Eli Lilly and Company today announced groundbreaking results from a Phase 3 clinical trial for its investigational triple agonist, retatrutide, demonstrating an average weight loss of 16.8% in participants with type 2 diabetes. This significant achievement positions retatrutide as a potentially transformative treatment in the ongoing battle against type 2 diabetes and its associated comorbidities, including obesity. The company confirmed that the drug met both primary and key secondary endpoints, showcasing superior reductions in glycated hemoglobin (A1C) and body weight over a 40-week treatment period when compared to a placebo.
Unpacking the Clinical Success: Key Data and Endpoints
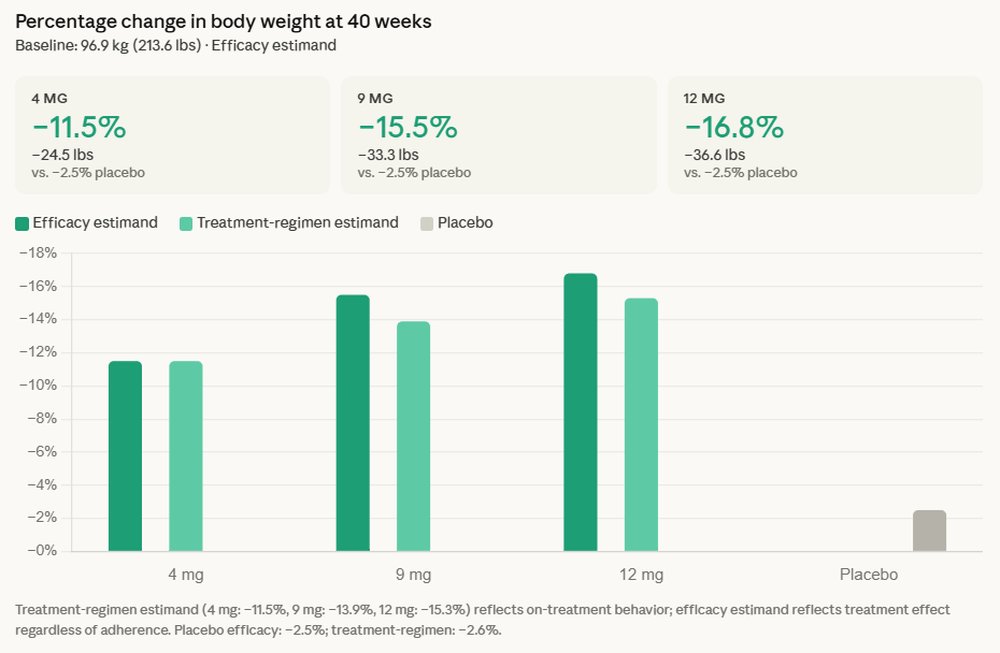
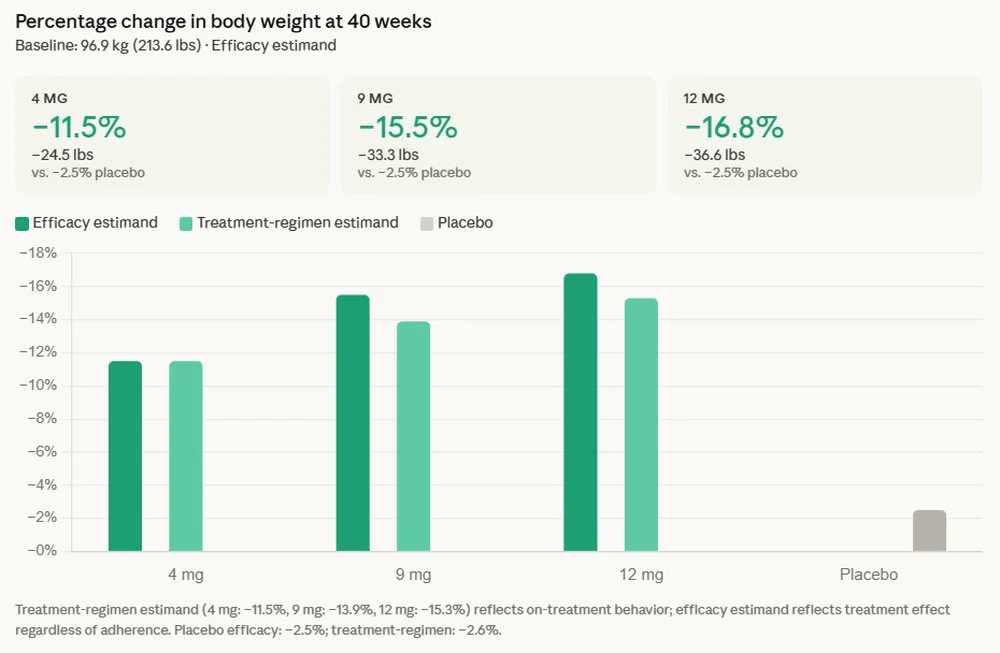
The reported average weight loss of 16.8% translates to an impressive average reduction of 36.6 pounds for participants in the trial. Beyond sheer weight reduction, the study highlighted a crucial observation: no weight-loss plateau was observed, with patients continuing to lose weight throughout the 40-week duration. This sustained efficacy is a significant differentiator and could represent a major advantage over existing therapies, where weight loss often slows or plateaus after an initial period.
