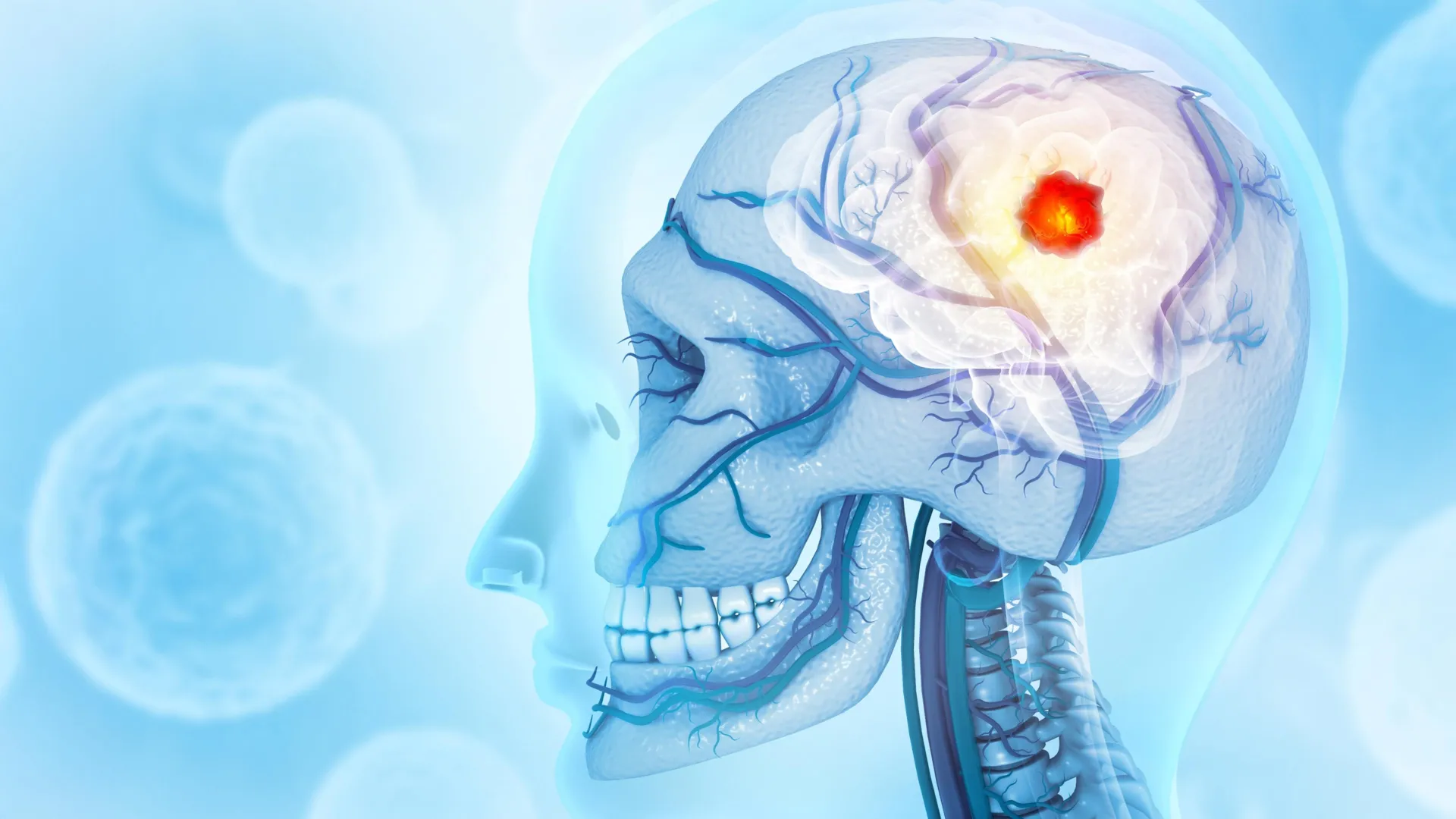
Researchers from the Montefiore Einstein Comprehensive Cancer Center (MECCC) and the Albert Einstein College of Medicine have fundamentally challenged the long-standing medical consensus that glioblastoma is a localized neurological malignancy. In a landmark study published on October 3 in the journal Nature Neuroscience, a multidisciplinary team of scientists revealed that glioblastoma—the most aggressive and lethal form of primary brain cancer—exerts a systemic influence that extends far beyond the soft tissues of the brain. The study demonstrates that these tumors actively degrade the structural integrity of the skull, hijack the bone marrow’s immune-cell production, and effectively neutralize the body’s natural defenses, providing a potential explanation for why traditional therapies have remained largely ineffective for decades.
