Photosynthetic bacteria, the ancient architects of our planet, played an instrumental role in transforming Earth into the vibrant world we inhabit today. Among these primordial life forms, cyanobacteria hold a special place in evolutionary history. It was their pioneering photosynthetic processes that gradually filled our atmosphere with oxygen, a monumental event that paved the way for the emergence of complex, aerobic life. Now, groundbreaking research from the Institute of Science and Technology Austria (ISTA) has unveiled a surprising evolutionary narrative, demonstrating how a biological system once thought to be solely dedicated to DNA segregation has been ingeniously repurposed to control the very shape of cyanobacterial cells. This discovery, published in the esteemed journal Science, offers profound new insights into the dynamic nature of protein systems over geological timescales and sheds light on the developmental pathways that led to multicellularity in these ecologically vital microorganisms.
Pioneers of Oxygen and Life
"Cyanobacteria are essentially pioneers of oxygenic photosynthesis," states Benjamin Springstein, a postdoctoral researcher in the Loose group at ISTA. His sentiment underscores the profound significance of these single-celled organisms. Their photosynthetic activity, dating back billions of years, was not merely an incidental biological process; it was a planetary-scale revolution. The accumulation of oxygen in Earth’s atmosphere, a phenomenon known as the Great Oxygenation Event (GOE) that occurred approximately 2.5 billion years ago, is directly attributed to the relentless activity of cyanobacteria. This dramatic shift in atmospheric composition fundamentally altered Earth’s habitability, creating an environment conducive to the evolution of organisms that rely on oxygen for energy production – a category that includes virtually all complex life forms, including humans. "Without them," Springstein emphasizes, "it’s safe to say that none of us would be here today."
The impact of cyanobacteria extends far beyond their ancient role in atmospheric transformation. Even in the present day, they remain indispensable to the global ecosystem. They contribute a substantial portion of the planet’s biomass and are central players in the crucial carbon and nitrogen cycles, processes fundamental to the sustenance of life. Their remarkable adaptability is evident in their ubiquitous presence across diverse and often extreme environments, from the searing heat of hot springs to the frigid expanse of the Arctic. They can be found colonizing surfaces in urban landscapes, such as rooftops and walls, demonstrating their resilience and versatility.
