A groundbreaking discovery by a research team spearheaded by David Reverter, a scientist at the Universitat Autònoma de Barcelona (UAB), has illuminated the intricate molecular mechanism governing bacterial cell division. The findings, published in the prestigious journal Nature Communications, detail precisely how the MraZ protein orchestrates this fundamental biological process by binding to the critical dcw gene cluster. This revelation provides an unprecedented atomic-level understanding of a process essential for the proliferation of nearly all life on Earth.
The Fundamental Dance of Bacterial Reproduction
Cell division, the process by which a single cell divides into two or more daughter cells, is a cornerstone of life. For bacteria, this mechanism is not merely about reproduction; it’s about population growth, adaptation, and the very survival of the species. This complex process is a symphony of molecular interactions, a precisely choreographed dance involving numerous proteins and regulatory elements working in concert. In the vast majority of bacterial species, the genetic blueprint for this essential function is encoded within a specific group of genes known as the dcw operon. This operon is not just a collection of genes; it’s a highly organized genetic unit containing the instructions necessary for producing proteins that are vital for both the physical act of splitting the cell and the construction of its protective outer layer, the cell wall. The integrity and proper functioning of the cell wall are paramount, as it provides structural support and protects the bacterium from environmental stresses. Disruptions to this delicate balance can lead to cell lysis or incomplete division, ultimately hindering bacterial survival and propagation.
The Maestro of the dcw Operon: MraZ’s Commanding Presence
The activation of the genes within the dcw operon, and consequently the initiation of cell division, is orchestrated by a class of proteins known as transcription factors. These molecular architects bind to specific DNA sequences called promoters, which serve as the starting signals for gene transcription – the process of copying genetic information from DNA into RNA. The promoter region is typically located just upstream of the gene’s coding sequence, acting as a molecular switch that dictates when and how strongly a gene is expressed.
Among these crucial transcription factors, MraZ holds a position of particular significance. Intriguingly, MraZ is itself the very first gene within the dcw operon across virtually all bacterial species. This self-regulatory arrangement underscores its pivotal role. When MraZ successfully activates the operon, it initiates a cascade of gene expression, leading to the production of the suite of proteins required for bacteria to undergo division. In essence, MraZ acts as the master regulator, the conductor of the molecular orchestra that governs cell division in the majority of bacterial life forms. Its presence and activity are indispensable for the perpetuation of bacterial populations.
Illuminating the Molecular Mechanism: A Glimpse into Atomic Precision
The research team at the UAB, under the astute leadership of David Reverter, a distinguished full professor in the Department of Biochemistry and Molecular Biology and a researcher at the Institute of Biotechnology and Biomedicine of the UAB (IBB-UAB), has meticulously unraveled the detailed molecular mechanisms underlying this critical regulation. To achieve this remarkable feat, the scientists employed state-of-the-art structural biology techniques, including X-ray crystallography and cryo-electron microscopy (cryo-EM). These advanced methods offer unparalleled resolution, allowing researchers to visualize the three-dimensional structures of molecules and their interactions with exquisite detail.
These powerful techniques enabled the scientists to pinpoint precisely how the MraZ transcription factor interacts with and binds to the promoter region of the dcw operon within the bacterium Mycoplasma genitalium. This particular microorganism was selected for study due to its exceptionally small genome, which simplifies genetic analysis and provides a clear model system for understanding fundamental biological processes. The relative simplicity of M. genitalium‘s genetic makeup makes it an ideal candidate for elucidating conserved molecular mechanisms that are likely to be present in a wider range of bacterial species.