Equally critical for patients with type 2 diabetes, retatrutide delivered an average A1C reduction of up to 2.0%. A1C is a key metric reflecting average blood sugar levels over the past two to three months, and substantial reductions are vital for managing the disease and preventing long-term complications. Achieving both significant A1C control and substantial weight loss simultaneously has historically been a major challenge for many individuals living with type 2 diabetes, as obesity often complicates disease management.
Kenneth Custer, executive vice president and president of Lilly Cardiometabolic Health, underscored the significance of these findings, stating, “For many people with type 2 diabetes, it is a struggle to achieve both A1C control and weight loss, since obesity has historically been harder to treat for those with type 2 diabetes. With triple agonist retatrutide, we set out to make a molecule that could help patients achieve substantial A1C reduction and weight loss. These results support the potential of this molecule for people living with type 2 diabetes.” His statement reflects Lilly’s strategic focus on developing comprehensive solutions for complex cardiometabolic conditions.
A Novel Mechanism: The Triple Agonist Advantage
Retatrutide is an investigational first-in-class GIP, GLP-1, and glucagon triple hormone receptor agonist. This unique pharmacological profile distinguishes it from current treatments and is central to its remarkable efficacy. To understand its impact, it’s essential to grasp the roles of these three endogenous hormones:
- Glucose-dependent insulinotropic polypeptide (GIP): A gut hormone that enhances insulin secretion in a glucose-dependent manner and plays a role in fat metabolism. Agonism of the GIP receptor has been shown to improve insulin sensitivity and promote weight loss.
- Glucagon-like peptide-1 (GLP-1): Another gut hormone that stimulates insulin release, suppresses glucagon secretion, slows gastric emptying, and reduces appetite. GLP-1 receptor agonists are a cornerstone of modern diabetes and obesity treatment.
- Glucagon: While primarily known for raising blood glucose, glucagon also has metabolic effects that can be leveraged therapeutically. Glucagon receptor agonism can increase energy expenditure and reduce food intake, contributing to weight loss, especially when combined with GIP and GLP-1 agonism, which counteract its hyperglycemic effects.
By simultaneously activating receptors for all three hormones, retatrutide aims to achieve a synergistic effect, addressing multiple metabolic pathways implicated in type 2 diabetes and obesity. This multi-pronged approach appears to enhance glycemic control, promote greater and sustained weight loss, and potentially offer broader cardiometabolic benefits compared to single or dual agonists. This represents a significant leap from earlier generations of GLP-1 agonists and even the dual GIP/GLP-1 agonists that have already shown impressive results.
Beyond Glycemic Control and Weight Loss: Cardiovascular and Metabolic Improvements
The trial data also revealed clinically meaningful improvements across several key cardiovascular risk factors. Participants treated with retatrutide experienced beneficial changes from baseline in non-HDL cholesterol, triglycerides, and systolic blood pressure. These improvements are particularly noteworthy because type 2 diabetes is often accompanied by dyslipidemia (abnormal cholesterol levels) and hypertension (high blood pressure), both of which significantly increase the risk of cardiovascular events like heart attacks and strokes. By positively impacting these parameters, retatrutide holds the promise of not only managing diabetes and obesity but also reducing the overall cardiovascular burden on patients, which is the leading cause of morbidity and mortality in this population. This holistic benefit underscores the potential for a profound impact on long-term patient health outcomes.
Safety Profile: Consistent with Class
Regarding safety, the common adverse effects reported in the trial included nausea, diarrhea, and vomiting. These are consistent with side effects observed with other incretin-based therapies, such as GLP-1 and GIP/GLP-1 receptor agonists. While these gastrointestinal side effects can be challenging for some patients, they are generally manageable and often diminish over time as the body adjusts to the medication. The consistency of the safety profile with similar therapies suggests that clinicians and patients will have a familiar framework for managing potential side effects, allowing for careful titration and patient education. The overall tolerability, especially in light of the significant efficacy, will be a key factor in its clinical adoption.
The Broader Clinical Development Program
The Phase 3 trial whose results were just announced, likely part of the TRANSCEND-T2D-1 study, is just one component of a much broader and ambitious clinical development program for retatrutide. Eli Lilly is currently investigating the triple agonist in several other Phase 3 clinical trials, exploring its potential efficacy and safety across a wide spectrum of cardiometabolic and related conditions. These include:
- Obesity and overweight with at least one weight-related medical problem: This indicates a direct pursuit of an obesity indication, similar to Lilly’s tirzepatide (Zepbound) and Novo Nordisk’s semaglutide (Wegovy).
