The burgeoning telehealth industry, lauded for its potential to democratize healthcare access, is facing intensified scrutiny as companies push the boundaries of regulatory compliance, particularly with compounded medications. At the forefront of this emerging controversy is Medvi, a company recently elevated to "unicorn" status by a high-profile New York Times feature, which is now drawing sharp criticism for its business practices, including the marketing of a unique erectile dysfunction (ED) product called QUAD. This four-drug compounded cocktail, which combines three widely known PDE5 inhibitors with apomorphine – a medication primarily approved for Parkinson’s disease – is raising significant safety concerns among medical professionals and regulatory observers due to its unapproved nature, the inherent risks of drug interactions, and alleged flaws in its patient screening processes.
The Expanding Shadow Over a "Unicorn" Startup
From NYT Profile to Regulatory Backlash
Medvi, and its CEO Matthew Gallagher, were recently presented by The New York Times on April 2, 2026, as a "vibe-coded unicorn," a rapidly scaling AI-driven startup poised to revolutionize healthcare. However, the glowing profile quickly ignited a firestorm of critical reporting from investigative outlets such as Futurism, Forrester, Drug Discovery & Development, and Business Insider. These subsequent reports peeled back the layers of Medvi’s rapid ascent, revealing a pattern of regulatory challenges and questionable operational tactics.
Key revelations included an FDA warning letter issued in February 2026 for the misbranding of compounded GLP-1 weight-loss drugs – a distinct product line from QUAD but indicative of broader compliance issues. Further investigations uncovered a series of lawsuits against the company, a history of employing Meta advertisements featuring what appeared to be AI-generated "doctor" personas, and serious questions regarding the oversight of its extensive affiliate marketing network. These reports painted a picture of a company prioritizing aggressive growth and novel marketing strategies, potentially at the expense of rigorous medical and regulatory standards. The initial New York Times article, intended to highlight innovation, inadvertently became a catalyst for a deeper examination of Medvi’s operational integrity and the wider implications for the telehealth sector.
QUAD: A Four-Drug Formulation Raising Alarms

Unapproved Compounding and Risky Combinations
At the heart of the current controversy surrounding Medvi’s ED offerings is QUAD, a compounded product that combines an unprecedented array of active pharmaceutical ingredients (APIs) into a single sublingual dose. The formulation includes sildenafil, tadalafil, and vardenafil – all well-known PDE5 inhibitors commonly prescribed for ED – alongside apomorphine. It is crucial to understand that compounded drugs, by their very nature, are not FDA-approved. Unlike commercially manufactured pharmaceuticals, they do not undergo the rigorous premarket review process by the FDA to ensure their safety, effectiveness, or quality when used in combination or for off-label indications. This lack of oversight is particularly concerning given the powerful and potentially interacting nature of QUAD’s components.
The Peril of "Stacking" PDE5 Inhibitors
PDE5 inhibitors, such as sildenafil (Viagra), tadalafil (Cialis), and vardenafil (Levitra), function by increasing blood flow to the penis, facilitating an erection. While individually effective and FDA-approved, they carry known side effects and contraindications. For instance, these drugs can cause a decrease in blood pressure and are strictly contraindicated in patients taking nitrates due to the risk of severe, potentially life-threatening hypotension. They also require caution with alpha-blockers and certain blood pressure medications.
The practice of combining multiple PDE5 inhibitors, often referred to as "stacking," is explicitly warned against by regulatory bodies. The FDA’s labeling for sildenafil, for example, clearly states that combinations of PDE5 inhibitors "have not been studied" and "may further lower blood pressure," concluding unequivocally that "the use of such combinations is not recommended." Professional drug-interaction databases used by pharmacies and electronic health record systems universally flag therapeutic duplication warnings when more than one erectile-dysfunction agent is prescribed simultaneously, often recommending a maximum of one. QUAD, by its very name, implies four active ingredients, three of which are PDE5 inhibitors, directly contravening established medical and regulatory guidance. This synergistic action of multiple vasodilators dramatically escalates the risk of dangerous hypotensive episodes, syncope (fainting), and other cardiovascular complications for patients.
