Researchers at Johns Hopkins Medicine have unveiled a significant breakthrough in the understanding of Alzheimer’s disease, identifying a specific protein pathway that could serve as a primary target for future therapeutic interventions. Supported by new funding from the National Institutes of Health (NIH), the study highlights the role of Cystathionine γ-lyase (CSE), an enzyme responsible for producing hydrogen sulfide ($H_2S$) within the brain. While hydrogen sulfide is most commonly recognized by its pungent, "rotten egg" odor, this research suggests that in trace amounts, the gas is an essential signaling molecule that maintains cognitive health and protects the structural integrity of the brain.
The findings, recently published in the Proceedings of the National Academy of Sciences (PNAS), indicate that the loss of CSE activity may be a driving factor in the onset of neurodegenerative symptoms. By utilizing genetically engineered mouse models, the research team demonstrated that a deficiency in this protein leads to a cascade of cellular failures, including increased oxidative stress, DNA damage, and a breakdown of the blood-brain barrier—all of which are hallmark indicators of Alzheimer’s disease in humans.
The Biological Significance of Hydrogen Sulfide
For decades, hydrogen sulfide was viewed primarily as a toxic environmental byproduct. However, in the late 20th and early 21st centuries, scientists began to categorize it as a "gasotransmitter," a family of gaseous molecules that includes nitric oxide and carbon monoxide. These gases are produced endogenously by the body to regulate various physiological processes. In the brain, $H_2S$ acts as a neuromodulator, influencing synaptic plasticity and protecting neurons from the harmful effects of reactive oxygen species.
The challenge for researchers has long been the "Goldilocks" nature of the gas: while it is vital for health in minute quantities, it is highly toxic in larger concentrations. This toxicity makes direct delivery of the gas to the brain an unsafe prospect for human medicine. Consequently, the Johns Hopkins study focused on the enzyme CSE, which acts as the biological "faucet" for $H_2S$. By understanding how to modulate the enzyme itself, scientists hope to find a way to maintain the safe, natural levels of the gas required to keep neurons functioning.
Experimental Methodology: The Barnes Maze and Cognitive Decline
To isolate the role of CSE in memory and learning, the research team, led by Bindu Paul, M.S., Ph.D., associate professor of pharmacology, psychiatry, and neuroscience at the Johns Hopkins University School of Medicine, conducted a series of longitudinal experiments. They compared "normal" wild-type mice with a strain of mice engineered to lack the CSE enzyme.
The primary tool for assessing cognitive function was the Barnes maze, a circular platform with multiple holes, only one of which leads to a dark, recessed "escape box." Because mice are naturally averse to open, brightly lit spaces, they are motivated to find the shelter. The test measures spatial memory—the ability to remember landmarks and directions.
The results provided a clear chronological view of how CSE deficiency impacts the brain over time:
- Early Stage (Two Months): At two months of age, the CSE-deficient mice performed almost identically to the control group. Both sets of mice were able to locate the escape box within three minutes, suggesting that early-life cognitive development remained relatively intact despite the lack of the protein.
- Middle Stage (Six Months): By the time the mice reached six months—roughly equivalent to middle age in humans—a stark divergence emerged. The control mice continued to navigate the maze efficiently. However, the mice lacking CSE showed significant struggle. They took longer to find the exit, made more errors, and appeared to lose the "spatial map" they had previously developed.
Suwarna Chakraborty, a researcher in Paul’s lab and the study’s first author, noted that this decline in spatial memory indicates a progressive onset of neurodegeneration. The fact that the symptoms worsened with age suggests that CSE is not just a component of the brain, but a critical maintenance tool that prevents the gradual "wear and tear" associated with Alzheimer’s.