- Knee osteoarthritis: Obesity is a major risk factor for osteoarthritis, and significant weight loss could alleviate symptoms and slow disease progression.
- Moderate-to-severe obstructive sleep apnea: A common comorbidity of obesity, which could be dramatically improved with weight reduction.
- Chronic low back pain: Another condition often exacerbated by excess body weight.
- Cardiovascular and renal outcomes: Dedicated trials to definitively establish the drug’s benefits in reducing major adverse cardiovascular events and slowing the progression of kidney disease, which are crucial for long-term health.
- Metabolic dysfunction-associated steatotic liver disease (MASLD), formerly known as NAFLD: A rapidly growing global health concern, often linked to obesity and type 2 diabetes, for which effective pharmacological treatments are urgently needed.
This extensive pipeline of trials highlights Lilly’s confidence in retatrutide’s broad therapeutic potential and its strategic vision to position the drug as a comprehensive solution for multiple interconnected health challenges. Success in these additional indications could significantly expand the addressable patient population and solidify retatrutide’s market position.
The Evolving Landscape of Diabetes and Obesity Treatment
The field of diabetes and obesity treatment has witnessed rapid advancements in recent years, largely driven by the development of incretin-based therapies. For decades, managing type 2 diabetes involved a range of medications focusing on blood sugar control, with weight loss often a secondary or even challenging outcome. The advent of GLP-1 receptor agonists like semaglutide (Ozempic for diabetes, Wegovy for obesity) revolutionized this approach by offering both A1C reduction and meaningful weight loss.
Eli Lilly further pushed the boundaries with tirzepatide (Mounjaro for diabetes, Zepbound for obesity), a dual GIP/GLP-1 receptor agonist, which demonstrated even greater efficacy in both glycemic control and weight reduction, often surpassing single-agonist GLP-1 drugs. Tirzepatide’s impressive results, with participants achieving average weight losses exceeding 20% in some trials, set a new benchmark.
Now, retatrutide, with its triple agonist mechanism, appears to be raising that bar once again. While direct head-to-head comparisons across different trials are challenging due to varying patient populations and study designs, the 16.8% average weight loss in type 2 diabetes patients for retatrutide is highly competitive and, in some contexts, exceeds the average weight loss observed with many existing dual agonists in similar populations. The "no plateau" observation also suggests a sustained effect that could offer an edge.
This intense innovation fosters a dynamic and competitive market. Novo Nordisk and Eli Lilly are at the forefront, continually developing next-generation compounds that push the boundaries of efficacy. This competition is ultimately beneficial for patients, offering more potent and comprehensive treatment options for conditions that impact hundreds of millions globally.
Broader Implications and Future Outlook
The positive Phase 3 results for retatrutide carry significant implications for various stakeholders:
- For Patients: These results offer renewed hope for individuals struggling with type 2 diabetes and obesity. The promise of significant and sustained weight loss combined with excellent glycemic control and cardiovascular benefits could drastically improve quality of life and reduce the long-term health complications associated with these chronic conditions.
- For Healthcare Providers: Clinicians will gain another powerful tool in their armamentarium. The comprehensive benefits of retatrutide could simplify treatment regimens by addressing multiple aspects of cardiometabolic disease with a single agent.
- For Eli Lilly: These findings represent a substantial win and reinforce Lilly’s leadership in the cardiometabolic space. With tirzepatide already a blockbuster, retatrutide has the potential to become another cornerstone product, driving significant revenue growth and expanding the company’s market dominance. The broad development program also positions Lilly to address a wider range of medical needs, solidifying its future pipeline.
- For the Pharmaceutical Market: The success of retatrutide will undoubtedly intensify research and development efforts across the industry into multi-agonist approaches. It sets a new standard for efficacy in weight management and diabetes, potentially accelerating the development of even more advanced therapies.
- Public Health Impact: Given the global epidemics of obesity and type 2 diabetes, highly effective treatments like retatrutide could have a profound public health impact, potentially reducing healthcare burdens related to associated complications such as heart disease, stroke, kidney failure, and liver disease.
Following these successful Phase 3 results, Eli Lilly will likely proceed with regulatory submissions to health authorities around the world, including the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA). The review process will scrutinize the full dataset, including efficacy, safety, and manufacturing quality, before a decision on approval is made. If approved, retatrutide would enter a rapidly expanding market, but one where its unique triple agonist mechanism and compelling clinical data could quickly establish it as a leading therapeutic option. The medical community eagerly anticipates the full publication of these trial results and the subsequent steps in retatrutide’s journey to market.














Leave a Reply