Apomorphine: A Parkinson’s Drug with a Troubled ED History
The inclusion of apomorphine in the QUAD formulation introduces another layer of significant concern. In the United States, apomorphine is FDA-approved exclusively for the treatment of motor fluctuations, or "OFF episodes," in adults with advanced Parkinson’s disease. Its veterinary use is even more distinct, serving as an emetic to induce vomiting in dogs.
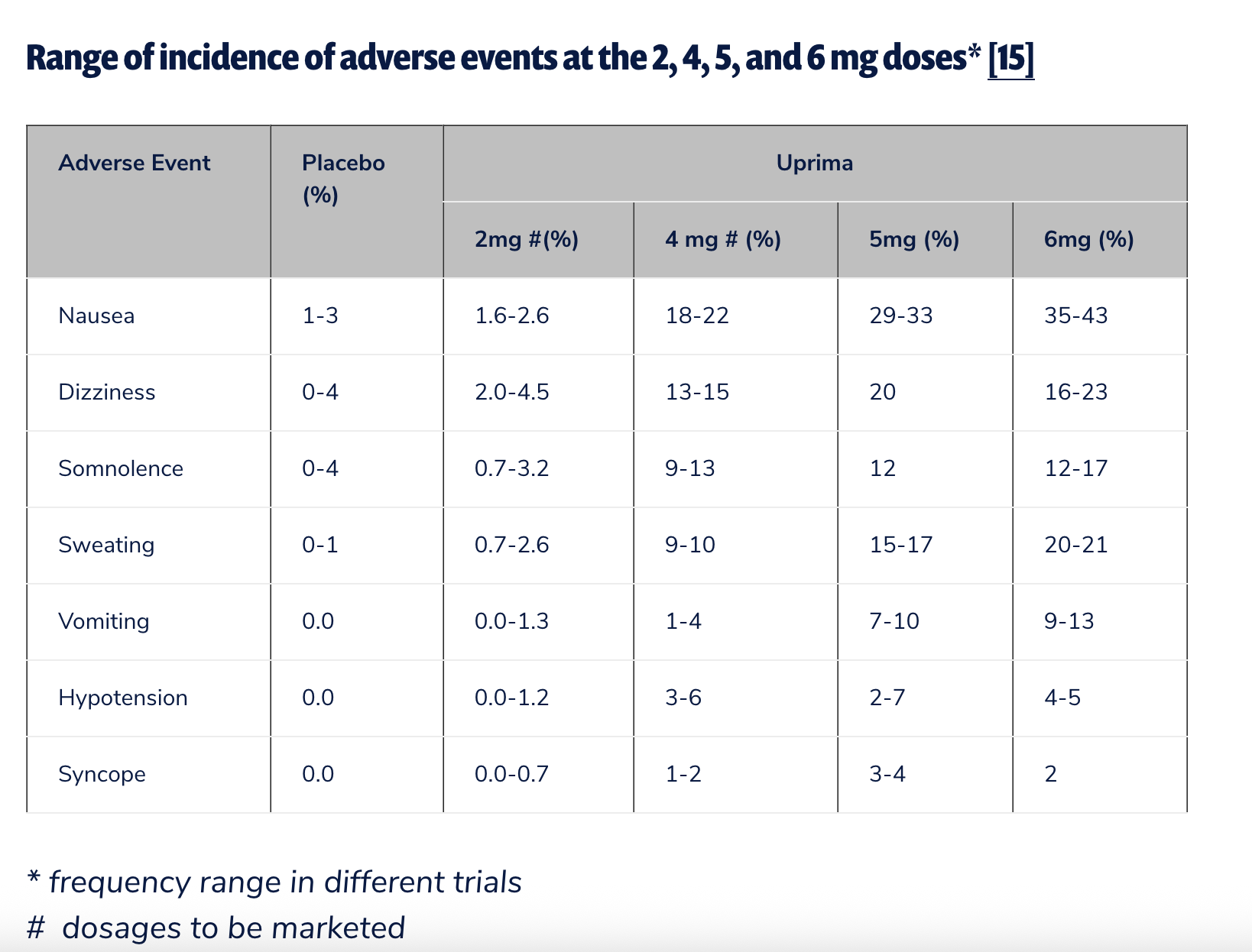
While apomorphine was marketed for erectile dysfunction in Europe starting in May 2001, its authorization expired in May 2006. Its then-developer, Abbott Laboratories, chose not to renew the marketing authorization, citing "commercial reasons." However, the decision was widely understood to be influenced by significant concerns regarding the drug’s efficacy and side effect profile. A notable UK study involving 11,185 patients found that a substantial two-thirds of men discontinued its use because they perceived it to be ineffective. Nausea was also reported as a fairly common side effect. A more recent 2020 systematic review and meta-analysis published in Archives of Sexual Behavior offered a somewhat more positive assessment, concluding that sublingual apomorphine was generally well tolerated only at lower doses (2 mg and 3 mg), acknowledging that discontinuation due to adverse events climbed significantly at higher doses.
