Eli Lilly and Company today announced groundbreaking results from a Phase 3 clinical trial for its investigational triple agonist, retatrutide, revealing an average weight loss of 16.8% in patients with type 2 diabetes. This significant outcome, alongside superior A1C reduction, positions retatrutide as a potentially transformative therapy in the challenging landscape of cardiometabolic health, addressing both glycemic control and weight management with unprecedented efficacy. The Indianapolis-based pharmaceutical giant shared that the drug met all primary and key secondary endpoints in the trial, demonstrating its robust potential for a patient population grappling with complex and often co-occurring metabolic conditions.
Unpacking the Clinical Data: A Dual Triumph in Glycemic and Weight Management
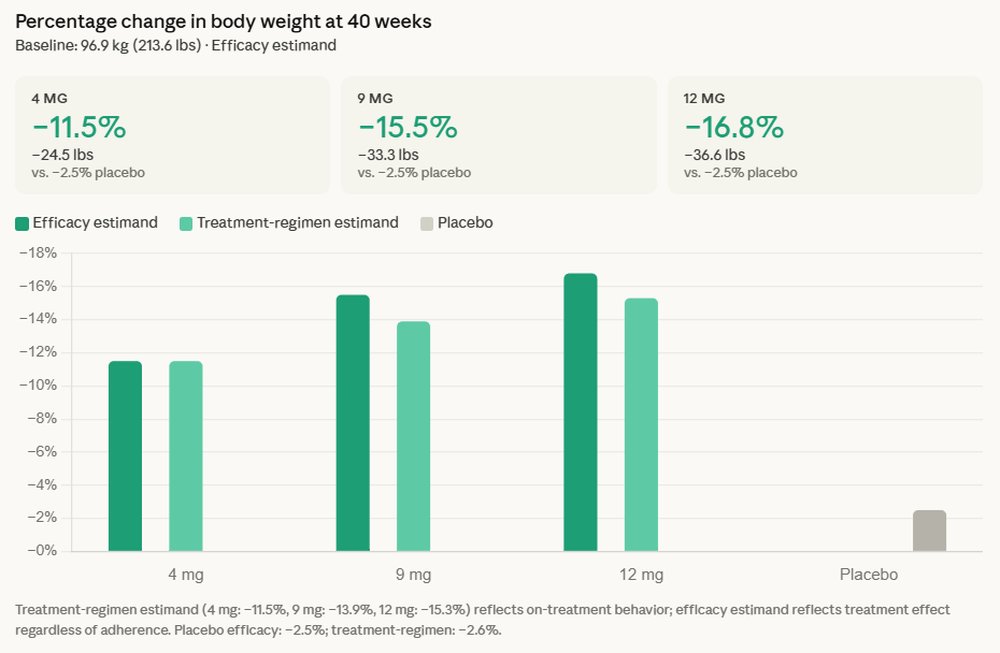
The Phase 3 trial, which focused on individuals with type 2 diabetes, showcased retatrutide’s remarkable ability to induce substantial improvements across critical metabolic parameters. Participants treated with retatrutide experienced an average weight loss of 16.8% over 40 weeks, translating to an average reduction of 36.6 pounds. A particularly noteworthy finding was the absence of a weight-loss plateau, with patients continuing their downward trajectory in body weight throughout the entire 40-week study period. This sustained efficacy is a crucial differentiator in a therapeutic area where initial weight loss often tapers off.
Beyond weight reduction, retatrutide delivered impressive glycemic control, achieving an average A1C reduction of up to 2.0% compared to placebo. A1C, or glycated hemoglobin, is a key marker reflecting average blood sugar levels over the preceding two to three months, and its substantial reduction is vital for preventing or delaying diabetes-related complications. The ability to simultaneously achieve such profound improvements in both weight and A1C levels is a testament to the drug’s multi-faceted mechanism of action.
Kenneth Custer, executive vice president and president of Lilly Cardiometabolic Health, underscored the significance of these findings. "For many people with type 2 diabetes, it is a struggle to achieve both A1C control and weight loss, since obesity has historically been harder to treat for those with type 2 diabetes," Custer stated in the company’s announcement. "With triple agonist retatrutide, we set out to make a molecule that could help patients achieve substantial A1C reduction and weight loss. These results support the potential of this molecule for people living with type 2 diabetes." His comments highlight the long-standing clinical challenge of managing the intertwined pathologies of type 2 diabetes and obesity, and the potential for retatrutide to offer a more comprehensive solution.
