The resolution of a geological enigma that has confounded the scientific community for over 200 years has finally been achieved through a collaborative effort between the University of Michigan and Hokkaido University. For more than two centuries, researchers have struggled to replicate the natural formation of the mineral dolomite in a laboratory setting. Despite its prevalence in ancient rock formations across the globe, dolomite has remained notoriously difficult to synthesize under conditions that mimic the natural environment. This discrepancy, famously known as the "Dolomite Problem," has finally been unraveled through a combination of cutting-edge atomic simulations and innovative electron microscopy.
The findings, published recently in the journal Science, suggest that the secret to dolomite growth lies not in a continuous process, but in a cyclical one involving the periodic removal of structural defects. This breakthrough not only provides an answer to a fundamental question in earth science but also offers transformative potential for the manufacturing of high-tech materials, including semiconductors and next-generation batteries.
The Historical Context of the Dolomite Problem
Dolomite, a calcium magnesium carbonate mineral, was first described in 1791 by the French naturalist and geologist Déodat de Dolomieu. He identified the mineral in the mountain range now known as the Dolomites in northeastern Italy. These peaks, characterized by their jagged profiles and pale coloration, are composed of vast quantities of the mineral. Beyond Italy, dolomite is a primary constituent of Niagara Falls and the striking "hoodoo" formations found in Utah’s Bryce Canyon National Park.
Geological records indicate that dolomite is incredibly abundant in rocks older than 100 million years. It constitutes approximately 10% of all sedimentary rocks on Earth. However, a paradox emerges when examining more recent geological strata. In modern environments, dolomite is exceedingly rare, and until this recent study, it was virtually impossible to grow in a laboratory at room temperature and pressure.
For decades, the inability to synthesize dolomite led scientists to speculate about the conditions of the ancient Earth. Some theorized that the oceans were once chemically different, perhaps more saline or warmer, to facilitate the mineral’s growth. Others searched for specific bacteria that might have acted as a catalyst. However, none of these theories could be consistently proven in a lab, leaving the "Dolomite Problem" as one of the most persistent mysteries in mineralogy.
The Atomic Barrier to Growth
The research team, led by Wenhao Sun, the Dow Early Career Professor of Materials Science and Engineering at the University of Michigan, approached the problem by examining the atomic-scale interactions that occur during crystal formation. In most minerals, growth occurs as atoms in a solution attach themselves to the surface of a crystal in a neat, orderly fashion.
