The narrative of MEDVi, an AI-powered telehealth startup, recently unfolded with a stark duality: hailed by The New York Times as a trailblazer fulfilling Sam Altman’s vision for AI-driven billion-dollar enterprises, while simultaneously grappling with a barrage of regulatory warnings, allegations of deceptive marketing, and multiple lawsuits. This complex saga underscores the rapidly evolving landscape of artificial intelligence in healthcare, highlighting both its disruptive potential and the profound ethical and legal challenges it introduces, particularly in a highly regulated sector like pharmaceuticals.
The Genesis of an AI Unicorn: Altman’s Prediction and Gallagher’s Ambition
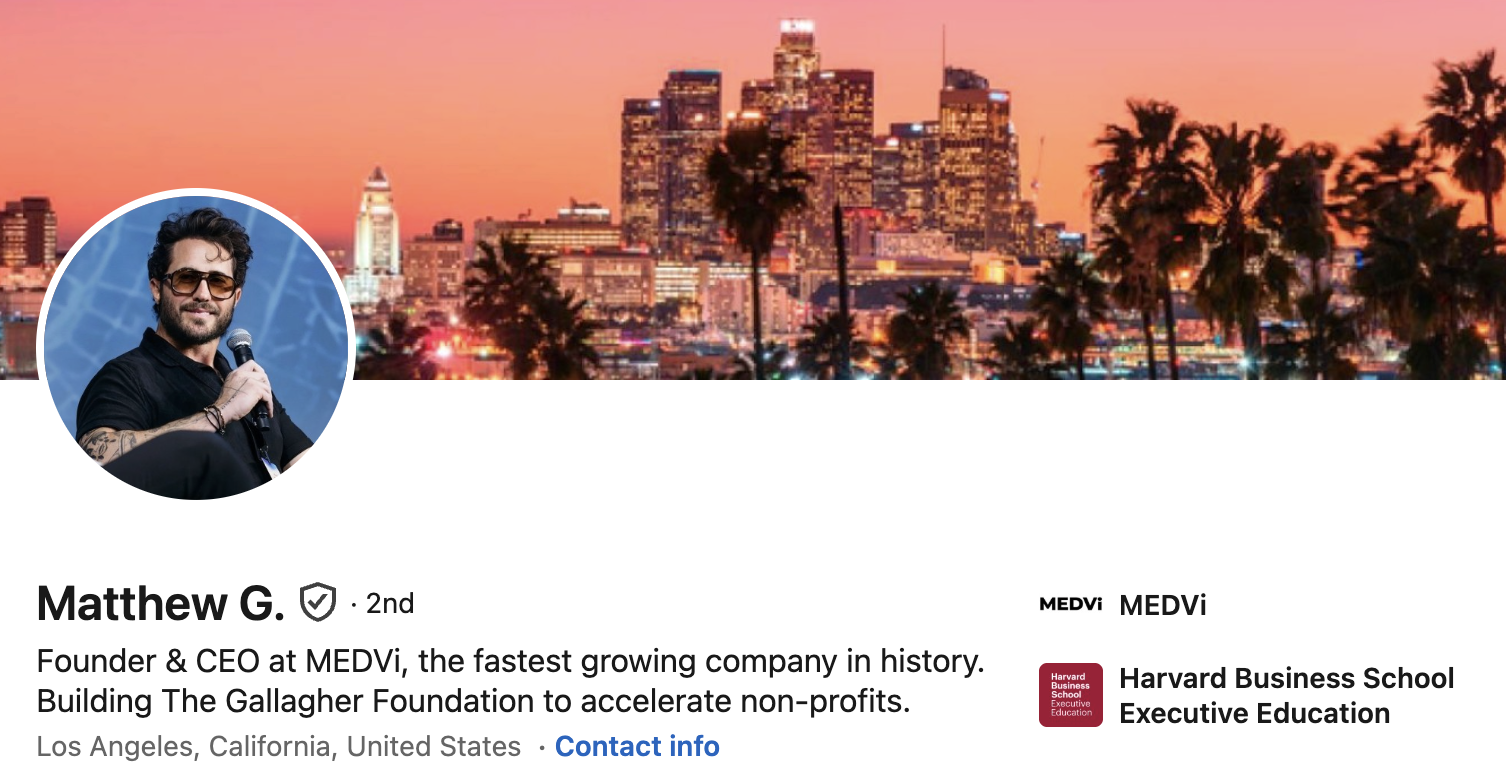
In 2024, OpenAI CEO Sam Altman made a bold prediction: a single entrepreneur, leveraging the power of artificial intelligence, would soon create a business valued at over one billion dollars. Matthew Gallagher, an entrepreneur based in Los Angeles, seemingly positioned his telehealth startup, MEDVi, as one of the earliest success stories to validate Altman’s prophecy. Gallagher, who previously boasted on LinkedIn about MEDVi being "the fastest growing company in history" (a claim he has since removed), garnered significant attention from The New York Times.
