The human bone marrow, often described as the body’s "blood factory," is a sophisticated organ of immense biological complexity, comprising a dense network of bone cells, nerves, blood vessels, and various immune cell types. In a groundbreaking development published in the journal Cell Stem Cell, researchers from the University of Basel and University Hospital Basel have successfully engineered a functional, three-dimensional model of this environment using exclusively human cells. This achievement marks a significant milestone in tissue engineering and regenerative medicine, offering a realistic platform for studying blood-related diseases, testing pharmacological interventions, and advancing the principles of personalized medicine while simultaneously addressing the ethical and scientific limitations of animal experimentation.
The Architecture of the Blood Factory: Understanding the Endosteal Niche
Bone marrow is not a uniform substance but a highly organized tissue situated within the cavities of the skeletal system. Its primary function is hematopoiesis—the continuous production of blood cells, including red blood cells, white blood cells, and platelets. This process occurs within specialized microenvironments known as "niches." Among these, the endosteal niche is of particular interest to oncologists and hematologists. Located near the inner surface of the bone, this niche serves as a sanctuary for hematopoietic stem cells and is frequently implicated in the progression of blood cancers such as leukemia.
The endosteal niche is characterized by its "quiet" or quiescent state, where stem cells are maintained in a protected environment. However, this same protection often extends to malignant cells. During chemotherapy, cancer cells within the bone marrow can retreat into these niches, where the microenvironment provides signals that help them resist treatment and eventually cause a relapse. Until now, recreating the sheer complexity of this niche—which integrates vascular structures, neural inputs, and a mineralized bone matrix—has proven nearly impossible using traditional laboratory techniques. Previous models often relied on simplified two-dimensional cultures or hybrid systems that combined human cells with animal-derived scaffolds, neither of which could accurately replicate the unique physiological signaling of the human body.
A Chronological Shift: From Animal Models to Human-Centric Systems
For more than half a century, the study of hematology has leaned heavily on murine (mouse) models. While these studies provided the foundational knowledge of how blood cells develop, they have consistently encountered the "translational gap"—the phenomenon where results observed in mice fail to replicate in human clinical trials. Human and mouse bone marrows differ significantly in their immunological profiles, signaling molecules, and structural density.
The timeline of this specific breakthrough began with the convergence of two major scientific fields: synthetic material science and stem cell biology. The team, led by Professor Ivan Martin and Dr. Andrés García García at the Department of Biomedicine, sought to move beyond the limitations of animal-based research. Their objective was to build a system that followed the "3Rs" principle—Replacement, Reduction, and Refinement of animal experiments.
The development process involved several distinct phases. First, the researchers selected a suitable structural foundation. They utilized hydroxyapatite, a naturally occurring mineral form of calcium apatite that constitutes the primary inorganic component of human bone and teeth. This provided the necessary mechanical cues for the cells. Second, the team leveraged advanced molecular biology to create induced pluripotent stem cells (iPSCs). These are adult cells that have been "reprogrammed" back into an embryonic-like state, giving them the potential to differentiate into any cell type within the human body.