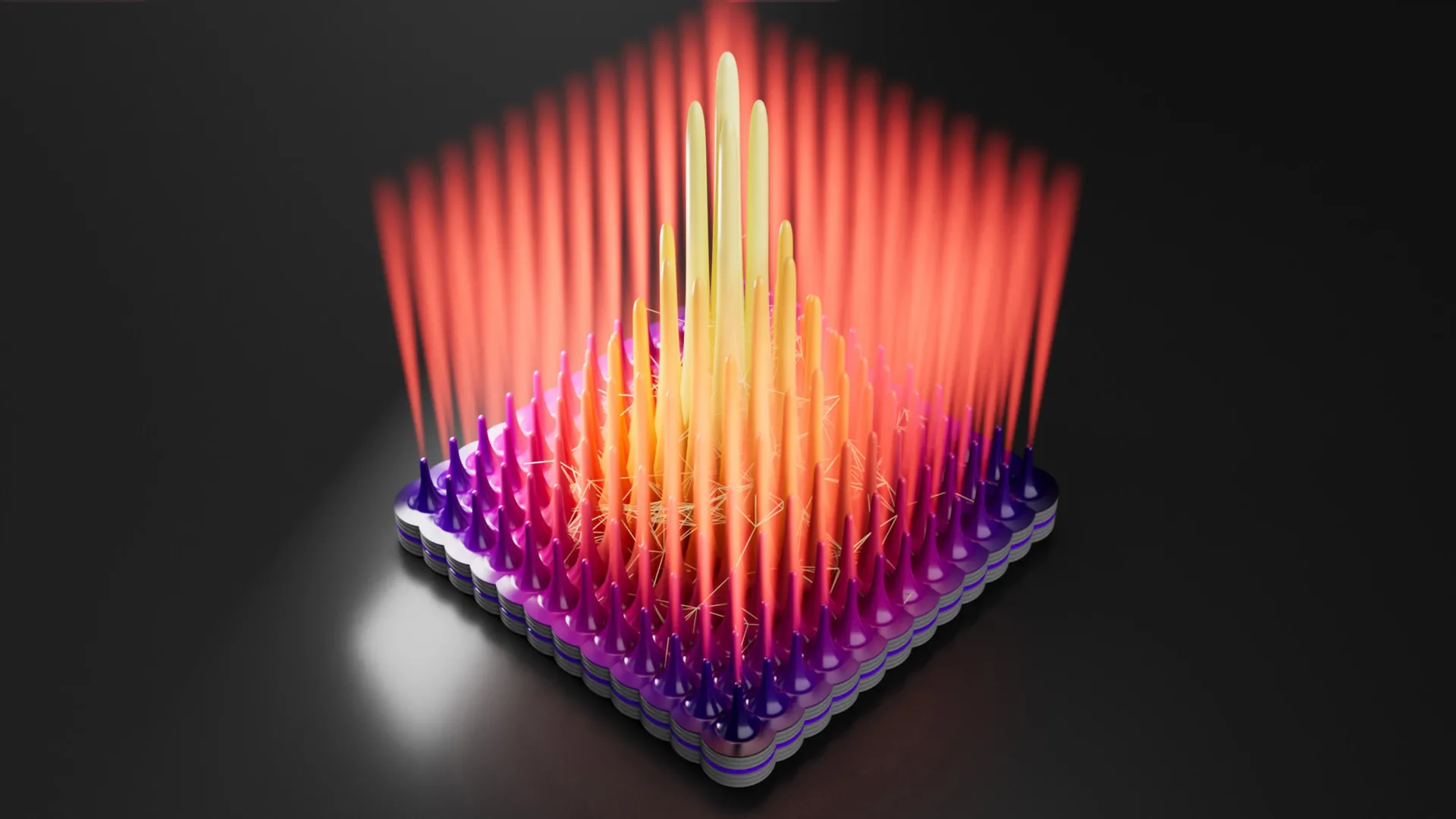
Dolomite, however, possesses a unique and highly organized structure. It consists of alternating layers of calcium and magnesium. For the crystal to grow correctly, these atoms must choose their specific positions with perfect precision. The difficulty arises because, in a solution, calcium and magnesium ions are chemically similar and often attach themselves to the wrong sites.
When a magnesium atom occupies a spot intended for calcium, or vice versa, it creates a structural defect. These defects act as a physical barrier, preventing new layers from forming on top of them. The research team calculated that at the slow rates of natural growth, it would take roughly 10 million years to form a single well-ordered layer of dolomite if the process were continuous. This "kinetic bottleneck" explains why dolomite appears to grow so slowly in nature and why it failed to form in traditional laboratory experiments that sought to grow crystals in a steady state.
A Paradigm Shift: The Role of Dissolution
The breakthrough came when the researchers shifted their focus from how dolomite grows to how it survives. Using a new theory based on detailed atomic simulations, the team realized that the structural defects that stop growth are not permanent. Because these "misplaced" atoms are not in their most stable positions, they are more easily dissolved back into the surrounding water than the correctly placed atoms.
In natural environments, conditions are rarely static. Environments where dolomite is currently found—such as coastal lagoons or tidal flats—experience frequent fluctuations. Cycles of rainfall, evaporation, and tidal changes cause the chemistry of the water to shift. The researchers hypothesized that these cycles provide a "reset" mechanism. When the water becomes less saturated, the unstable defects dissolve away. When the water becomes saturated again, a clean, ordered surface is available for new, correct layers to attach.
"If we understand how dolomite grows in nature, we might learn new strategies to promote the crystal growth of modern technological materials," Sun stated. This realization shifted the focus from seeking a constant "growth environment" to seeking a "dynamic environment" that allowed for periodic cleaning of the crystal surface.
Computational Innovation at the PRISMS Center
To validate their hypothesis, the team required a way to simulate the growth of dolomite over geological timescales, a task that would normally exceed the capabilities of even the most powerful supercomputers. Calculating the energy involved in every interaction between electrons and atoms is computationally expensive.