The findings indicate that glioblastoma induces a state of "systemic subversion," where the tumor communicates with the skull bone marrow through microscopic channels to recruit pro-inflammatory cells that facilitate tumor growth. Furthermore, the research sounds a cautionary note regarding the use of common bone-density medications, such as those used to treat osteoporosis, suggesting that these drugs may inadvertently accelerate the progression of the disease or interfere with emerging immunotherapies.
The Clinical Challenge of Glioblastoma Multiforme
Glioblastoma multiforme (GBM) has long been regarded as one of the most formidable challenges in modern oncology. According to the National Cancer Institute (NCI), approximately 15,000 individuals in the United States are diagnosed with this condition annually. Despite advancements in surgical techniques, precision radiation, and specialized chemotherapies like temozolomide, the prognosis for patients remains grim. The median survival time for those receiving the current standard of care is approximately 15 months, a statistic that has seen little improvement over the last twenty years.
The difficulty in treating GBM has traditionally been attributed to the blood-brain barrier, which prevents many drugs from reaching the tumor, and the highly infiltrative nature of the cancer cells, which makes complete surgical resection nearly impossible. However, the new data from MECCC and Albert Einstein College of Medicine suggests that the failure of current treatments may also stem from a fundamental misunderstanding of the tumor’s environment. By viewing glioblastoma as a "local" disease confined to the brain, clinicians may have been overlooking a critical "skull-brain axis" that the tumor exploits to survive and thrive.
Discovery of the Skull-Brain Communication Axis
The research team, led by corresponding author Jinan Behnan, Ph.D., an assistant professor in the Leo M. Davidoff Department of Neurological Surgery and the Department of Microbiology & Immunology at Einstein, was inspired by recent anatomical discoveries. Prior studies had identified the existence of extremely thin, microscopic channels connecting the skull’s bone marrow directly to the meninges—the protective layers surrounding the brain. These channels allow for a bidirectional exchange of molecular signals and immune cells.
Utilizing advanced imaging technologies and mouse models of two distinct types of glioblastoma, Dr. Behnan’s team observed a startling phenomenon: the presence of the tumor triggered significant erosion of the skull bone. This degradation was most pronounced along the cranial sutures—the fibrous joints where the various bones of the skull fuse together.
Crucially, the researchers determined that this bone loss was a specific pathological signature of glioblastoma and other highly aggressive brain tumors. When the team examined mice that had suffered strokes, traumatic brain injuries, or had developed cancers in other parts of the body, they found no evidence of similar skull erosion. This suggests that glioblastoma possesses a unique mechanism for "remodeling" the bone that houses it. To validate these findings in a clinical context, the team reviewed CT scans of human glioblastoma patients. The results confirmed that human patients exhibited the same localized thinning of the skull in regions corresponding to the tumor’s location, mirroring the observations in the lab.
The Hijacking of Skull Marrow and Immune Dysregulation
The structural erosion of the skull is not merely a side effect of the cancer; it serves a functional purpose for the tumor’s survival. The research found that as the skull bone erodes, the microscopic channels between the bone marrow and the brain increase in both size and number. This "widening of the gates" allows the tumor to send molecular distress signals into the skull marrow, effectively "reprogramming" the immune cell production facilities located there.
Using single-cell RNA sequencing—a high-resolution technique that allows scientists to see which genes are active in individual cells—the researchers analyzed the immune landscape of the skull marrow. They discovered a profound shift in the types of cells being produced. In the presence of glioblastoma, the marrow nearly doubled its production of pro-inflammatory myeloid cells, specifically neutrophils. These cells, while normally part of the body’s first line of defense, are often co-opted by tumors to create a "suppressive" environment that protects the cancer from the immune system.
Simultaneously, the marrow’s production of beneficial immune cells plummeted. The researchers observed the near-total elimination of various types of B cells, which are responsible for producing antibodies. This creates a "tilt toward inflammation" that favors the tumor while disarming the body’s adaptive immune response.