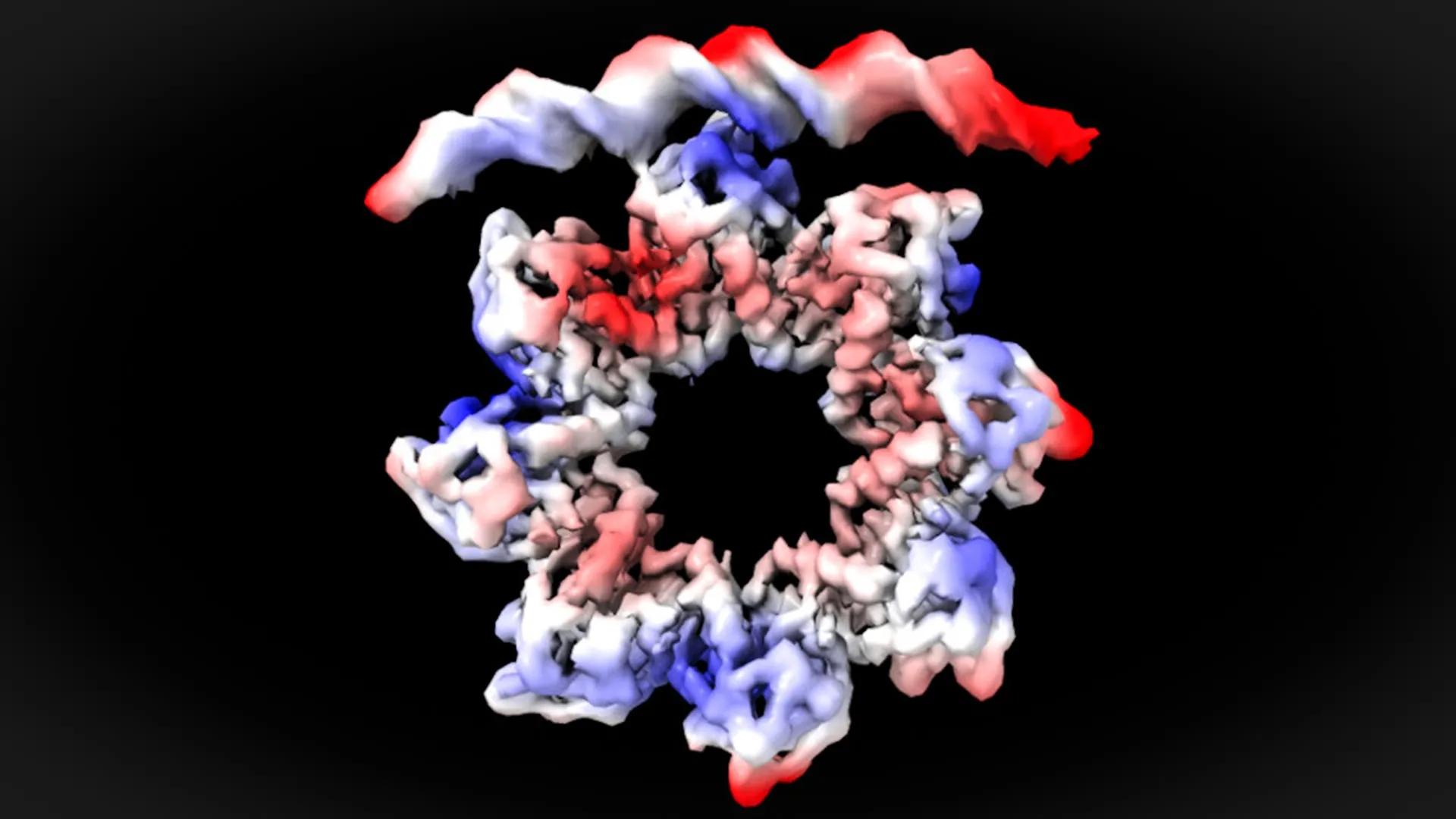
An Atomic-Level Blueprint: MraZ’s Unexpected Molecular Ballet
The promoter region of the dcw operon, the specific DNA sequence that MraZ targets, is characterized by the presence of four repeated segments, often referred to as "boxes." Each of these boxes is composed of six nucleotide bases, the fundamental building blocks of DNA. These repetitive DNA sequences are not accidental; they are crucial for the precise regulation of transcription initiation, ensuring that the dcw operon is activated at the appropriate time and under the correct cellular conditions.
Through the application of cryo-electron microscopy, the researchers were able to achieve an astonishingly high-resolution view of the interaction between the MraZ protein and the DNA bases within these four repeated boxes. Their observations revealed a surprising and dynamic molecular maneuver. For MraZ to successfully bind to the promoter, it must undergo a significant structural transformation.
"This is a surprising observation," stated David Reverter, elaborating on the findings. "The MraZ protein, in its resting state, forms an octamer, meaning it’s composed of eight identical subunits. These subunits are arranged in a donut-like shape, but with a distinct curvature. This initial conformation would seemingly preclude its direct union with the four ‘boxes’ of the promoter. However, to regulate cell division, we observed how this donut-like structure breaks and deforms in such a way that four of the subunits can precisely join with the four corresponding boxes on the promoter DNA." This conformational change, a remarkable feat of molecular plasticity, allows MraZ to adopt the necessary shape to effectively engage with its DNA target, thereby initiating the transcription of essential cell division genes.