The history of apomorphine for ED in the U.S. is even more fraught. In 2000, a joint venture between Abbott and Takeda, TAP Pharmaceuticals, sought FDA approval for sublingual apomorphine under the brand name Uprima. However, the application was withdrawn in June 2000 after an advisory committee expressed serious concerns about risks of hypotension and syncope. Public Citizen, a prominent consumer advocacy organization, played a critical role in opposing its approval. Sidney Wolfe, M.D., then director of Public Citizen’s Health Research Group, penned a blistering letter to then-CDER director Janet Woodcock, M.D., urging the FDA to reject the drug outright. Dr. Wolfe warned the agency that approval would inevitably lead to serious injuries and a near-certain post-market ban. He famously stated, "If the FDA approves this drug, it is certain that it will have to be banned within a short time after marketing begins because of the high probability of further occurrence of the serious injuries it has already been found to cause." FDA approval for ED in the U.S. never materialized, a testament to the profound safety concerns raised at the time.
The mechanism of apomorphine for ED is distinct from PDE5 inhibitors, acting as a central dopamine receptor agonist to facilitate erections. Combining a central nervous system-acting drug with multiple peripheral vasodilators in an unstudied formulation significantly increases the complexity and unpredictability of drug interactions, amplifying the risks of both central (e.g., nausea, dizziness) and peripheral (e.g., severe hypotension) adverse events.
Navigating the Regulatory Labyrinth of Compounding
FDA’s Stance on "Essentially a Copy" Products
The legality and appropriateness of Medvi’s QUAD product are further complicated by the FDA’s regulatory framework for compounded drugs. As previously noted, compounded drugs are not subject to the same pre-market approval process as commercially manufactured drugs. They are intended for individual patients with specific, documented clinical needs that cannot be met by an FDA-approved product.
In response to questions about compounded products, including those similar to QUAD, an HHS public affairs spokesperson referred Drug Discovery & Development to an April 1, 2026 FDA statement titled "FDA clarifies policies for compounders as national GLP-1 supply begins to stabilize." This document, published just a week before Drug Discovery & Development‘s testing of Medvi’s intake process, outlines critical FDA policy. It states that the FDA "intends to consider a compounded drug product to be essentially a copy of a commercially available drug product" when it "contains the same APIs as two or more commercially available drug products in the same, similar or easily substitutable strength." This is highly pertinent to QUAD, as sildenafil, tadalafil, and vardenafil – three of its four active ingredients – are each commercially available in the U.S. as FDA-approved products under brand names like Viagra, Cialis, and Levitra, as well as in generic versions.
The guidance does provide a narrow exception for cases where a prescriber documents a "significant clinical difference" for an identified individual patient, such as an allergy to a dye in a commercial product or the need for a liquid formulation for a patient unable to swallow pills. Furthermore, the FDA clarifies that it does not intend to take action against compounders making "essentially-a-copy" products if they fill "four or fewer prescriptions of that compounded drug product during a calendar month." This threshold is designed to accommodate genuine individualized compounding by a local pharmacy for specific, rare patient needs, a scenario starkly different from a large-scale telehealth operation like Medvi marketing a standardized "stack" to a broad consumer base. The volume implied by Medvi’s marketing and the rapid patient onboarding process suggests their operations far exceed this minimal threshold, placing their QUAD product squarely within the "essentially a copy" category without the requisite clinical justification.
