In a landmark study published in The Lancet Digital Health, researchers from the USC Mark and Mary Stevens Neuroimaging and Informatics Institute (Stevens INI) have uncovered a remarkable neurological phenomenon: the human brain appears to undergo a structural "rejuvenation" in specific undamaged regions following a severe stroke. This finding, which suggests a profound and paradoxical form of neuroplasticity, provides a new window into how the central nervous system attempts to compensate for catastrophic tissue loss. By utilizing advanced artificial intelligence and a massive global database of brain scans, the research team demonstrated that while a stroke accelerates aging in the hemisphere where the injury occurred, the opposite, undamaged hemisphere may adopt a structural signature that appears biologically "younger" than the patient’s chronological age.
This study represents a significant leap forward in our understanding of stroke recovery, particularly for patients who suffer from chronic, long-term physical impairments. Traditionally, stroke research has focused primarily on the site of the lesion—the area where blood flow was restricted and neurons died. However, this new data suggests that the brain’s response is a whole-organ event, involving complex reorganization across both hemispheres. The discovery of "younger" brain structures in the contralesional side (the side opposite the stroke) points toward a compensatory mechanism where the brain attempts to rewire itself to take over lost functions, even if those attempts do not result in a full return of motor control.
The Global Burden of Stroke and the Search for Biomarkers
Stroke remains one of the leading causes of long-term disability worldwide. According to the World Health Organization (WHO), over 15 million people suffer a stroke each year, with approximately five million left permanently disabled. Despite advancements in acute care—such as clot-busting drugs and mechanical thrombectomies—the long-term rehabilitation process remains a challenge. Many patients reach a plateau in their recovery approximately six months after the initial event, entering what is known as the "chronic phase."
Understanding why some patients recover more mobility than others is a primary goal of modern neurology. Historically, clinicians relied on physical examinations and standard MRI scans to assess damage. However, these tools often fail to capture the subtle, microscopic changes in neural networks that dictate recovery potential. The Stevens INI study addresses this gap by introducing "brain age" as a biomarker. By comparing a patient’s biological brain age to their actual chronological age, researchers can quantify the "brain-predicted age difference" (brain-PAD). A positive brain-PAD indicates a brain that looks older than it should, often a sign of neurodegeneration or poor health, while a negative brain-PAD suggests a brain that has maintained a more youthful, resilient structure.
Methodology: Harnessing Big Data and Artificial Intelligence
The scale of this research was made possible through the Enhancing NeuroImaging Genetics through Meta-Analysis (ENIGMA) Stroke Recovery Working Group. ENIGMA is a global consortium that pools neuroimaging data from over 50 countries, allowing researchers to study brain conditions with a level of statistical power that individual labs cannot achieve. For this specific study, the team analyzed MRI scans from 535 stroke survivors across 34 research centers in eight different countries.
To process this vast amount of data, the researchers employed a sophisticated type of artificial intelligence known as a graph convolutional network (GCN). Unlike traditional AI models that treat images as flat grids of pixels, GCNs are designed to understand the complex, non-linear relationships between different points in a network—much like the interconnected regions of the human brain. The AI was first trained on a massive baseline of tens of thousands of MRI scans from healthy individuals to learn the typical structural changes associated with normal aging.
Once trained, the AI was tasked with estimating the biological age of 18 specific brain regions in each stroke survivor. By comparing these estimates to the survivors’ actual ages and their motor function scores, the researchers were able to map out how a stroke redistributes the "age" of the brain.