On April 2, 2026, the prestigious publication profiled MEDVi as a prime example of an AI-fueled success story, detailing Gallagher’s projected path to an astounding $1.8 billion in sales by 2026. What made this projection particularly remarkable was the lean operational structure: MEDVi claimed only two full-time employees—Gallagher himself and his brother, Elliot—supplemented by contract engineers, account managers, and external agencies. The Times reported that it was granted access to MEDVi’s financial records to corroborate these impressive revenue and profit figures.

Gallagher detailed his reliance on a suite of AI services, including ChatGPT, Claude, Grok, MidJourney, and Runway, for the company’s foundational development. He outsourced critical medical and logistical functions, entrusting CareValidate, a "telehealth-in-a-box" platform, and OpenLoop Health with the management of doctors, pharmacies, shipping, and regulatory compliance. This model, often referred to as white-label telemedicine, allows companies to rapidly scale by leveraging existing infrastructure, a strategy Gallagher defended on social media, equating it to "drop shipping" and praising its potential to democratize healthcare access.
A Storm on the Horizon: The FDA’s Early Warning
Even as MEDVi was being lauded for its innovative approach, regulatory alarms had already been sounded. Six weeks prior to The New York Times profile, on February 20, 2026, the U.S. Food and Drug Administration (FDA) issued a warning letter to MEDVi, LLC, citing significant concerns regarding the misbranding of the compounded drugs that constituted a substantial portion of its revenue.
The FDA’s letter specifically admonished MEDVi for creating a false impression that it was the direct compounder of the semaglutide and tirzepatide it sold. Furthermore, the agency took issue with explicit claims on MEDVi’s website, such as "Same active ingredient as Wegovy® and Ozempic®" and "Same active ingredient as Mounjaro® and Zepbound®." The FDA deemed these statements to falsely imply that the compounded versions had received official FDA approval or evaluation, which they had not. The warning stipulated that failure to rectify these violations could lead to severe enforcement actions, including product seizure or injunctions. Crucially, the FDA noted that the cited violations were not exhaustive, suggesting that further issues might exist.
MEDVi, in an April 8 statement, attempted to deflect the warning, claiming the FDA-cited website (medvi.io) belonged to an affiliate marketing agency and asserting that MEDVi itself had "never received a letter from the FDA." However, the FDA’s warning letter was explicitly addressed to "MEDVi, LLC dba MEDVi" at its registered Delaware address, utilized MEDVi’s corporate email address, and directly referenced the problematic, MEDVi-branded claims found on medvi.io. This direct evidence contradicted MEDVi’s attempt to distance itself from the regulatory action.
The Unmasking of Deception: AI-Generated Personas and Dubious Marketing
Concurrent with the FDA’s actions, independent journalistic investigations began to uncover a pattern of highly questionable marketing practices employed by MEDVi and its affiliates. A review conducted by Drug Discovery & Development on April 3, 2026, revealed widespread use of apparent AI-generated personas, some even presented with medical titles, across MEDVi’s website and Facebook advertising.