Off-Label Use and Enforcement Challenges
The April 1 FDA document does not explicitly address the compounding of drugs for uses outside their FDA-approved indications. However, the regulatory landscape generally discourages and scrutinizes off-label compounding, especially when it involves drugs with known safety concerns for the intended off-label use, as is the case with apomorphine for ED. The HHS spokesperson, while declining to discuss specific compliance matters, reiterated that "when violations occur, the agency takes action as appropriate."
The challenge for the FDA lies in regulating the rapidly evolving telehealth sector, where companies can quickly scale and reach a national audience, blurring the lines between traditional pharmacy practice and mass-market drug distribution. The intent behind compounding pharmacy laws is to provide customized medications for unique patient needs, not to circumvent the drug approval process for combination products or off-label uses that lack robust safety and efficacy data. Medvi’s model, by offering a complex, unapproved combination of drugs, appears to stretch the boundaries of compounding’s intended purpose, raising critical questions about regulatory oversight in the digital age.
Medvi’s Marketing and Patient Intake: A Closer Look
Aggressive Promotion and Affiliate Disclosures
Medvi’s marketing for QUAD is characterized by aggressive claims and a direct comparison to established ED therapies. On its website, Medvi promotes apomorphine as a component that "ignites desires" and promises that QUAD "hits your system in 10 minutes" while offering a "36-hour response window." The company’s marketing materials feature head-to-head bar charts comparing QUAD by name to Viagra, Cialis, and Levitra, suggesting superior efficacy or convenience without robust clinical evidence for the combined product. This is further bolstered by terms like "Complete Stack," implying a comprehensive and potent solution.
Affiliate marketing pages further complicate the promotional landscape. An apparent affiliate marketing page titled "MEDVi QUAD Reviews: Is This Fast 4-in-1 ED Formula Worth the Hype?" states that users can "check your eligibility for MEDVi QUAD in minutes." While this page does include a vague disclaimer that the therapy "may not be suitable for individuals with certain medical conditions or those taking specific medications" and that "a clinician review is required to determine safety," it presents the product as a compelling option for men who have "already tried traditional ED therapies such as sildenafil or tadalafil individually and want something different." Crucially, the same page admits that the combined formulation "is not evaluated as a single product and is not FDA-approved as a single product," a disclosure that stands in stark contrast to the aggressive marketing claims.
Another affiliate page, posted on MedicalFoundationofNC.org, dated April 4, 2026, and updated April 6, 2026, attempts to distance Medvi’s ED therapies from the ongoing regulatory scrutiny. It asserts that "the FDA warning letter and the GLP-1 regulatory crackdown are about a different product line" and specifically states that the February 2026 warning letter "did not reference MEDVi’s QUAD ED product line." This page, however, also contains extensive disclaimers, advising readers to "ask hard questions before enrolling," disclosing potential affiliate commissions, stating it is "not medical advice," and clarifying that MedicalFoundationofNC.org is "not a medical practice, pharmacy, or licensed healthcare provider" and "not affiliated with The Medical Foundation of North Carolina, Inc., UNC Health Foundation, UNC Health Care, the UNC School of Medicine, or the University of North Carolina at Chapel Hill." This intricate web of promotion and disclaimers highlights the challenges in discerning credible medical information from marketing hype in the digital space.
Critical Flaws in Patient Screening
Beyond the marketing, Medvi’s patient intake and screening processes for QUAD, as documented by Drug Discovery & Development during test runs on April 7, 2026, reveal significant and alarming flaws that directly jeopardize patient safety.
