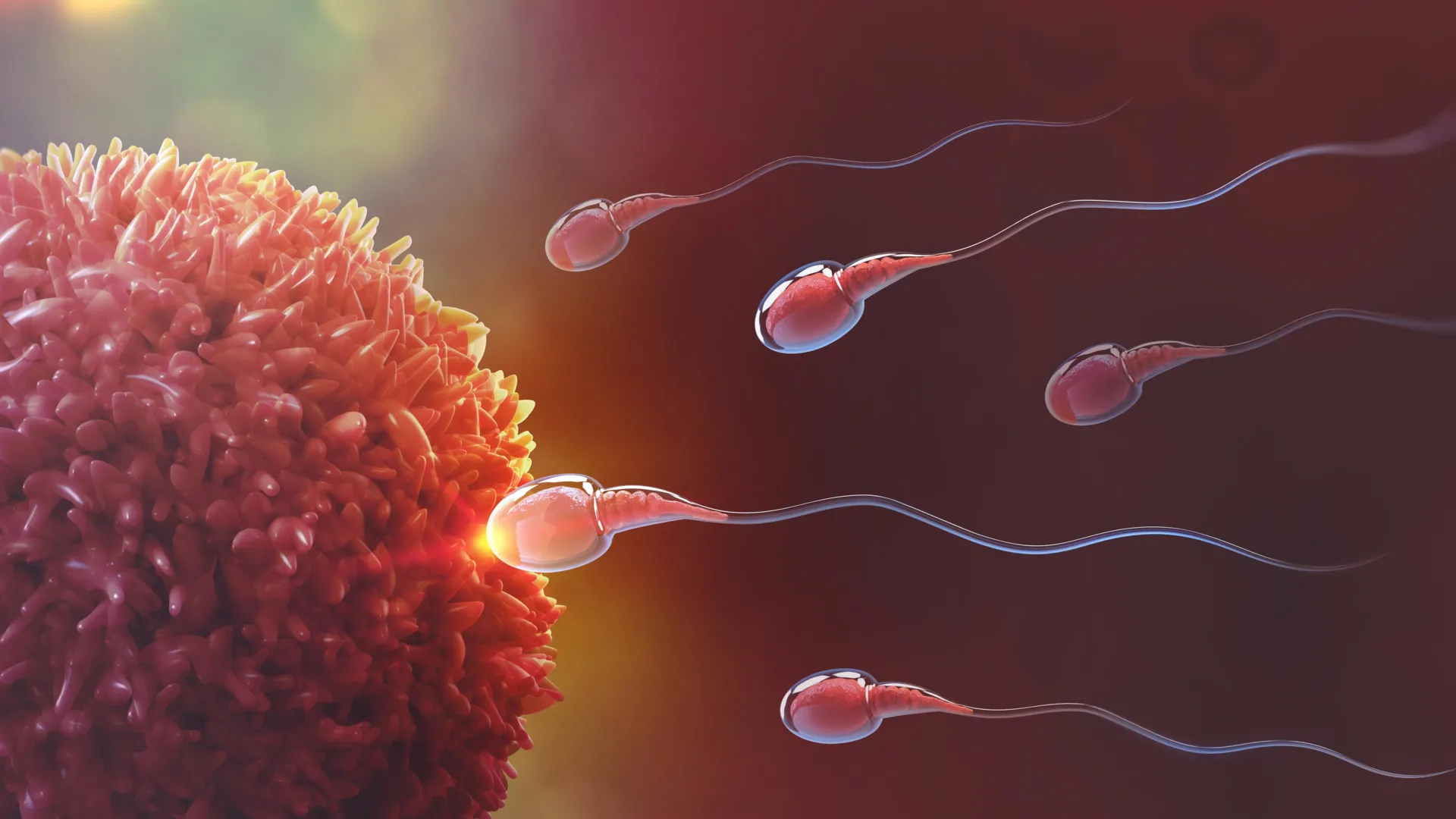
In vitro fertilization (IVF) has long been a cornerstone of both human reproductive medicine and global agricultural productivity. However, despite decades of refinement, the process remains fraught with variables that can lead to failure. One of the most persistent challenges in the laboratory setting is the rapid decline of sperm viability once samples are removed from the natural reproductive tract. Researchers at the University of Illinois Urbana-Champaign have recently published a study in Scientific Reports that addresses this hurdle by mimicking the natural environment of the female reproductive system. By utilizing specific complex sugars known as glycans, the team has developed a method to select high-quality sperm and significantly prolong their functional lifespan, potentially revolutionizing the efficiency of IVF across species.