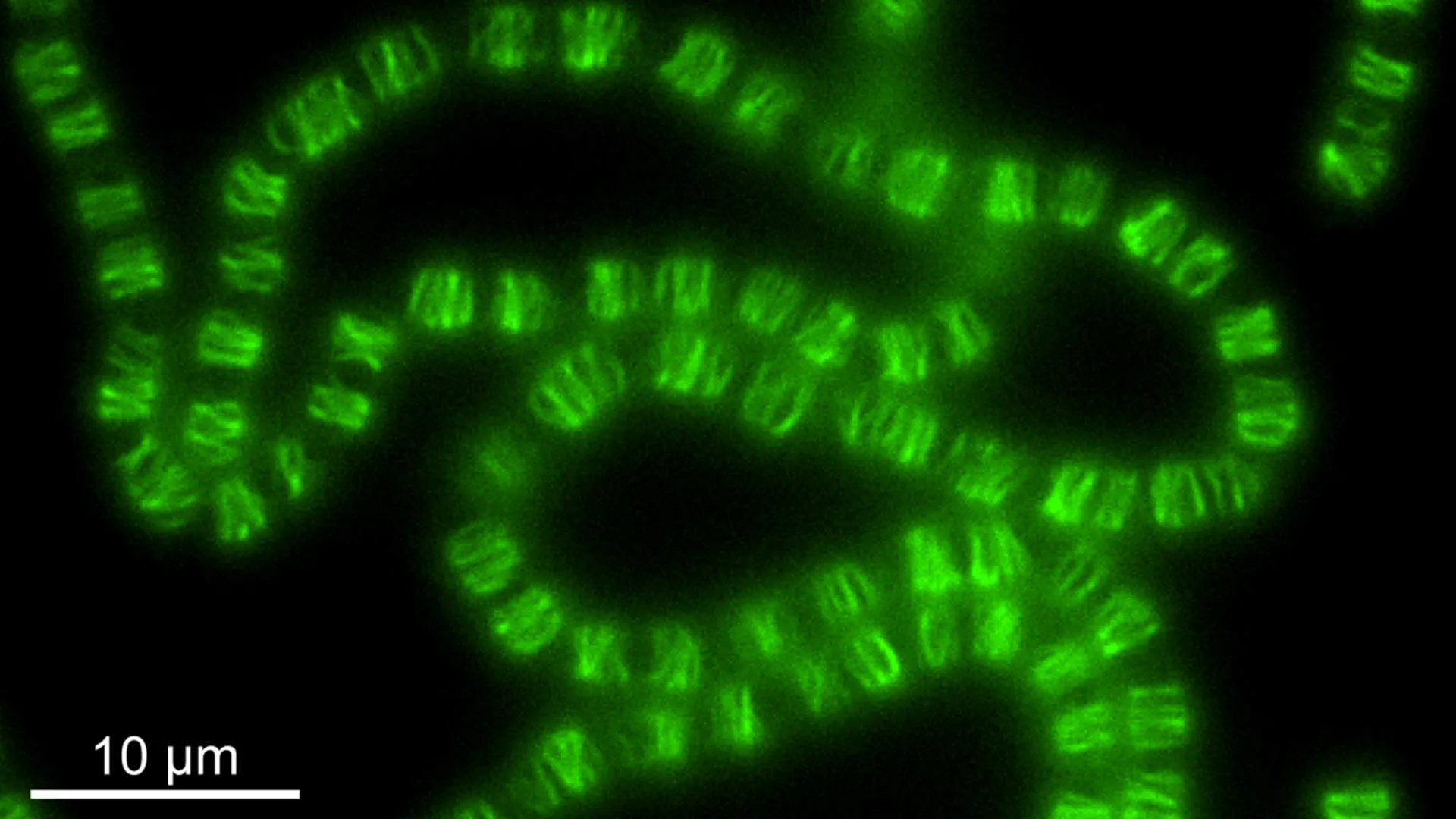
For decades, the species Anabaena sp. PCC 7120, often referred to simply as Anabaena, has served as a crucial model organism for scientific inquiry. Its study has provided invaluable insights into the biology and evolution of multicellular cyanobacteria, a fascinating evolutionary leap for organisms typically thought of as unicellular.
An Evolutionary Masterstroke: Repurposing DNA Machinery for Cell Shaping
The recent findings by Springstein and his collaborators from ISTA, the Institut Pasteur de Montevideo in Uruguay, Kiel University in Germany, and the University of Zurich in Switzerland, reveal a significant evolutionary pivot in Anabaena and, it is hypothesized, in other multicellular cyanobacteria. They have uncovered evidence that an ancient system, originally evolved to ensure the accurate segregation of DNA during cell division, has undergone a remarkable metamorphosis. This system has been repurposed into a sophisticated, cytoskeleton-like structure, now instrumental in dictating and maintaining the characteristic shape of cyanobacterial cells.
Understanding Bacterial DNA: A Foundation for the Discovery
To fully appreciate the significance of this discovery, it’s essential to understand the basics of DNA within bacteria. Like all prokaryotic organisms, Anabaena reproduces through binary fission, a process where a single cell divides into two identical daughter cells. For this to occur successfully, the genetic material – the DNA – must be meticulously replicated and then accurately distributed to each new cell. Bacterial DNA is organized into chromosomes, which are analogous to tightly wound spools of thread, containing the essential genetic blueprint for survival. In many bacteria, including Anabaena, chromosomes are not the only form of genetic material; they also possess plasmids.
Plasmids are smaller, circular DNA molecules that are distinct from the main chromosome. They often carry genes that confer advantageous traits, such as antibiotic resistance or metabolic capabilities, which are not strictly essential for basic survival but can enhance adaptability. A key characteristic of plasmids is their ability to be transferred between bacteria, a process known as horizontal gene transfer. This mechanism allows for the rapid dissemination of beneficial traits throughout a bacterial population, enabling swift adaptation to changing environmental conditions.