One critical issue emerged when the intake questionnaire asked users if they had been "diagnosed with high blood pressure (hypertension) or low blood pressure (hypotension)." Despite explicitly mentioning both conditions, the system offered only two answer cards, both referring to "high blood pressure." There was no option for "low blood pressure" (hypotension), a condition that, as previously discussed, is a major contraindication for PDE5 inhibitors and apomorphine due to the severe risk of additive hypotensive effects. In the test runs, selecting "Yes, I have high blood pressure" correctly halted the intake with an "Eligibility Status" message indicating that the assessment could not be completed. However, the absence of an off-ramp for hypotension means that individuals with dangerously low blood pressure – a population at extremely high risk for adverse events from QUAD – could potentially proceed through the screening process unchecked. This is a profound patient safety loophole that could lead to serious medical incidents.
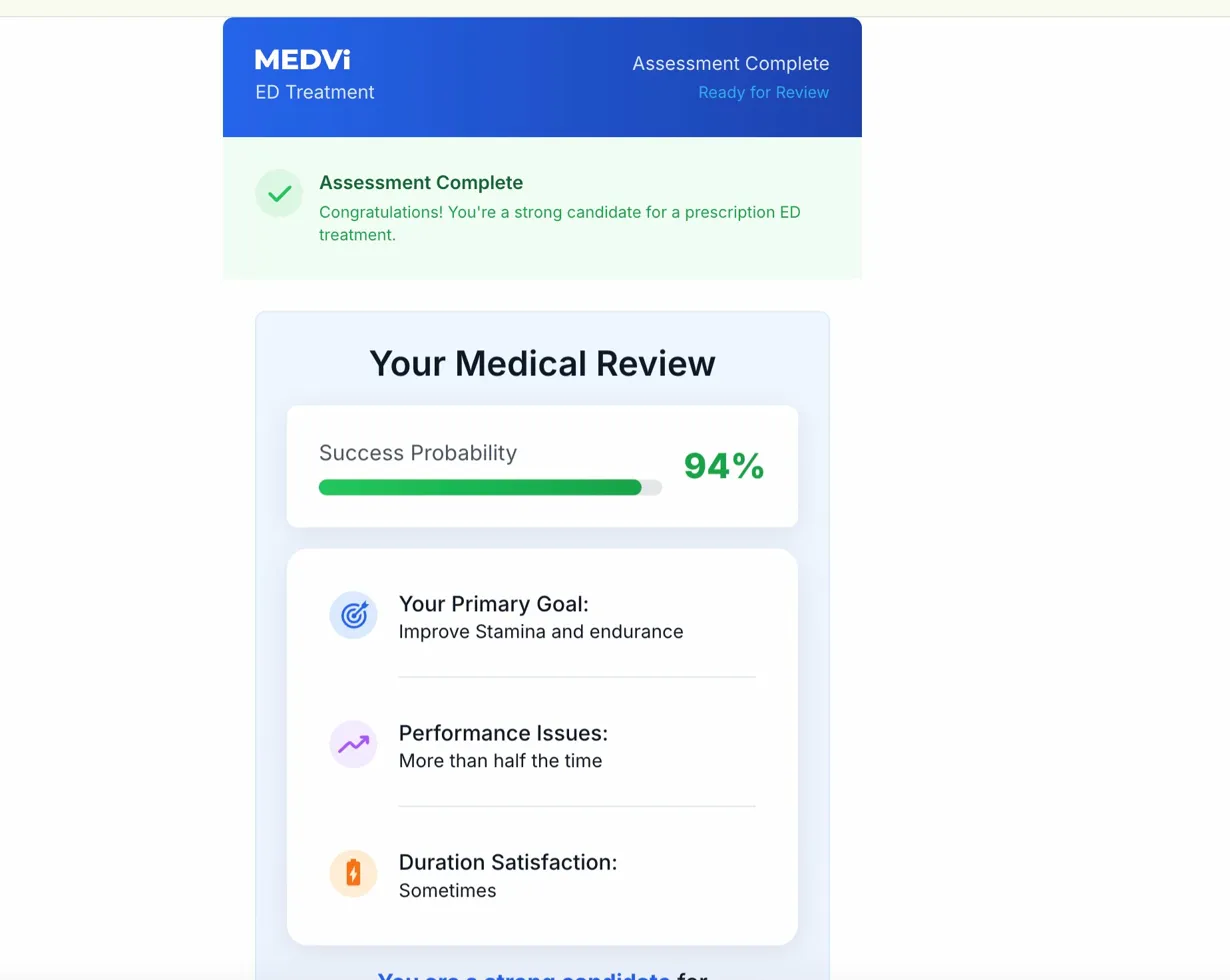
Furthermore, the onboarding portal displayed a "Your Medical Review" screen showing a "Success Probability" of 94% for users not screened out from QUAD. This exact 94% figure has been observed in Medvi’s GLP-1 weight-loss intake process and is preserved in a screen recording filed as an exhibit in Day v. OpenLoop Health, a federal class action lawsuit in Delaware (where Medvi is mentioned but not a defendant). Across at least three QUAD intake test runs conducted with varied answer combinations, the 94% figure remained constant. This static, seemingly arbitrary success rate raises ethical concerns, as it suggests a lack of personalized assessment and could be misleading to patients, creating a false sense of assured efficacy regardless of individual medical profiles or the inherent risks of an unapproved, multi-drug combination.
