A groundbreaking discovery by a research team spearheaded by Universitat Autònoma de Barcelona (UAB) scientist David Reverter has unveiled the intricate molecular mechanism governing bacterial cell division. This pivotal research, published in the prestigious journal Nature Communications, details how the MraZ protein orchestrates this fundamental biological process by binding to the critical dcw gene cluster. The findings offer unprecedented insight into the precise regulation of bacterial reproduction, a process essential for life and a target for therapeutic interventions.
The Genesis of Bacterial Reproduction: The dcw Operon
Cell division, the fundamental process by which all living organisms grow and reproduce, relies on a highly coordinated symphony of protein interactions and regulatory elements. In the vast majority of bacterial species, the genetic blueprint for this essential function is consolidated within a specific region of DNA known as the dcw operon. This gene cluster acts as a central command center, encoding the proteins necessary for both the physical act of dividing the cell and the crucial construction of the bacterial cell wall, the protective outer layer that maintains cell integrity. Understanding how this operon is activated is paramount to comprehending bacterial life cycles.
Historically, the activation of genes within operons is controlled by proteins called transcription factors. These molecular architects bind to specific DNA sequences known as promoters, which serve as the starting point for gene transcription – the process of copying genetic information from DNA into RNA. The promoter region typically precedes the start codon, the three-nucleotide sequence that signals the initiation of protein synthesis.
Within the dcw operon, the MraZ protein holds a position of supreme importance. It is not only a key transcription factor but also the very first gene within the operon in virtually all bacteria. When MraZ is activated, it triggers the transcription of the entire dcw operon, leading to the production of the essential proteins required for bacterial cell division. Consequently, MraZ acts as the master switch, the primary regulator that dictates the activity of the operon responsible for cell division across a broad spectrum of bacterial species. Its precise function, however, remained elusive until the recent work by Reverter’s team.
Illuminating the Mechanism: Advanced Imaging Techniques
The UAB research group, under the leadership of David Reverter, a distinguished full professor in the Department of Biochemistry and Molecular Biology and a researcher at the Institute of Biotechnology and Biomedicine of the UAB (IBB-UAB), has succeeded in precisely mapping the molecular dance of MraZ and the dcw operon. Employing cutting-edge structural biology methodologies, including X-ray crystallography and cryo-electron microscopy (cryo-EM), the scientists were able to visualize these molecular interactions with unprecedented clarity.
These sophisticated techniques allowed the researchers to meticulously determine how the MraZ transcription factor interacts with the promoter region of the dcw operon. For their detailed investigation, the team focused on the bacterium Mycoplasma genitalium. This microorganism is a favored subject in laboratory research due to its exceptionally small and streamlined genome, which simplifies the analysis of complex genetic pathways. By studying this model organism, the researchers aimed to establish fundamental principles applicable to a wider range of bacterial species.
An Atomic-Level Glimpse: MraZ’s Remarkable Transformation
The promoter region of the dcw operon, the crucial DNA sequence that initiates transcription, contains a distinctive feature: four repetitive segments, often referred to as "boxes." Each of these boxes is comprised of six nucleotides, the building blocks of DNA. These repeating sequences are critical for the precise regulation of gene expression, acting as binding sites for transcription factors like MraZ.