The solution came from the University of Michigan’s Predictive Structure Materials Science (PRISMS) Center. The center developed a specialized software package capable of streamlining these complex calculations. Instead of calculating every single interaction, the software identifies the energy for a selection of atomic arrangements and then uses the inherent symmetry of the crystal structure to extrapolate the energies for all other possible arrangements.
"Our software calculates the energy for some atomic arrangements, then extrapolates to predict the energies for other arrangements based on the symmetry of the crystal structure," explained Brian Puchala, an associate research scientist at U-M and a lead developer of the software.
This computational shortcut allowed the researchers to simulate the growth of dolomite over periods that would have previously required 5,000 CPU hours on a supercomputer. With the PRISMS software, the same calculations were completed in just two milliseconds on a standard desktop computer. These simulations confirmed that a "pulsed" growth strategy, where growth and dissolution alternate, could indeed allow dolomite to form millions of times faster than in a steady-state environment.
Laboratory Confirmation via Electron Microscopy
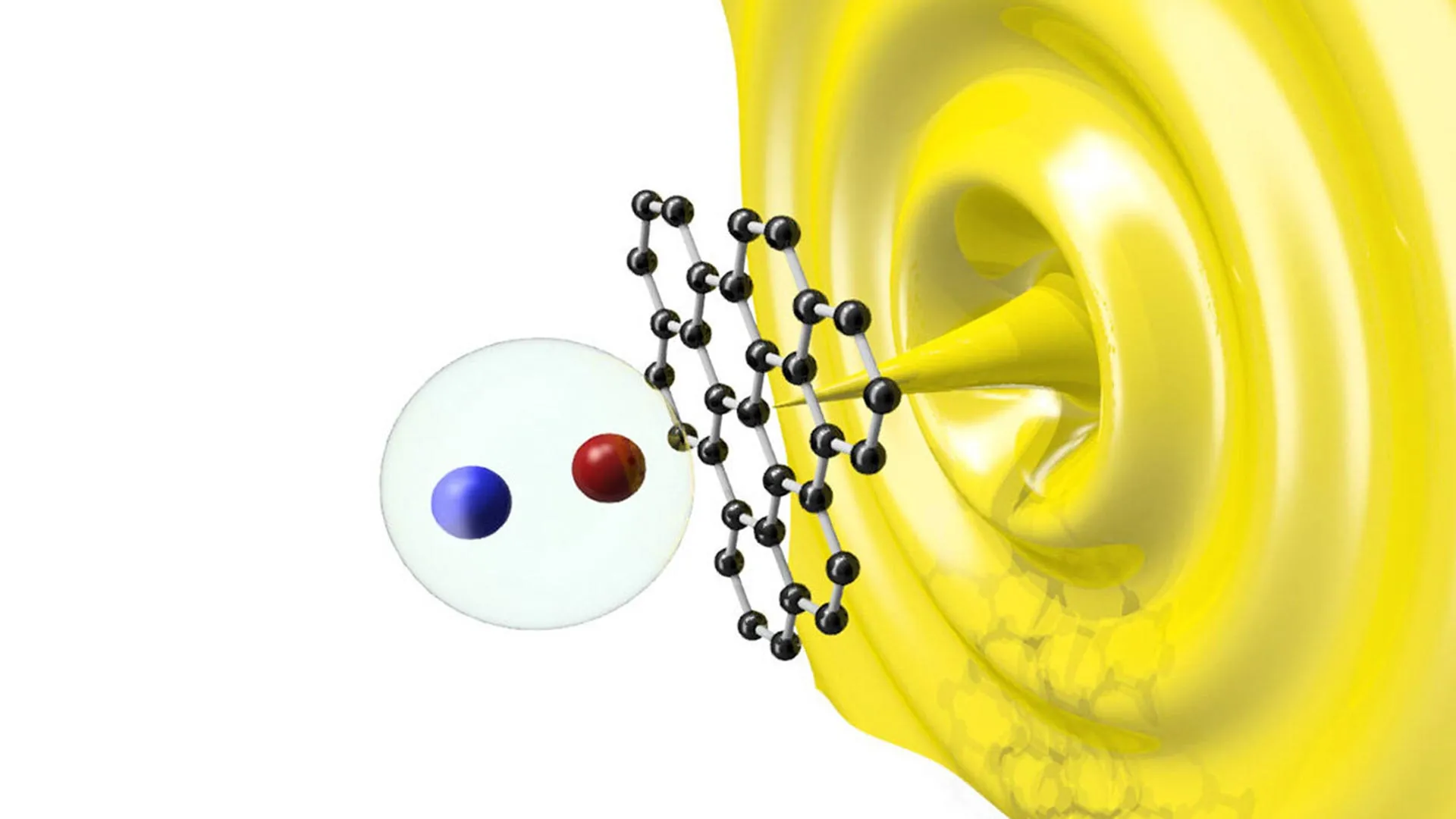
The theoretical model required experimental proof. This task was undertaken by Professor Yuki Kimura and postdoctoral researcher Tomoya Yamazaki at Hokkaido University in Japan. They utilized a Transmission Electron Microscope (TEM) to observe the growth of a dolomite crystal in real-time, but with a clever twist on the technology’s typical use.