The Paradox of Contralesional Rejuvenation
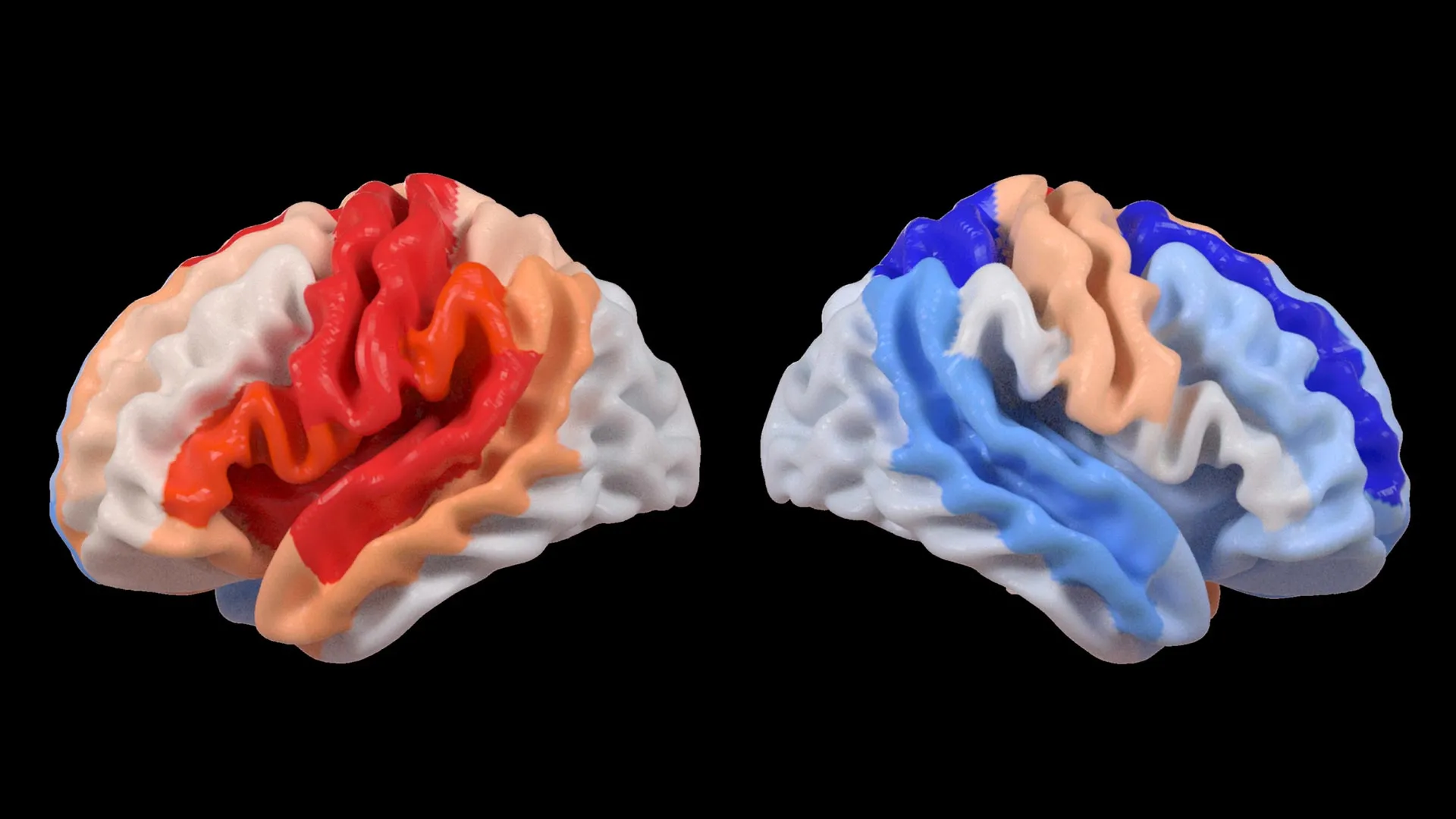
The most striking finding of the study was the divergence between the two hemispheres of the brain. In the hemisphere where the stroke occurred (the ipsilesional side), the AI detected signs of accelerated aging. This was expected, as the death of neurons and the subsequent inflammation and tissue atrophy naturally make the brain appear more degraded.
However, the opposite side of the brain (the contralesional side) told a different story. In survivors with the most severe motor impairments, the undamaged hemisphere appeared significantly "younger" than their actual age. This phenomenon was most pronounced in the frontoparietal network, a critical circuit involved in motor planning, coordination, and attention.
"We found that larger strokes accelerate aging in the damaged hemisphere but paradoxically make the opposite side of the brain appear younger," stated Hosung Kim, PhD, associate professor of research neurology at the Keck School of Medicine of USC and co-senior author of the study. Dr. Kim explained that this pattern suggests a massive reorganization of neural resources. Essentially, the brain may be "rejuvenating" undamaged networks to compensate for the functions lost in the damaged side.