The study, led by David Miller, a professor in the Department of Animal Sciences at the University of Illinois, focuses on the oviduct—commonly known as the fallopian tube in humans. In a natural setting, the oviduct does far more than serve as a conduit for eggs and sperm; it acts as a sophisticated biological reservoir. It possesses the innate ability to bind sperm, maintaining their viability and regulating their release to ensure they are ready for fertilization at the precise moment of ovulation. Until now, recreating this protective "sperm reservoir" in a laboratory setting has proven elusive, leading to a narrow window of opportunity for successful fertilization during IVF procedures.
The Role of Glycobiology in Reproductive Success
The foundation of this breakthrough lies in the 2020 discovery by Miller’s team that complex sugars, or glycans, are the primary components of the oviduct responsible for sperm storage. These glycans act as molecular anchors, binding to the surface of the sperm and keeping them in a quiescent yet viable state. For the current study, the researchers collaborated with specialized chemists to screen hundreds of different oviduct glycans. Their goal was to identify which specific sugar structures possessed the highest affinity for sperm binding.
After extensive testing, the team identified a specific compound: sulfated Lewis X trisaccharide, or suLeX. This glycan demonstrated a superior ability to bind pig sperm, which served as the primary model for the research. The choice of porcine subjects was strategic; pig reproduction is a vital component of the global food supply chain, and pig IVF faces a unique challenge known as polyspermy. Polyspermy occurs when multiple sperm fertilize a single egg, a phenomenon that results in inviable embryos and significant economic loss for producers. By using suLeX to tether sperm to the surface of a culture dish, the researchers aimed to regulate the number of free-swimming sperm, thereby reducing the risk of multiple penetrations.
Experimental Methodology and Chronology
The research team established a rigorous experimental timeline to test the efficacy of suLeX in maintaining sperm longevity. The process began by coating the bottom of laboratory culture dishes with the suLeX glycan. Sperm samples were introduced to these dishes and allowed 30 minutes to adhere to the sugar molecules. Once the sperm were securely bound, the researchers introduced eggs at four distinct intervals: 0, 6, 12, and 24 hours.
This chronological approach was designed to simulate the "timing mismatch" often encountered in clinical and agricultural IVF. In many cases, sperm may be ready for fertilization before the harvested eggs have reached full maturity, or vice versa. By introducing eggs at later time points, the researchers could measure how effectively the suLeX-bound sperm maintained their fertilization potential compared to traditional methods.
To further refine the process, the researchers implemented a "wash" step. Because the viable, high-quality sperm were bound to the suLeX-coated surface, the team could wash away the excess, free-swimming sperm that did not adhere. This ensured that only the sperm with the specific receptors necessary for glycan binding—often an indicator of higher health and maturity—remained in the dish when the eggs were finally introduced.
Supporting Data: A Significant Leap in Efficiency
The results of the study provided clear, quantifiable evidence of the benefits of glycan-integrated IVF. At the initial 0-hour mark, the IVF efficiency—defined as the ratio of successfully fertilized zygotes to the total number of eggs—was 53% for the suLeX-treated group. In contrast, the control group, which utilized standard IVF culture dishes without oviduct compounds, showed an efficiency of only 36%. Two other alternative "control" compounds were also tested, both yielding efficiency rates of approximately 40%, further highlighting the specific effectiveness of suLeX.
The most dramatic findings emerged during the extended time trials. As time passed, fertilization rates naturally declined across all groups as sperm lost their motility and structural integrity. However, the suLeX group maintained a significantly higher success rate than the control. By the 24-hour mark, the control group’s fertilization rate had plummeted to a mere 1%, effectively rendering the samples useless. In stark contrast, the suLeX-bound sperm maintained a 12% fertilization rate after 24 hours. While a 12% rate may seem modest in isolation, in the context of IVF, it represents a twelvefold increase in the window of viability, offering a critical buffer for laboratory technicians and clinicians.