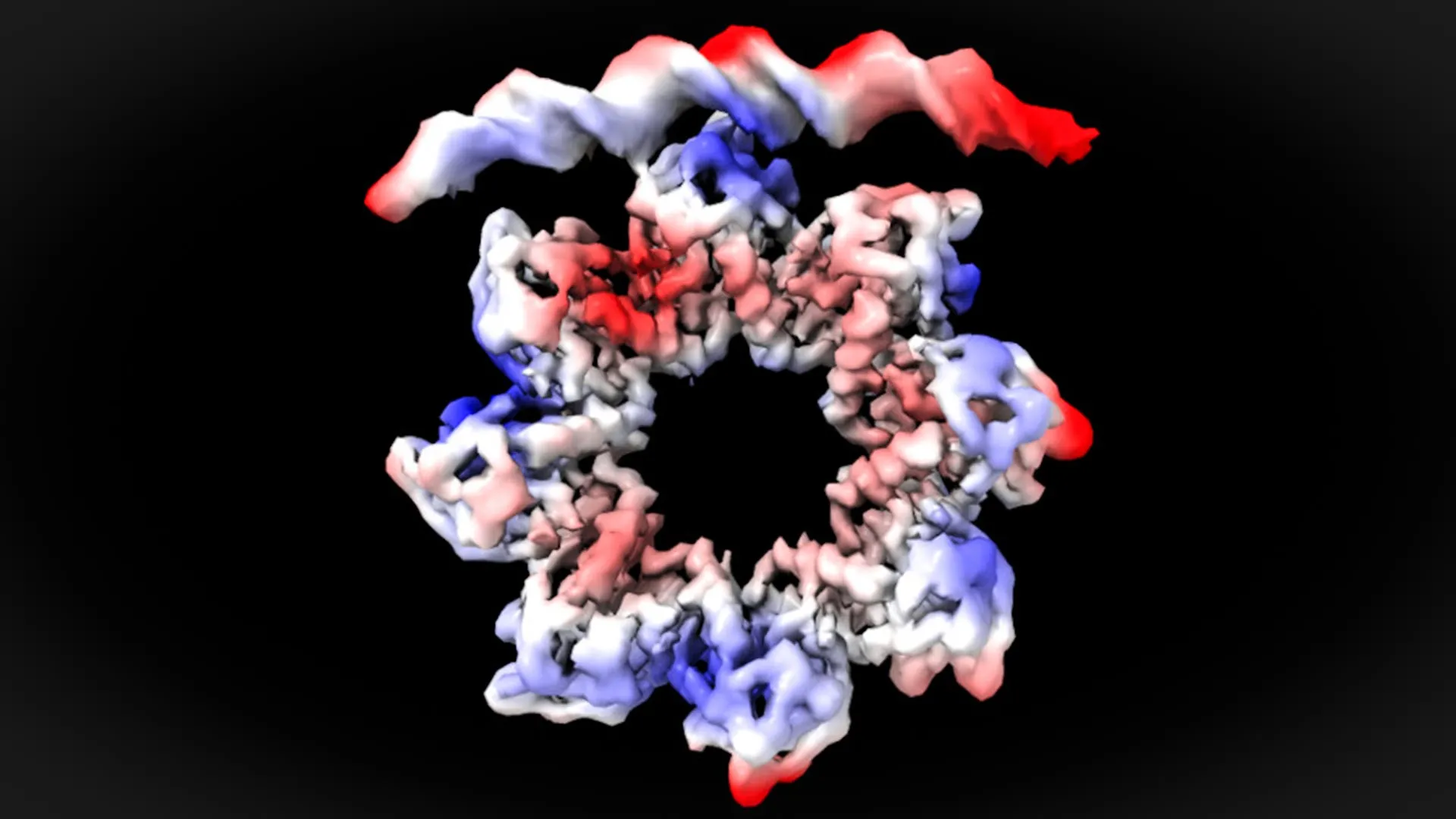
Through the application of cryo-electron microscopy, the researchers achieved a near-atomic resolution view of the interaction between the MraZ protein and the DNA bases within these four repeated boxes. The observations revealed a surprising and dynamic process: MraZ must undergo a significant structural alteration to successfully bind to the operon and initiate cell division.
"This is a surprising observation," stated David Reverter. "The MraZ protein normally exists as an octamer – a complex formed by eight identical subunits assembled in a donut-like structure. However, its inherent curvature would typically prevent it from binding to the four distinct ‘boxes’ on the promoter DNA. Yet, to regulate cell division, we observe how this donut-shaped structure undergoes a remarkable transformation. It breaks apart and deforms in such a way that four of its subunits can precisely align and attach to the four specific boxes on the promoter." This conformational change is a critical step, allowing MraZ to exert its regulatory control.
A Paradigm Shift in Understanding Bacterial Division
The ability to directly visualize the molecular mechanism by which MraZ interacts with the promoter DNA that initiates cell division represents a monumental leap forward in the field. Prior to this study, researchers relied heavily on indirect evidence gleaned from biochemical experiments and computational modeling to infer the workings of this complex system. While these methods provided valuable insights, they lacked the direct, visual confirmation that structural biology techniques now afford. This new evidence solidifies hypotheses and opens avenues for more targeted investigations.