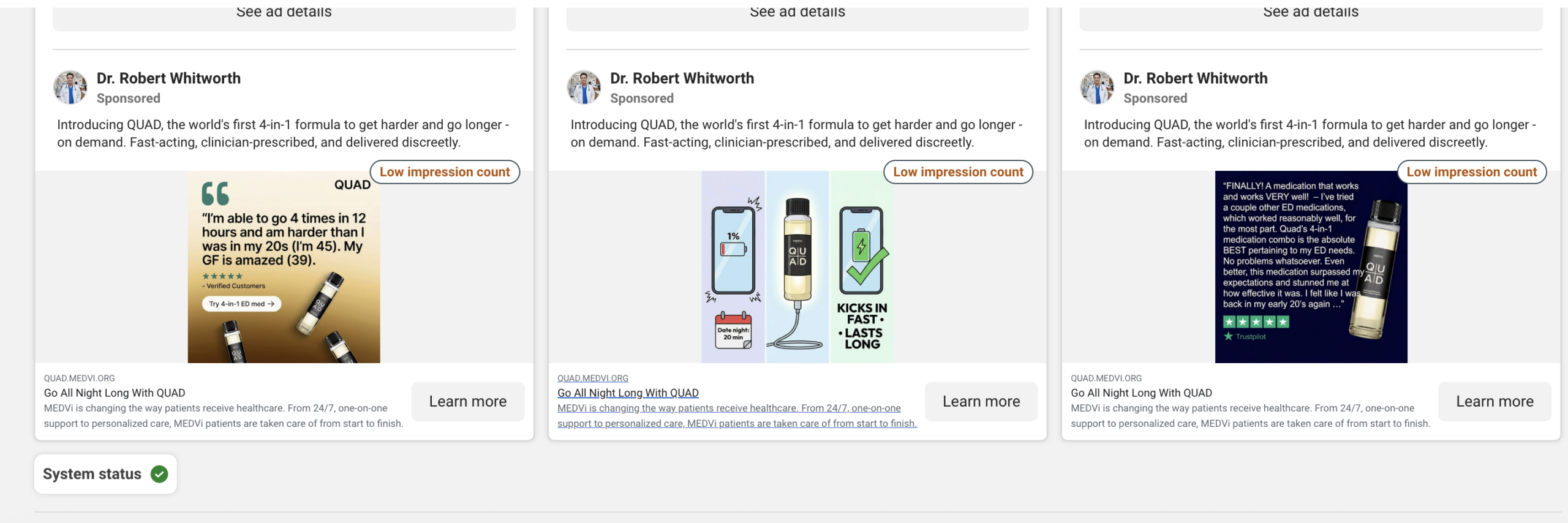
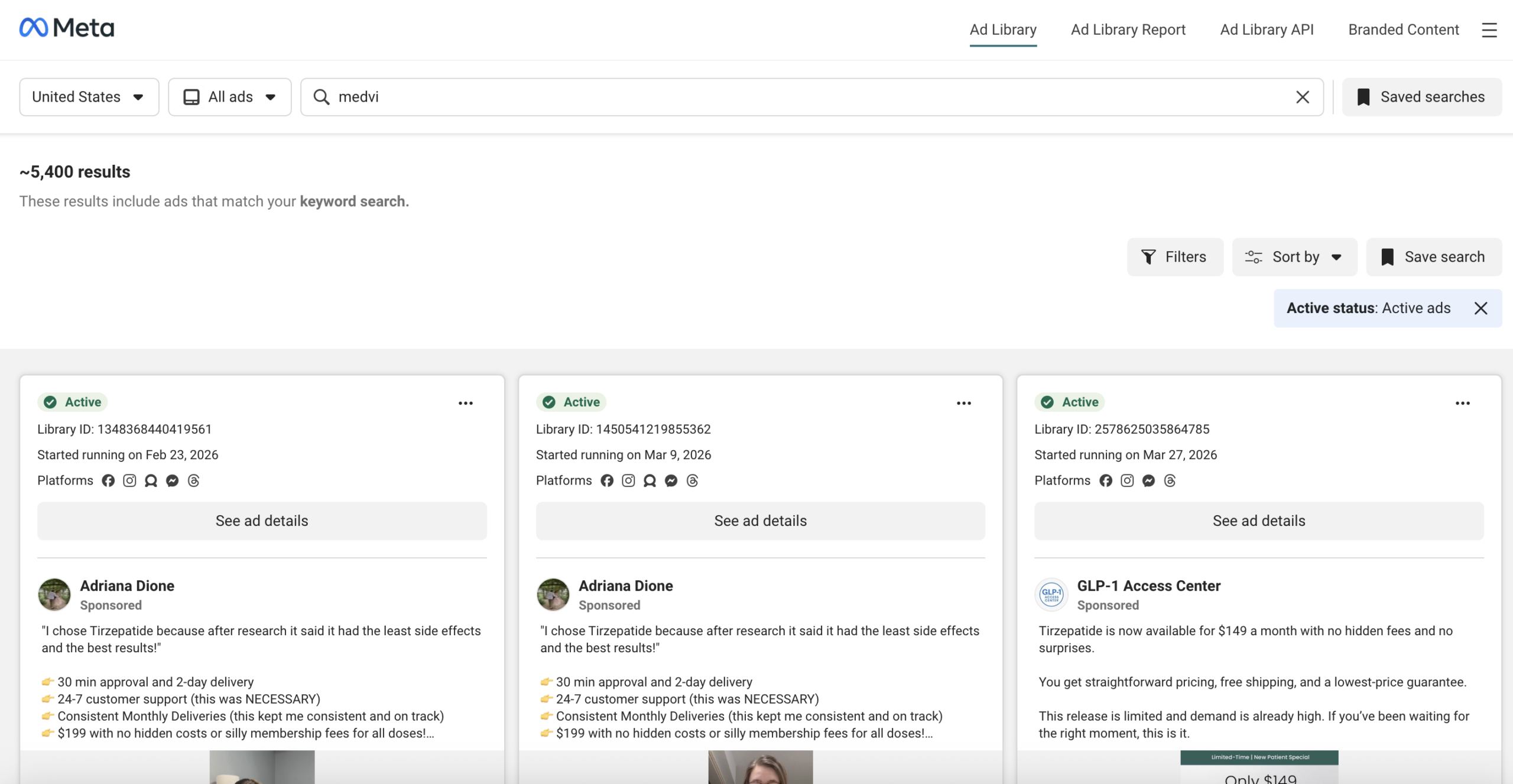
A search of Meta’s Ad Library for "medvi" yielded over 5,000 active advertisements, many operating under what appeared to be fabricated physician personas. For instance, a Facebook page for "Dr. Robert Whitworth," which sponsored ads for MEDVi’s QUAD erectile dysfunction product, was bizarrely categorized as an "Entertainment website" and listed a non-existent address in Cameron, MT. Other ads were attributed to fantastically named individuals like "Professor Albust Dongledore" and "Dr. Richard Hörzgock," featuring AI-generated video testimonials and recycling identical scripts across multiple fabricated identities. In some egregious cases, a doctor’s headshot would be displayed on the page, while the accompanying ad featured an entirely different person delivering a patient testimonial. Following the initial journalistic inquiries, many of these seemingly fabricated doctor personas were reportedly removed from the site by April 6.
A Business Insider investigation published on the same day further corroborated these findings, scrutinizing the historical use of AI-generated doctor profiles in Meta ads for MEDVi, often linked to affiliate marketers. Gallagher informed Business Insider that approximately "30%" of MEDVi’s advertising was conducted through affiliates. Business Insider identified profiles such as "Dr. Matthew Anderson MD," linked to an Angolan phone number and a gospel musician, and "Dr. Spencer Langford MD," associated with a clothing store in the Republic of Congo. Many profiles exhibited tell-tale signs of AI generation, including garbled text and Gemini watermarks. Following Business Insider’s inquiries, the number of active Medvi-related ads dropped from over 5,000 to roughly 2,800.
Gallagher, in response to Business Insider, stated, "In line with the FTC, we have a clear policy of providing disclosure on any actor or AI portrayal of a doctor or not using them at all. If we find an affiliate doing this we work to take these ads down." However, this statement appeared to contradict his earlier social media posts where he mocked critics and equated the use of AI-generated physician imagery to