Technical Methodology and Functional Capacity of the 3D Model
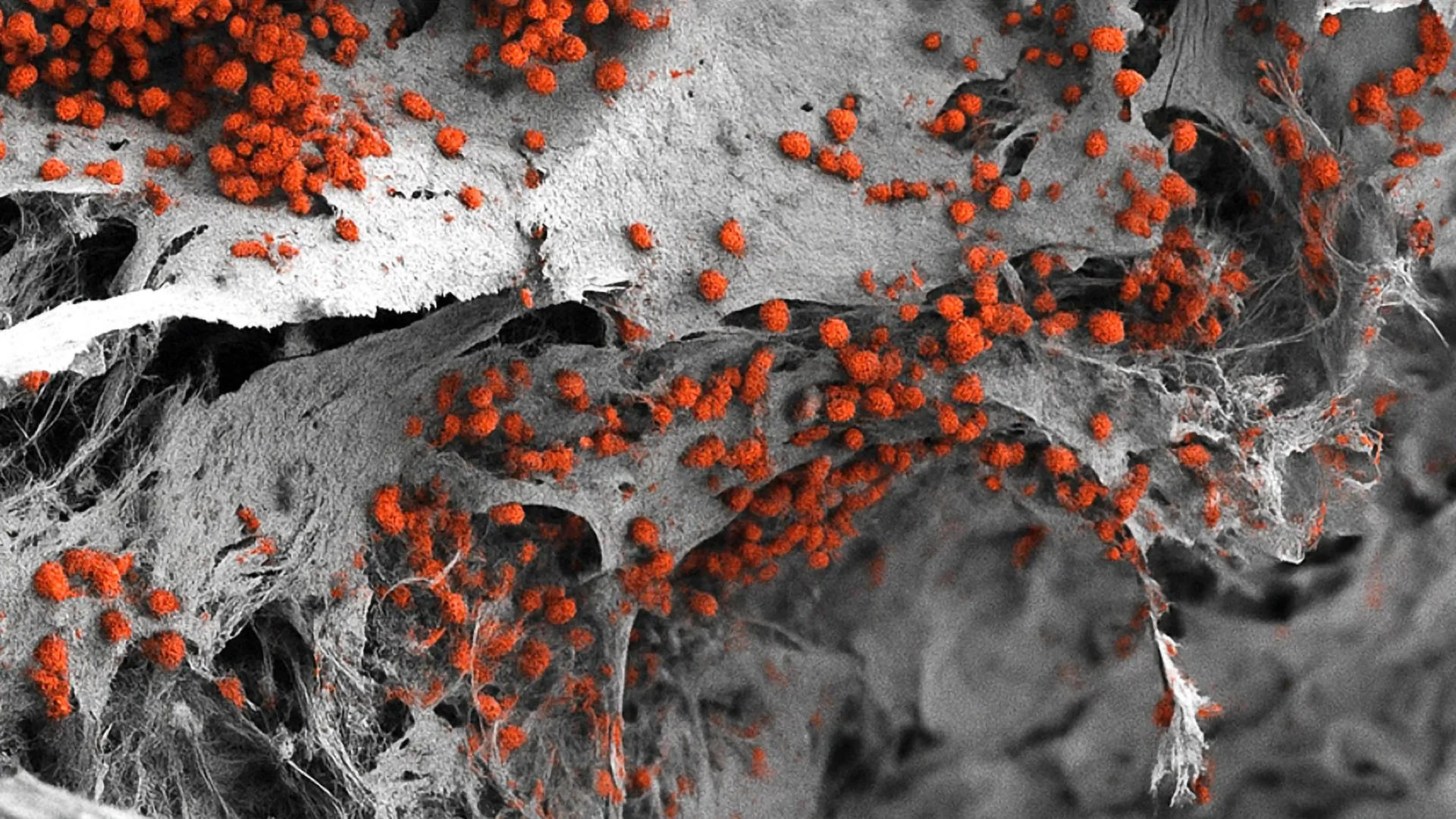
The assembly of the model required precise orchestration of cellular development. By introducing human iPSCs into the hydroxyapatite scaffold and applying specific growth factors and environmental signals, the researchers guided the cells to transform into the various components of the bone marrow. This included the formation of osteoblasts (bone-forming cells), endothelial cells (which form blood vessels), and mesenchymal stromal cells (which provide structural support).
One of the most impressive aspects of the study is the scale and longevity of the resulting model. Measuring eight millimeters in diameter and four millimeters in thickness, the engineered tissue is substantially larger than typical "organ-on-a-chip" systems. This macro-scale approach allows for a more realistic distribution of cells and nutrients. Data collected during the study confirmed that the model remained functional for several weeks, successfully supporting the continuous formation and maturation of human blood cells in a laboratory setting. This duration is critical for longitudinal studies on disease progression or the long-term effects of drug exposure.