The implications of this discovery extend far beyond the specific bacterium studied. According to Reverter, the regulatory system involving MraZ and the dcw operon is likely to be conserved across a vast majority of bacterial species. "This regulatory system is likely universal to most bacteria," Reverter explained. "This is because all MraZ proteins exhibit significant structural similarity, maintaining the same octameric structure. Furthermore, the DNA sequences of the promoters of the operons that regulate cell division are also remarkably similar across diverse bacterial phyla." This universality suggests that the fundamental principles uncovered by this research have broad applicability in understanding bacterial biology.
A Collaborative Endeavor: Bridging Institutions and Expertise
The seminal research was spearheaded by David Reverter’s dedicated team at the Institute of Biotechnology and Biomedicine and the Department of Biochemistry and Molecular Biology at UAB. The success of this complex project was further amplified through invaluable collaboration with the ALBA synchrotron, a leading facility for synchrotron light generation in Spain, and the cryo-electron microscopy service at the Institute of Genetics and Molecular and Cellular Biology in Strasbourg, France. This international and interdisciplinary collaboration brought together diverse expertise and state-of-the-art instrumentation, underscoring the global nature of modern scientific discovery.
Broader Implications and Future Directions
The precise understanding of bacterial cell division regulation has profound implications across various scientific and medical domains.
Antimicrobial Drug Development
Bacterial cell division is a critical process for bacterial survival and proliferation. Disrupting this process is a highly effective strategy for developing new antimicrobial agents. By elucidating the precise molecular targets and mechanisms involved, this research provides invaluable information for the design of novel antibiotics that specifically inhibit MraZ function or its interaction with the dcw operon. Such targeted therapies could overcome existing antibiotic resistance mechanisms, a growing global health crisis. The identification of the structural requirements for MraZ binding offers a direct avenue for designing molecules that either mimic or block this interaction.
Understanding Bacterial Diversity and Evolution
The universality of the MraZ-dcw operon regulatory system across diverse bacterial species offers insights into bacterial evolution. The conservation of this pathway suggests it is a fundamental mechanism that has been preserved over evolutionary time due to its critical role in bacterial fitness. Further comparative studies across different bacterial lineages could reveal subtle variations and adaptations in this system, shedding light on the evolutionary pressures that have shaped bacterial diversity. For instance, investigating why certain bacteria might have slightly different promoter sequences or MraZ variants could reveal novel regulatory strategies.
Fundamental Biological Research
Beyond its applied implications, this research significantly advances our fundamental understanding of gene regulation and protein-DNA interactions. The discovery of the MraZ octamer’s dynamic conformational change highlights the plasticity of protein structures and their ability to adapt to specific DNA binding sites. This finding contributes to the broader field of structural biology and provides a compelling case study for how proteins achieve specificity in their interactions. It also offers a powerful example of how advanced imaging techniques are revolutionizing our ability to observe molecular processes in action.
Potential for Therapeutic Interventions in Human Health
While the current research focuses on bacteria, understanding fundamental cellular processes in model organisms can sometimes illuminate analogous pathways in more complex organisms, including humans. Although human cell division is far more complex, the principles of protein-DNA interaction and transcriptional regulation are universal. Future research might explore whether similar regulatory mechanisms or protein families exist in eukaryotic systems, potentially opening doors for novel therapeutic strategies in areas beyond infectious diseases. However, it is crucial to emphasize that direct applications in human health are currently speculative and require extensive further investigation.
The work by David Reverter and his team represents a significant milestone in our quest to understand the fundamental processes of life. By dissecting the molecular choreography of bacterial cell division, they have not only provided a clearer picture of bacterial reproduction but have also laid the groundwork for future innovations in medicine and biology. The detailed atomic-level visualization of MraZ’s transformative binding event serves as a testament to the power of scientific inquiry and the relentless pursuit of knowledge. The ongoing research into this critical bacterial pathway promises to yield further insights with far-reaching consequences.













Leave a Reply