standard medical stock photography, suggesting an awareness of the practice that predated his claims of "recently becoming aware." Nancy Glick, Director of Food, Nutrition, and Obesity at the National Consumers League, a prominent consumer advocacy group that has previously warned against unregulated GLP-1 products, voiced strong criticism of MEDVi’s practices to Business Insider.

MEDVi’s own website contained a subtle, yet telling, disclaimer at the bottom: "Individuals appearing in advertisements may be actors or AI portraying doctors and are not licensed medical professionals." This fine print, while technically a disclosure, often goes unnoticed by consumers and stands in stark contrast to the overt portrayal of medical authority in the advertisements themselves.
Earlier in May 2025, Futurism had already documented similar issues with MEDVi’s website, including AI-generated patient photos, deepfaked before-and-after weight loss images (traced to photos with faces swapped by AI), and AI-generated Ozempic box imagery containing clear errors. At that time, logos from The New York Times, Bloomberg, and Forbes were displayed, implying editorial coverage that had not yet occurred. When Futurism contacted doctors listed on MEDVi’s site, at least one, osteopathic practitioner Tzvi Doron, confirmed he had no association with the company and demanded his removal. Gallagher later acknowledged in The New York Times profile that MEDVi’s initial site had indeed featured AI-generated photos and deepfaked before-and-after weight loss images.
Furthermore, MEDVi’s site had previously listed two physicians, Dr. Ana Lisa Carr and Dr. Kelly Tenbrink, both associated with Ringside Health, a concierge practice in Wellington, Florida. MEDVi’s site did not clarify their affiliation. While Dr. Tenbrink was listed as certified by the American Board of Emergency Medicine, and an ABEM credential page confirmed this, the Florida Department of Health practitioner profiles for both physicians indicated that neither held specialty board certifications recognized by the Florida board. On April 10, following inquiries from Drug Discovery & Development, MEDVi entirely removed both Dr. Carr and Dr. Tenbrink from its website.