Reducing Polyspermy and Improving Embryo Quality
Beyond simply extending the lifespan of the sperm, the suLeX method addressed the critical issue of embryo quality. In porcine IVF, the high concentration of sperm required to ensure fertilization often leads to the aforementioned polyspermy. By binding the sperm to the glycan-coated surface and washing away the excess, the researchers were able to control the "sperm-to-egg ratio" more effectively.
"Because the sperm were bound securely to the glycan compound, we could reduce the overall number of sperm, which meant fewer cases where more than one sperm fertilized the eggs," Miller explained. This reduction in polyspermy leads to a higher yield of healthy, diploid embryos, which are necessary for successful pregnancy and live birth. This aspect of the research is particularly relevant to the livestock industry, where the production of high-genetic-merit embryos is a multi-million dollar enterprise.
Broader Implications for Agriculture and Human Medicine
The implications of this study extend far beyond the laboratory. In the agricultural sector, particularly in dairy and beef production, IVF is used to propagate the genetics of elite animals. These "high-genetic-merit" embryos result in livestock that can produce milk more efficiently or reach market weight with fewer resources, contributing to global food security and environmental sustainability.
"There are companies, especially related to dairy cattle, that use IVF to produce and sell high-genetic-merit embryos that, after they are delivered, will produce milk more efficiently," Miller noted. "This technology could potentially help produce meat and milk more efficiently." By increasing the success rate of each IVF cycle, producers can lower costs and accelerate the genetic improvement of their herds.
In the realm of human medicine, the discovery offers a promising path toward improving the success rates of fertility treatments. Human IVF is often an emotionally and financially draining process, with success rates hovering around 30% per cycle depending on the age and health of the patients. One of the primary causes of failure is the "timing mismatch" between sperm preparation and egg readiness.
Miller pointed out that while the specific glycans that bind human sperm have not yet been identified, the methodology established in this porcine study provides a roadmap for future human-centric research. "Both eggs and sperm have to undergo a maturation phase before they’re ready for fertilization, so the timing is critical," Miller said. "There’s variability in the time it takes sperm to complete their final major maturation step. We think glycan-IVF could lengthen the fertile window of sperm and possibly increase IVF rates."
Analysis of Future Research and Challenges
While the study marks a significant milestone, several hurdles remain before glycan-based IVF becomes a standard clinical practice. The primary challenge lies in the diversity of glycans across different species. The suLeX compound worked exceptionally well for porcine sperm, but human reproductive biology may rely on a different set of complex sugars. Identifying these specific human glycans will require extensive screening and collaboration between reproductive biologists and glycochemists.
Furthermore, the transition from a laboratory culture dish to a commercial IVF kit requires rigorous testing for safety and consistency. The researchers must ensure that the glycan coating does not interfere with the long-term development of the embryo or the health of the resulting offspring. However, the foundational nature of this study, supported by the Eunice Kennedy Shriver National Institute of Child Health and Human Development (part of the NIH), suggests a strong institutional belief in the potential of this technology.
The study, titled "Porcine sperm bind to an oviduct glycan coupled to glass surfaces as a model of sperm interaction with the oviduct," represents a shift toward more "bio-inspired" reproductive technologies. By looking to the natural mechanisms of the oviduct, Miller and his team have found a way to bridge the gap between the artificial environment of the lab and the highly optimized environment of the body.
Conclusion
The work of the University of Illinois researchers provides a compelling example of how basic biological research can lead to practical technological advancements. By identifying suLeX as a key player in sperm longevity, the team has opened a new door for improving the efficiency of IVF. Whether applied to the goal of increasing sustainable food production in the livestock industry or helping families achieve their dreams of parenthood, the integration of glycobiology into reproductive medicine promises to make the process more predictable, more successful, and more reflective of the natural miracle of life. As researchers move toward identifying human-specific glycans, the window of opportunity for successful fertilization—and the hope that comes with it—continues to expand.












Leave a Reply