This "youthful" signature is not necessarily a sign of a return to health, but rather an indicator of intense neuroplasticity. When the primary motor pathways are destroyed, the brain may recruit these "younger" appearing regions to attempt to bridge the gap. In patients with severe impairment, this recruitment is more desperate and visible, though it may not be enough to restore full movement.
Chronology of Recovery and the Role of the Frontoparietal Network
The study specifically looked at patients in the chronic stage of recovery—those who were at least six months post-stroke. This timeline is crucial because it is the period during which the brain’s initial inflammatory response has subsided, and the long-term structural changes have solidified.
The focus on the frontoparietal network is also significant. In healthy individuals, this network helps manage complex tasks by integrating sensory input and planning motor output. When a stroke occurs in the primary motor cortex, the frontoparietal network in the opposite hemisphere often becomes hyperactive. The USC study provides the first structural evidence that this hyperactivity is accompanied by a physical change in the brain’s architecture, making it look more like the brain of a younger person who has a higher capacity for learning and adaptation.
This discovery challenges the traditional view of the "static" chronic brain. It suggests that even months or years after a stroke, the brain remains in a state of flux, actively trying to reorganize its resources.
Expert Reactions and the Move Toward Personalized Rehabilitation
The implications of this research have resonated throughout the neurological community. Experts suggest that the ability to measure regional brain age could lead to a new era of "precision rehabilitation." Currently, stroke rehabilitation is often a one-size-fits-all approach, involving physical and occupational therapy based on visible symptoms.
Arthur W. Toga, PhD, director of the Stevens INI and Provost Professor at USC, emphasized the power of the AI-driven approach. "By pooling data from hundreds of stroke survivors worldwide and applying cutting-edge AI, we can detect subtle patterns of brain reorganization that would be invisible in smaller studies," Toga said. He noted that these findings could eventually guide personalized strategies, where therapies—such as non-invasive brain stimulation or specific motor exercises—are tailored to the specific "age" and plasticity of a patient’s brain regions.
Independent observers in the field of neurorehabilitation have noted that this study provides a structural explanation for why some patients respond better to certain therapies than others. If a patient’s contralesional frontoparietal network shows a "younger" signature, they may have a higher "plasticity reserve," making them better candidates for intensive neuro-rehabilitative interventions.
Future Implications and Longitudinal Research
While the current study provides a snapshot of the brain in the chronic phase, the researchers at USC Stevens INI are already planning the next steps. One of the primary goals is to conduct longitudinal studies that track patients from the moment of their stroke through the various stages of recovery. By observing how brain-PAD changes over weeks and months, doctors may be able to predict which patients are at risk for poor outcomes and intervene earlier.
Furthermore, this research opens the door to studying how other factors—such as genetics, pre-existing health conditions like diabetes or hypertension, and even social determinants of health—influence the brain’s ability to "rejuvenate" itself after an injury. The use of the ENIGMA dataset ensures that these future findings will be applicable to a diverse global population, rather than a limited demographic.
The study also raises intriguing questions about the limits of neuroplasticity. If the brain can make certain regions appear "younger" to compensate for damage, are there ways to pharmacologically or technologically enhance this process? The intersection of AI, neuroimaging, and regenerative medicine is likely where the next generation of stroke treatments will be born.
Conclusion
The findings published in The Lancet Digital Health mark a pivotal shift in stroke neurology. By moving beyond the site of the injury and looking at the global "age" of the brain, the USC Stevens INI team has revealed a hidden landscape of adaptation and resilience. The discovery that severe impairment triggers a youthful structural shift in undamaged brain regions provides a powerful testament to the brain’s enduring ability to change, even in the face of profound trauma. As AI continues to refine our understanding of these complex neural patterns, the hope for more effective, personalized stroke recovery becomes increasingly tangible. For the millions of survivors worldwide living with the long-term effects of stroke, this research offers not just a better understanding of their condition, but a roadmap toward more targeted and effective therapies in the future.














Leave a Reply