Beyond Glucose and Weight: Comprehensive Cardiometabolic Benefits
The positive impact of retatrutide extended beyond glycemic control and weight loss. The trial also revealed clinically meaningful improvements from baseline across several key cardiovascular risk factors. These included reductions in non-HDL cholesterol, triglycerides, and systolic blood pressure – all critical markers associated with increased risk of cardiovascular disease, which remains the leading cause of morbidity and mortality in patients with type 2 diabetes. This broader cardiometabolic benefit suggests that retatrutide could offer a holistic approach to managing the complex health profile of these patients, potentially reducing their overall risk burden.
The safety profile observed in the trial was consistent with that of other incretin-based therapies. Common adverse effects reported included nausea, diarrhea, and vomiting. These gastrointestinal side effects are frequently associated with drugs in this class and are generally manageable, often dose-dependent, and tend to diminish over time as patients adapt to treatment. The overall tolerability, combined with the profound efficacy, reinforces the drug’s potential for widespread clinical application.
The Science Behind the Breakthrough: A Novel Triple Agonist
Retatrutide is an investigational, first-in-class GIP, GLP-1, and glucagon triple hormone receptor agonist. This innovative mechanism of action differentiates it from existing treatments and is central to its exceptional efficacy. To understand its significance, it’s essential to briefly delve into the roles of these three gut hormones:
- Glucose-dependent insulinotropic polypeptide (GIP): A hormone that enhances glucose-dependent insulin secretion, promoting satiety, and influencing fat metabolism.
- Glucagon-like peptide-1 (GLP-1): Another incretin hormone that stimulates insulin release, suppresses glucagon secretion (which raises blood sugar), slows gastric emptying, and promotes satiety, leading to reduced food intake.
- Glucagon: While primarily known for raising blood sugar, glucagon also plays a role in energy expenditure and may contribute to weight loss by increasing metabolism and potentially affecting satiety pathways when modulated in combination with GIP and GLP-1.
By simultaneously activating receptors for all three of these hormones, retatrutide leverages multiple physiological pathways involved in glucose homeostasis, appetite regulation, and energy balance. This multi-pronged approach allows for a more comprehensive and potent metabolic modulation compared to single or dual agonists, leading to superior outcomes in both blood sugar control and weight reduction. This synergistic action is believed to be the driving force behind the unprecedented levels of weight loss and glycemic improvement observed in the trial.
The Global Burden of Type 2 Diabetes and Obesity: An Unmet Need
Type 2 diabetes and obesity represent two of the most pressing public health crises of the 21st century. According to the International Diabetes Federation (IDF), an estimated 537 million adults worldwide were living with diabetes in 2021, a number projected to rise to 783 million by 2045. The vast majority of these cases are type 2 diabetes, which is strongly linked to overweight and obesity. The World Health Organization (WHO) reports that globally, over 1 billion people are obese, a figure that has tripled since 1975.
These conditions not only reduce quality of life but also lead to a cascade of severe complications, including cardiovascular disease, kidney disease, neuropathy, retinopathy, and certain cancers. The economic burden is staggering, encompassing direct healthcare costs, lost productivity, and premature mortality. For decades, clinicians have struggled to effectively manage both blood sugar and weight in patients with type 2 diabetes. Traditional therapies often lead to modest weight loss or even weight gain, creating a vicious cycle where managing one condition exacerbates the other. The emergence of highly effective GLP-1 receptor agonists, and more recently, GIP/GLP-1 dual agonists like Eli Lilly’s own tirzepatide (Mounjaro/Zepbound), has begun to transform this landscape. Retatrutide’s even more potent results suggest a further paradigm shift, offering hope for patients who have exhausted other options or who require more aggressive intervention.
Lilly’s Expanding Footprint in the Cardiometabolic Arena
Eli Lilly has rapidly established itself as a frontrunner in the cardiometabolic space. The company’s GLP-1 receptor agonist, dulaglutide (Trulicity), has been a cornerstone in diabetes management for years. More recently, tirzepatide, a GIP/GLP-1 dual agonist, has achieved blockbuster status, first approved as Mounjaro for type 2 diabetes and then as Zepbound for chronic weight management. Tirzepatide itself demonstrated impressive weight loss (up to 22.5% in non-diabetic individuals with obesity) and A1C reductions, setting a new benchmark.