In standard microscopy, the electron beam is used solely for imaging. However, the beam can also interact with the water in a solution, a process called radiolysis, which splits water molecules and creates a mild acidic environment. While this is usually considered a drawback because it can damage samples, Kimura and Yamazaki used it to their advantage.
By pulsing the electron beam, they could alternate between a state that allowed for crystal growth and a state that caused slight dissolution. They placed a small seed crystal of dolomite in a solution of calcium and magnesium. Over a two-hour period, the team pulsed the beam 4,000 times.
The results were unprecedented. The dolomite crystal grew by approximately 100 nanometers, consisting of roughly 300 new layers of the mineral. In all previous attempts over the last two centuries, scientists had never managed to grow more than five layers of dolomite in a lab. This experiment provided the first direct evidence that periodic dissolution is the key to overcoming the kinetic barriers of dolomite formation.
Implications for Modern Material Science
While the resolution of the "Dolomite Problem" is a landmark achievement for geologists, its implications extend far into the realm of materials science and industrial manufacturing. Many of the high-performance materials used in today’s technology—such as the wide-bandgap semiconductors found in electric vehicles or the specialized components in solar panels—require crystals that are entirely free of defects.
Traditionally, the industry has operated under the assumption that the best way to avoid defects is to grow crystals very slowly. The U-M and Hokkaido study suggests a counterintuitive approach: growing materials quickly but periodically "washing" them to remove flaws.
"In the past, crystal growers who wanted to make materials without defects would try to grow them really slowly," Sun said. "Our theory shows that you can grow defect-free materials quickly, if you periodically dissolve the defects away during growth."
This concept of "pulse-growth" could potentially revolutionize the production of:
- Semiconductors: Reducing the time and energy required to grow high-purity silicon or gallium nitride wafers.
- Batteries: Improving the structural integrity of cathode materials, which could lead to longer-lasting and faster-charging batteries.
- Carbon Sequestration: Understanding dolomite formation could lead to better methods for mineralizing carbon dioxide, potentially aiding in the removal of greenhouse gases from the atmosphere by locking them into stable mineral forms.
Conclusion and Future Research
The study, which was supported by the American Chemical Society PRF New Doctoral Investigator grant, the U.S. Department of Energy, and the Japanese Society for the Promotion of Science, marks a turning point in mineralogical research. It demonstrates that the answers to long-standing natural mysteries often lie at the intersection of classical geology, advanced computation, and innovative laboratory techniques.
Moving forward, the research team plans to investigate whether this "dissolution-assisted growth" theory applies to other minerals that are difficult to synthesize. There are several other carbonate and silicate minerals that exhibit similar growth problems. Furthermore, the industry application of the "pulsed growth" method will likely become a major area of focus for materials engineers looking to optimize the next generation of technological hardware.
By looking back at how the Earth built its most iconic mountain ranges, scientists have discovered a method that may help build the future of technology. The "Dolomite Problem," having stood for 222 years, is finally considered solved, opening a new chapter in our understanding of the crystalline world.














Leave a Reply