Broader Implications: Patient Safety, Regulatory Oversight, and Telehealth Ethics

Unacceptable Patient Safety Risks
The most pressing implication of Medvi’s QUAD offering and its associated practices is the unacceptable risk posed to patient safety. The combination of three PDE5 inhibitors with apomorphine, a drug with its own history of efficacy and safety concerns for ED, creates a pharmacologically complex and potentially dangerous cocktail. The additive hypotensive effects, explicitly warned against by the FDA and professional drug databases, coupled with the documented flaws in Medvi’s patient screening (particularly the failure to adequately screen for hypotension), form a critical safety gap. Patients, unaware of the compounded nature of the drug and relying on aggressive marketing claims and a potentially misleading "success probability," could be exposed to severe adverse events, including profound drops in blood pressure, fainting, and other serious cardiovascular complications, without adequate medical oversight or pre-screening.
The Future of Telehealth Regulation
Medvi’s case underscores the formidable challenges faced by regulatory bodies like the FDA in keeping pace with the rapid innovation and expansion of the telehealth industry and the evolving landscape of compounded medications. The existing regulatory framework for compounding was designed for individualized prescriptions by local pharmacies, not for the mass-market distribution of standardized, unapproved combination products via online platforms. There is an urgent need for clearer, more robust guidelines and proactive enforcement mechanisms to address off-label use, the "stacking" of drugs, and the ethical marketing and screening practices of telehealth providers. The current ambiguities and enforcement lags create a grey area that companies like Medvi appear to be exploiting, potentially compromising public health.
Ethical Imperatives for Online Healthcare Platforms
Beyond regulatory compliance, Medvi’s practices raise profound ethical questions for the entire online healthcare sector. Telehealth platforms, while offering convenience, have an inherent responsibility to prioritize patient safety, ensure transparent communication about the nature of the medications they prescribe (e.g., compounded vs. FDA-approved), and implement rigorous, medically sound patient screening processes. The use of AI in medical assessments, while promising, must be meticulously validated and subject to stringent ethical oversight to prevent biases, errors, and the dissemination of misleading information like a fixed "success probability." The commercial pressures to scale rapidly must not supersede the fundamental ethical duty to provide safe, effective, and responsible medical care.
Conclusion: A Call for Scrutiny
The unfolding narrative around Medvi and its QUAD product serves as a potent reminder of the critical balance between innovation and regulation in healthcare. The company’s marketing of an unapproved, multi-drug erectile dysfunction cocktail, featuring a Parkinson’s drug with a checkered past for ED, in conjunction with potentially dangerous drug interactions and demonstrably flawed patient screening, demands immediate and thorough regulatory intervention. The combination of these factors – an unapproved product, highly concerning drug interactions, aggressive and potentially misleading marketing, and inadequate patient safety mechanisms – paints a troubling picture that necessitates urgent attention from both regulatory authorities and the broader public to safeguard patient well-being and uphold the integrity of online healthcare.















Leave a Reply