Cellular Destruction and the Blood-Brain Barrier
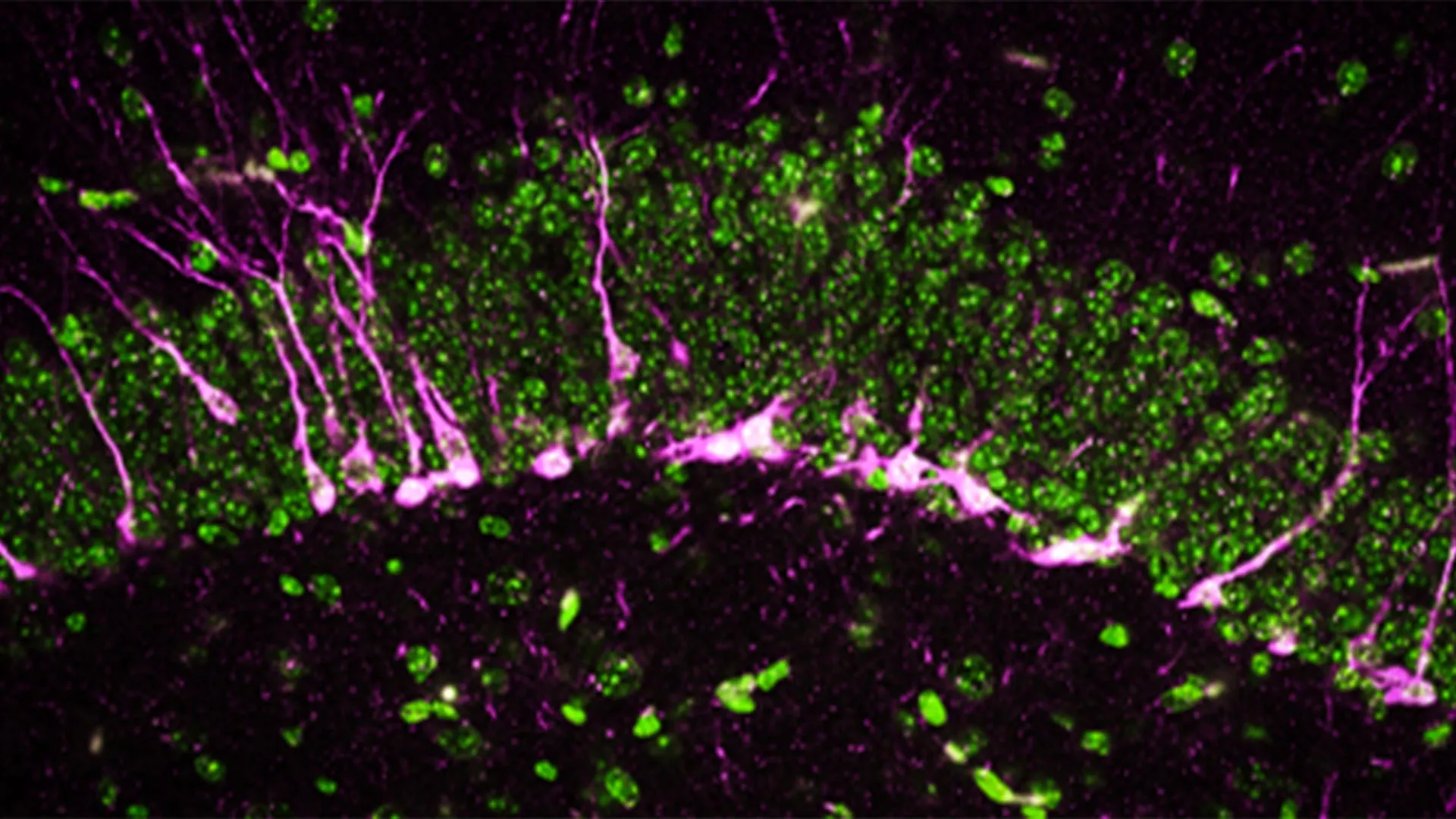
Beyond behavioral changes, the study utilized high-powered electron microscopy and biochemical analysis to examine the physical state of the brain. The team focused heavily on the hippocampus, the region of the brain responsible for converting short-term memories into long-term ones.
The researchers discovered that in the absence of CSE, the hippocampus was unable to effectively produce new neurons, a process known as neurogenesis. The proteins required to signal the birth and integration of these new cells were either reduced or entirely absent. Furthermore, the newly formed neurons that did manage to develop had difficulty migrating to their proper positions within the hippocampal circuit.