The Regulatory Tightrope: Compounded GLP-1s and the "Regulatory Gap"
MEDVi’s business model heavily relies on navigating a complex and narrowing regulatory environment surrounding compounded drugs, particularly GLP-1s. Federal law generally prohibits compounders from creating copies of FDA-approved drugs. However, Sections 503A and 503B of the Federal Food, Drug, and Cosmetic Act permit compounding under specific circumstances, such as during official drug shortages.

The FDA had officially declared the shortage of tirzepatide injection resolved on December 19, 2024, and the semaglutide injection shortage resolved on February 21, 2025. This resolution significantly impacts the legality of compounding these specific drugs. In response, many sellers, including those potentially associated with MEDVi, pivoted to offering "personalized" formulations, often adding ingredients like vitamin B-12, to argue that their products were not "essentially a copy" of the FDA-approved versions.
However, the FDA clarified its stance on April 1, 2026, stating that a compounded product combining semaglutide with another active ingredient, such as vitamin B-12, could still be deemed "essentially a copy" unless a prescriber could document a patient-specific "significant difference" justifying the unique formulation. Pharmaceutical giant Eli Lilly, the manufacturer of Mounjaro and Zepbound (tirzepatide), explicitly warned against the practice of compounding tirzepatide with vitamin B-12 in March 2026, citing potential safety and efficacy concerns. This tightening regulatory environment poses a direct threat to the core product offerings of companies like MEDVi.
Mounting Legal Challenges: Data Breaches and "Snake Oil" Allegations
Beyond regulatory warnings, MEDVi and its partners face a growing wave of legal challenges. MEDVi reported an impressive 250,000 customers by the end of 2025, a number Gallagher described to the Times as "insane" and accelerating. Its homepage now boasts "500,000+ MEDVi patients," promising "unlimited 24/7 support" and "doctor-led plans & coaching." The company’s reliance on OpenLoop Health clinicians, who "retain the decision to prescribe compounded GLP-1s to patients," links it directly to a major data security incident.
OpenLoop Health, a Des Moines, Iowa-based telehealth infrastructure company, disclosed a significant cyber breach on January 7, 2026. A threat actor reportedly breached OpenLoop’s systems and claimed to have exfiltrated records from approximately 1.6 million patients, encompassing sensitive data such as names, contact information, dates of birth, and medical information. OpenLoop notified the Texas Attorney General in March 2026, confirming at least 68,160 affected individuals in that state alone, and now faces multiple class-action lawsuits.
Adding to the legal woes, a North Carolina consumer filed a class-action complaint in the U.S. District Court for the District of Delaware in November 2025 against OpenLoop Health and compounding pharmacy Triad Rx. This lawsuit, first disclosed by Fierce Healthcare, alleges that the firms manufactured and sold "modern-day snake oil": compounded oral tirzepatide tablets with "no demonstrated mechanism of absorption or efficacy." While MEDVi is not explicitly named as a defendant, the complaint asserts that the plaintiff purchased these pills through MEDVi, which it describes as one of dozens of consumer-facing telehealth storefronts operating within a network managed by OpenLoop.
The scientific basis of the allegation is critical: tirzepatide, a large peptide molecule, is typically destroyed by digestive enzymes before it can enter the bloodstream. The only FDA-approved oral GLP-1, Rybelsus (semaglutide), requires a specialized absorption enhancer called SNAC to achieve a mere 1% bioavailability, and even then, it must be taken under strictly controlled conditions. The complaint thus raises serious questions about the fundamental efficacy and safety of the oral tirzepatide products being marketed.
The lawsuit brings claims under the Racketeer Influenced and Corrupt Organizations Act (RICO Act), state consumer protection statutes, and common law fraud, highlighting the severity of the alleged misconduct. It paints a picture of MEDVi as one of at least a dozen nearly identical telehealth storefronts, all marketing the same product through a shared backend infrastructure, and using a "Meet the incredible doctors we’ve partnered with" presentation as part of a broader marketing scheme.