The successful Phase 3 results for retatrutide, a triple agonist, indicate that Lilly is pushing the boundaries even further. This strategic development positions Lilly to potentially dominate the evolving market for highly effective weight loss and diabetes drugs, which is projected to reach tens of billions of dollars annually. While Novo Nordisk, with its GLP-1 drugs semaglutide (Ozempic/Wegovy) and the investigational cagrisema (a combination of semaglutide and amylin analogue cagrilintide), is a fierce competitor, Lilly’s multi-agonist approach appears to be delivering superior efficacy. The competition between these pharmaceutical giants is driving rapid innovation, ultimately benefiting patients with more potent and comprehensive treatment options.
A Broader Therapeutic Horizon: Beyond Type 2 Diabetes
The current Phase 3 trial results specifically pertain to type 2 diabetes, but Lilly’s ambition for retatrutide extends far beyond this single indication. The company is engaged in a comprehensive Phase 3 clinical program, evaluating retatrutide’s potential efficacy and safety across a wide array of interconnected metabolic and obesity-related conditions. This extensive program includes studies in:
- Obesity and overweight with at least one weight-related medical problem: Building on the weight loss observed in diabetic patients, this is a natural next step to address the primary obesity market.
- Knee osteoarthritis: Obesity is a major risk factor for osteoarthritis, and significant weight loss could alleviate symptoms and slow disease progression.
- Moderate-to-severe obstructive sleep apnea: A common complication of obesity, often improved with weight reduction.
- Chronic low back pain: Another condition frequently exacerbated by excess weight.
- Cardiovascular and renal outcomes: Crucial studies to determine if the improvements in risk factors translate into hard outcomes like reduced heart attacks, strokes, and kidney failure.
- Metabolic dysfunction-associated steatotic liver disease (MASLD), formerly NAFLD: A liver condition strongly linked to obesity and insulin resistance, for which effective pharmacological treatments are urgently needed.
This broad investigative pipeline underscores Lilly’s confidence in retatrutide’s potential as a foundational metabolic therapeutic. Positive results in these additional indications could position retatrutide as a multi-purpose drug capable of addressing several chronic conditions simultaneously, profoundly impacting public health and healthcare economics.
Looking Ahead: Regulatory Pathway and Market Impact
Following these robust Phase 3 results, Eli Lilly is expected to move swiftly towards regulatory submissions. Typically, after successful completion of Phase 3 trials, companies compile extensive data packages for review by regulatory bodies such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA). While the exact timeline is subject to the review process, an approval for type 2 diabetes could potentially occur within the next 12-24 months, with subsequent approvals for other indications following suit as their respective trials mature.
The market implications of retatrutide’s potential approval are substantial. Given the high prevalence of type 2 diabetes and obesity globally, and the superior efficacy demonstrated, retatrutide could quickly become a major revenue driver for Eli Lilly. Industry analysts are likely to revise their forecasts upwards for Lilly’s cardiometabolic portfolio, recognizing the drug’s potential to capture significant market share and expand the overall market for highly effective weight loss and diabetes treatments.
However, the introduction of such potent therapies also raises questions regarding accessibility and cost. These novel drugs are typically expensive, and ensuring equitable access for all patients who could benefit will be a critical challenge for healthcare systems and policymakers worldwide. Long-term safety data, particularly beyond the 40-week duration of this trial, will also be crucial for a full understanding of the drug’s profile and its role in chronic disease management.
In conclusion, Eli Lilly’s retatrutide has emerged from its Phase 3 trial for type 2 diabetes with data that signals a significant leap forward in metabolic medicine. Its ability to deliver unprecedented weight loss and profound A1C reduction, coupled with improvements in cardiovascular risk factors, positions it as a potential game-changer. As the company continues its extensive clinical development program, the medical community and patients alike will be eagerly anticipating its journey to market, hopeful for a new era of comprehensive and highly effective treatment for type 2 diabetes and its myriad comorbidities.












Leave a Reply