"The skull-to-brain channels allow an influx of these numerous pro-inflammatory cells from the skull marrow to the tumor, rendering the glioblastoma increasingly aggressive and, all too often, untreatable," explained study co-author E. Richard Stanley, Ph.D., a professor of developmental and molecular biology at Einstein.
Systemic Disruption: Skull vs. Femur Marrow
One of the most significant aspects of the study is the evidence that glioblastoma affects bone marrow differently depending on its proximity to the tumor. While the skull marrow was pushed into a state of hyper-inflammation, the marrow in the femur (the thigh bone) reacted in the opposite manner. In the femur, the cancer suppressed the genes necessary for producing various immune cells, leading to a state of systemic immune exhaustion.
This distinction is vital for future research. It proves that glioblastoma does not just send out a general signal to the whole body; it engages in a sophisticated, localized manipulation of the skull marrow while simultaneously dampening the rest of the body’s immune reservoir. This dual-action strategy makes the cancer exceptionally resilient to systemic therapies.
The Paradox of Osteoporosis Medications
Perhaps the most clinically urgent finding of the study involves the use of anti-osteoporosis drugs. Given that the tumor causes bone loss, the researchers hypothesized that preventing this erosion might slow the disease. They tested two FDA-approved drugs: zoledronic acid and denosumab.
While both medications successfully halted the erosion of the skull bone, the results regarding the cancer itself were unexpected and concerning. In one type of glioblastoma, zoledronic acid actually accelerated tumor progression. Furthermore, both drugs were found to block the efficacy of anti-PD-L1, a prominent immunotherapy drug designed to boost tumor-fighting T cells.
This suggests that the relationship between bone health and brain cancer is highly complex. While bone erosion is a symptom of the disease, simply stopping the erosion with current medications may interfere with the immune signaling required for other treatments to work. This finding may require oncologists to rethink how they manage bone health in brain cancer patients, particularly those enrolled in immunotherapy clinical trials.
Timeline of Discovery and Collaborative Effort
The research published in Nature Neuroscience is the culmination of years of collaborative work involving institutions across the globe. The project began with the initial identification of the skull-brain channels, a breakthrough that occurred within the last five years. Building on that anatomical foundation, the MECCC team spent the last three years utilizing mouse models and human retrospective data to map the immune changes.
The study involved a massive team of experts from MECCC and Albert Einstein College of Medicine, including Abhishek Dubey, Biljana Stangeland, Imane Abbas, and several others. International collaboration played a key role, with contributions from researchers at Osaka University in Japan, Karolinska Hospital in Sweden, Duke University, the University of California, San Francisco, and the German Rheumatism Research Center in Berlin.
Implications for Future Treatment Strategies
The discovery that glioblastoma is a systemic disease involving the skull marrow opens a new frontier for therapeutic intervention. Rather than focusing solely on the brain, future treatments may need to target the "calvarial" (skull) environment.
"Our discovery that this notoriously hard-to-treat brain cancer interacts with the body’s immune system may help explain why current therapies—all of them dealing with glioblastoma as a local disease—have failed," said Dr. Jinan Behnan.
Potential new strategies suggested by the research include:
- Marrow-Targeting Therapies: Developing drugs that can reset the immune balance in the skull marrow, specifically by suppressing the overproduction of neutrophils and restoring B and T cell populations.
- Channel Modulation: Investigating ways to "close" or regulate the microscopic channels between the skull and the brain to prevent the tumor from recruiting inflammatory cells.
- Refining Immunotherapy: Adjusting immunotherapy protocols to account for the systemic immune suppression caused by the tumor, ensuring that drugs like anti-PD-L1 are not neutralized by the marrow’s altered state.
- Re-evaluating Bone Health Protocols: Conducting further clinical reviews to determine how bone-modifying agents should be used in patients with primary brain tumors.
As the oncology community digests these findings, the focus shifts toward clinical trials that can translate this biological understanding into improved patient outcomes. By recognizing the skull as an active participant in the progression of glioblastoma, researchers may finally have found the "missing link" needed to break the stalemate in the fight against this devastating disease.












Leave a Reply