MEDVi itself has also been involved in direct litigation. It was served and subsequently defaulted in Siuksta v. MEDVi, LLC, a federal Telephone Consumer Protection Act (TCPA) lawsuit filed in May 2025, which was later voluntarily dismissed. More recently, James v. Medvi LLC (No. 8:26-cv-00641) was filed in the Central District of California on March 20, 2026. This class-action case accuses MEDVi of benefiting from affiliate spam, citing an email with the subject line "This might be the easiest way to start Ozempic" sent from a nonsensical email address, which routed to a MEDVi landing page with tracking parameters. Filed under California’s anti-spam law, the suit seeks statutory damages of $1,000 per violating email, with an estimated class size of at least 100,000 individuals.
The CEO’s Social Media Counteroffensive and Industry Skepticism
In the wake of The New York Times profile and the subsequent critical feedback, Matthew Gallagher took to social media to defend MEDVi. In one deleted X post, he derided detractors as "Karens of the internet" with "low testosterone." In another notable exchange, he pushed back against Jon Oringer, founder of Shutterstock, who had sarcastically proposed a "fake-doctor affiliate-as-a-service platform." Gallagher equated MEDVi’s AI-generated physician personas to standard medical stock art used in advertising, writing: "The guy who SELLS images of doctors to marketers pretends not to understand marketing. The irony is beautiful," accompanied by a screenshot of a generic stock doctor image. This public posture directly contradicted MEDVi’s April 8 statement claiming they had only "recently become aware" of ads featuring "potentially AI-generated medical practitioners."
On April 3, 2026, Gallagher posted a more expansive defense of the white-label telemedicine model on LinkedIn, portraying critics as unsophisticated and ignorant of modern business practices: "Watching in real-time as people learn about white label, drop shipping, and affiliate marketing is like seeing cavemen ‘fire bad.’ White label telemedicine is a huge benefit with a net positive for humanity. It has been the driving force behind big pharma lowering prices and making healthcare accessible to everyone from home. Low energy people think offering life-changing weight loss medication, prescribed by a doctor, is a trendy ‘pill mill.’ Wait until you see what’s possible with longevity services and peptides. I’m bullish on humanity. If you are too, I’ll help you build."
While some on LinkedIn congratulated him on his AI-first execution and philanthropic aspirations, others voiced skepticism. An SEO professional noted that MEDVi’s reported website traffic seemed insufficient to support the claimed user base and revenue. A digital health founder commented, "Something doesn’t add up," and an accountant recommended an independent audit. A viral YouTube video titled "billion dollar ai company was built on lies" garnered over a million views within three days, and several alleged customers posted direct complaints.
Drug Discovery & Development reached out to MEDVi, Matthew Gallagher, OpenLoop Health, Dr. Kelly Tenbrink, and Dr. Ana Lisa Carr for comment but did not receive an immediate response. Nicholas Chimicles, lead counsel for the plaintiffs in one of the class-action lawsuits, declined further comment on the litigation but confirmed awareness of The New York Times profile of MEDVi.
Broader Implications for AI, Telehealth, and Consumer Protection

The MEDVi saga serves as a critical case study at the intersection of technological innovation, healthcare, and consumer protection. It highlights the immense potential of AI to drive efficiency and scale, but also exposes the significant risks when ethical boundaries are blurred, and regulatory compliance is neglected. The rapid deployment of AI in marketing, particularly the creation of deceptive personas, presents a formidable challenge for platforms like Meta, which have been criticized for profiting from fraudulent advertisements. A bipartisan coalition of 35 attorneys general had already written to Meta in December 2025, urging the company to crack down on misleading, AI-generated weight-loss ads, noting that "The use of artificial intelligence to fabricate images, spokespeople, and medical claims crosses a line and makes these ads particularly dangerous." Reuters also reported in late 2025 that Meta was "earning a fortune on a deluge of fraudulent ads," with internal documents suggesting that one-tenth of its 2024 revenue came from "scams and banned goods."
For the telehealth industry, the controversies surrounding MEDVi could erode public trust, making it harder for legitimate, compliant companies to thrive. Consumers, lured by enticing AI-generated advertisements and promises of easy access to popular medications, face risks ranging from ineffective products to privacy breaches and potential health complications from unregulated or misbranded drugs.
The legal actions, particularly the RICO Act claims and class-action lawsuits, could set important precedents for accountability in the rapidly evolving digital health space. They underscore the need for greater transparency from companies leveraging AI and white-label services, and robust enforcement from regulatory bodies like the FDA and FTC to ensure patient safety and prevent consumer fraud. The MEDVi narrative is far from over, but it has already sparked crucial conversations about responsible innovation, ethical marketing, and the imperative for robust oversight in the age of AI-driven enterprise.














Leave a Reply