A Paradigm Shift in Understanding Bacterial Growth
The ability to directly visualize, at near-atomic resolution, the precise manner in which MraZ interacts with the promoter DNA that triggers cell division represents a monumental leap forward in the field of bacterial biology. Prior to this study, researchers relied heavily on indirect evidence derived from biochemical experiments and computational modeling to infer the workings of this complex regulatory system. While these methods provided valuable insights, they lacked the direct visual confirmation that structural biology can offer. The UAB team’s work now provides a concrete, tangible understanding of the molecular choreography involved.
Professor Reverter expressed confidence in the broad applicability of their findings. "The regulatory system identified in this study is likely widespread among bacteria," he asserted. "This is because all MraZ proteins are remarkably similar in their structure, maintaining the same octameric donut-like form. Furthermore, the DNA sequences of the promoters of the operons that regulate cell division are also highly conserved across different bacterial species." This suggests that the mechanism unveiled in Mycoplasma genitalium is not an isolated phenomenon but rather a fundamental aspect of bacterial physiology, applicable to a vast array of bacterial organisms.
A Symphony of International Expertise
The ambitious research initiative was meticulously led by David Reverter’s dedicated team at the Institute of Biotechnology and Biomedicine and the Department of Biochemistry and Molecular Biology at UAB. The success of this project was further amplified by crucial collaborations with leading scientific institutions. The ALBA synchrotron, a facility renowned for its advanced light source, played a vital role in providing the necessary infrastructure for certain experimental analyses. Additionally, the cryo-electron microscopy service at the Institute of Genetics and Molecular and Cellular Biology in Strasbourg, France, provided indispensable expertise and resources, showcasing the power of international scientific cooperation in tackling complex biological questions.
Broader Implications: Implications for Health and Biotechnology
The profound implications of this research extend far beyond fundamental biological understanding. Bacteria are ubiquitous, playing critical roles in ecosystems, human health (both beneficial and pathogenic), and industrial processes. Understanding the precise mechanisms that govern their growth and reproduction is paramount.
Antibiotic Development: Many antibiotics target essential bacterial processes, including cell division. By precisely understanding how MraZ regulates this vital function, scientists may be able to identify novel targets for developing new classes of antibiotics. Targeting the MraZ protein itself, or the specific interactions it mediates, could offer a pathway to developing drugs that selectively inhibit bacterial growth without harming human cells. The emergence of antibiotic resistance is a pressing global health crisis, making the discovery of new therapeutic strategies a critical imperative.
Biotechnology and Synthetic Biology: The ability to precisely control bacterial cell division has significant potential in various biotechnological applications. For instance, in industrial fermentation processes, where bacteria are used to produce valuable compounds like biofuels, pharmaceuticals, and enzymes, optimizing cell division can lead to increased yields and efficiency. In synthetic biology, where scientists engineer novel biological systems, a deep understanding of fundamental processes like cell division is crucial for designing and building complex microbial factories.
Understanding Microbial Ecology: Bacteria are the most abundant organisms on Earth, forming complex communities in diverse environments. The ability of these communities to grow and thrive is directly linked to their capacity for cell division. Understanding the conserved mechanisms of cell division, like the one involving MraZ, helps scientists decipher how different bacterial species compete, cooperate, and colonize various niches. This knowledge is vital for fields ranging from environmental science and agriculture to medicine and public health.
The research, initiated approximately three years ago with initial structural investigations and progressing through detailed mechanistic studies, represents a significant investment in understanding a fundamental life process. The publication in Nature Communications signifies peer validation and recognition of the study’s scientific rigor and impact. While specific quantitative data on the efficacy of targeting MraZ in therapeutic contexts are still in early stages of exploration, the foundational knowledge provided by Reverter’s team lays the groundwork for future drug discovery and engineering efforts. The universality of the MraZ mechanism across bacteria suggests that interventions based on this discovery could have broad-spectrum efficacy, a highly desirable trait in the face of widespread antibiotic resistance. The ongoing dialogue with colleagues in the fields of microbiology and pharmacology is expected to accelerate the translation of these fundamental insights into practical applications.












Leave a Reply