One of the most alarming findings involved the blood-brain barrier (BBB). The BBB is a highly selective semipermeable border of endothelial cells that prevents solutes in the circulating blood from non-selectively crossing into the extracellular fluid of the central nervous system. In the CSE-deficient mice, researchers observed large physical breaks in the blood vessels.
"The mice lacking CSE were compromised at multiple levels," explained co-first author Sunil Jamuna Tripathi. The structural damage to the blood vessels allows toxins to leak into the brain, while the lack of $H_2S$ leaves the neurons vulnerable to oxidative stress. This "double hit" of environmental toxicity and internal vulnerability mirrors the complex pathology seen in human Alzheimer’s patients.
A Decade of Discovery: Chronology of the Research
The current study is the culmination of more than fifteen years of investigation into the CSE protein. The timeline of discovery highlights the evolving understanding of how this enzyme affects the body:
- 2008: The CSE-deficient mouse model was first developed. At the time, researchers focused on the peripheral nervous system, linking the protein to blood vessel function and the regulation of blood pressure.
- 2014: A team led by Solomon Snyder, M.D., D.Sc., D.Phil., reported that CSE played a protective role in Huntington’s disease. This was one of the first major indications that the protein was vital for brain health specifically in the context of neurodegeneration.
- 2021: The Johns Hopkins group found that CSE was malfunctioning in existing Alzheimer’s mouse models. They discovered that administering very small, controlled injections of hydrogen sulfide could temporarily restore some cognitive function.
- Present Day: The latest research isolates CSE as a primary player. Unlike previous studies that used mice with multiple genetic mutations, this study proved that removing CSE alone is sufficient to cause Alzheimer’s-like symptoms.
Solomon Snyder, professor emeritus and a co-corresponding author on the study, emphasized that this work confirms CSE as a major player in cognitive function. Snyder, who retired in 2023, has spent decades pioneering the study of neurotransmitters, and this final collaboration underscores a potential paradigm shift in how the scientific community approaches dementia.
Implications for the Global Alzheimer’s Crisis
The search for an Alzheimer’s cure has been marked by decades of high-profile clinical trial failures. Most traditional research has focused on the "Amyloid Hypothesis"—the idea that clearing amyloid-beta plaques from the brain will stop the disease. However, drugs targeting these plaques have shown limited success in reversing cognitive decline once it has begun.
The Johns Hopkins research offers a different "upstream" approach. By focusing on CSE and hydrogen sulfide, scientists are looking at how the brain protects itself from damage in the first place. If a therapy can be developed to boost CSE activity or safely mimic the effects of $H_2S$, it might be possible to preserve the blood-brain barrier and maintain neurogenesis, effectively slowing the progression of the disease before the damage becomes irreversible.
According to the U.S. Centers for Disease Control and Prevention (CDC), more than 6 million Americans are currently living with Alzheimer’s, a number expected to nearly triple by 2060. The economic burden is equally staggering, with costs related to care and lost productivity reaching hundreds of billions of dollars annually. The discovery of the CSE pathway provides a much-needed new "avenue for treatment" in a field that has struggled to find consistent results.
Collaborative Effort and Future Outlook
The scale of this research is reflected in its diverse funding and the wide range of contributing institutions. Major support came from the National Institutes of Health, the Department of Defense, and the American Heart Association. Private foundations, including the Valour Foundation and the Wick Foundation, also played critical roles in sustaining the long-term study.
The research was a collaborative effort involving experts from Johns Hopkins University, Case Western University, the Leibniz Institute for Analytical Sciences in Germany, the Medical University of South Carolina, and the West Virginia University School of Medicine. This multidisciplinary approach allowed the team to bridge the gap between behavioral psychology, biochemistry, and advanced imaging.
As the team moves forward, the next step will be identifying small-molecule compounds that can cross the blood-brain barrier and stimulate CSE production. While the transition from mouse models to human clinical trials is a long and rigorous process, the isolation of CSE as a primary cognitive protector gives drug developers a specific, measurable target. For the millions of families affected by Alzheimer’s, this "rotten egg" gas—once considered a mere nuisance—may represent one of the most promising leads in modern neuroscience.















Leave a Reply