Furthermore, the researchers utilized high-resolution imaging and transcriptomic analysis to verify that the spatial organization of the cells matched the architecture of the human endosteal niche. The presence of functional blood vessel-like structures within the model ensures that nutrients can reach the interior cells, mimicking the vascularization necessary for healthy marrow function.
Supporting Data and Comparative Analysis
The necessity for such a model is underscored by the high failure rate of new drugs in the oncology sector. Currently, approximately 90% of drugs that pass animal testing fail in human clinical trials, often due to unforeseen toxicity or lack of efficacy in the complex human environment. By providing a 100% human-based platform, the Basel researchers offer a "human-first" filter that could identify these failures much earlier in the development pipeline.
Data from the study highlighted several key advantages:
- Species Specificity: The model avoids the cross-species signaling errors that occur when human cancer cells are transplanted into immunocompromised mice.
- Controlled Environment: Unlike living organisms, the 3D model allows researchers to isolate specific variables, such as the effect of a single cytokine or a specific genetic mutation, without the interference of systemic biological noise.
- Resource Efficiency: While the initial creation of iPSCs is resource-intensive, the ability to replicate models consistently in a lab setting can eventually reduce the costs associated with maintaining large animal colonies for research.
Official Responses and Ethical Implications
The research has been met with enthusiasm from both the scientific community and animal welfare advocates. Professor Ivan Martin emphasized that while mouse studies have been instrumental in the past, the shift toward human-centric biology is a natural evolution of medical science. "Our model brings us closer to the biology of the human organism," Martin stated, noting that it serves as a vital complement to existing research methods.
Dr. Andrés García García pointed out that while the current model is a triumph of complexity, future iterations will need to focus on scalability. For high-throughput drug screening—where thousands of compounds are tested simultaneously—the current 8mm size is too large. The next phase of research will likely involve miniaturizing the system to fit into standard laboratory plates while maintaining the intricate cellular relationships that make the model effective.
From an ethical standpoint, the University of Basel has reaffirmed its commitment to the 3Rs. This project represents a tangible step toward reducing the reliance on animal models for complex organ systems. As public and regulatory pressure to move away from animal testing increases, such humanized models provide a scientifically rigorous alternative that does not compromise on the quality of data.
Broader Impact: Towards Personalized Oncology
Perhaps the most promising application of this technology lies in the realm of personalized medicine. In the current standard of care, a patient with a blood cancer like multiple myeloma or acute myeloid leukemia often undergoes a "trial and error" approach to chemotherapy. If one drug fails, the doctor moves to the next.
The vision for the future, enabled by this research, involves creating "patient avatars." A clinician could take a small sample of a patient’s own cells, use them to grow a personalized 3D bone marrow model, and then test an array of different therapeutic combinations on that specific patient’s marrow in the lab. This would allow doctors to identify the most effective treatment with the least toxicity before the patient ever receives a dose.
Furthermore, the model provides a new lens through which to study "minimal residual disease"—the small number of cancer cells that survive treatment and lead to relapse. By observing how these cells interact with the human endosteal niche in the 3D model, researchers may discover new ways to "flush out" cancer cells from their protective environments, making them more vulnerable to existing treatments.
Future Outlook and Conclusion
The development of a fully humanized bone marrow model is not merely a technical achievement; it is a paradigm shift in how we approach hematological research. By successfully integrating the mineral, vascular, and cellular components of the bone marrow into a single functional system, the team at the University of Basel has provided a roadmap for the future of tissue engineering.
While challenges remain—particularly regarding the integration of a full immune system into the model and the scaling of the platform for industrial use—the foundation has been laid. As this technology matures, it promises to accelerate the discovery of life-saving drugs, provide deeper insights into the mechanisms of cancer, and move the scientific community toward a more ethical and accurate era of human-centric medical research. The "blood factory" in the lab is no longer a theoretical concept; it is a working reality that brings us one step closer to conquering some of the most challenging diseases of our time.














Leave a Reply