A DNA Segregation System Finds a New Calling
Benjamin Springstein’s engagement with Anabaena began in 2014, where he delved into its intricate biology and evolutionary history. The serendipitous observation that led to this groundbreaking discovery occurred during the COVID-19 pandemic, a period of enforced pause in laboratory work that prompted extensive review of existing scientific literature.
"I made a serendipitous observation," Springstein recounts, highlighting the unexpected nature of his find. He noticed that Anabaena and certain related cyanobacteria possessed a system known as ParMR, encoded within their chromosomal DNA. Traditionally, the ParMR system was understood to be exclusively associated with plasmid segregation and had only ever been identified on plasmids themselves. Its presence within the chromosome of Anabaena was highly unusual and sparked Springstein’s suspicion that this system might have adapted to a different role, potentially involving chromosome segregation.
Upon joining ISTA as an IST-Bridge Fellow, Springstein embarked on a series of experiments to rigorously test his hypothesis. The results, however, unveiled a scenario far removed from his initial expectations. It became apparent that one key component of the system, ParR, no longer exhibited binding to DNA. Instead, its affinity shifted to lipid membranes, particularly the inner cell membrane. Concurrently, the other component, ParM, did not assemble into structures within the cytoplasm to facilitate DNA movement. Rather, it formed extensive filament networks situated just beneath the inner membrane. These networks, composed of protein polymers, created a layer that bore a striking resemblance to a cell cortex, a structural component found in more complex eukaryotic cells.
Instead of functioning as a typical DNA segregation apparatus, which would typically form spindle-like structures within the cell’s interior to move chromosomes, this system operated at the cellular membrane level. Its primary function appeared to be the organization and maintenance of cellular structure.
Filaments That Mimic a Cellular Skeleton
To gain a deeper understanding of this novel function, the research team employed a combination of in vitro reconstitution experiments and advanced imaging techniques. In these experiments, they recreated the ParMR system outside of living cells using purified protein components. The reconstituted filaments displayed a behavior known as dynamic instability, characterized by periods of growth followed by rapid collapse. This dynamic behavior is remarkably similar to that observed in microtubules, a key component of the cytoskeleton in more complex eukaryotic cells.
Further investigation into the structural architecture of these filaments was conducted in collaboration with ISTA Professor Florian Schur and his PhD student Manjunath Javoor. Utilizing cryo-electron microscopy (cryo-EM), a technique that allows for atomic-level visualization of molecular structures, they meticulously examined how these protein filaments are assembled. Their detailed analysis revealed a significant distinction: unlike the polar filaments formed by analogous systems in other bacteria, where growth and shrinkage occur predominantly at one end, the filaments in Anabaena were found to be bipolar. This means they possess the capacity to grow and retract from both ends simultaneously, a feature that likely contributes to their robust structural role.
Loss of Function Reveals Critical Role in Cell Shape
The definitive proof of the system’s true function emerged when it was experimentally removed from living Anabaena cells. The consequences were stark and revealing.
"Cells lacking the system lost their normal rectangular-like cell shape and instead became round and swollen," Springstein explains. This dramatic alteration in cellular morphology is highly significant. In other bacterial species, changes of this nature are typically observed when genes directly responsible for maintaining cell shape are disrupted. This strongly indicated that the system’s primary role was not DNA distribution but rather the active control and maintenance of cellular structure.
In light of its altered function and location within the cell, the researchers proposed a new designation for this repurposed system: "CorMR," short for "cortex-related protein system."
The Evolutionary Trajectory: A Masterclass in Biological Innovation
The evolution of multicellularity in cyanobacteria was a gradual process, unfolding over vast stretches of time from single-celled ancestors. To trace the evolutionary path of the CorMR system, the team collaborated with Daniela Megrian, a researcher at the Institut Pasteur in Montevideo, Uruguay. Megrian conducted extensive bioinformatic analyses, which provided crucial insights into how this ancient DNA segregation mechanism was transformed into a cell-shaping apparatus.
The transformation was not a singular event but rather a step-by-step evolutionary cascade. The initial shift involved the relocation of the ParMR system from plasmids to the bacterial chromosome. Subsequently, the protein components themselves underwent modifications in size and structure, adapting to their new context. A critical juncture was the development of the ability for these components to bind to the cell membrane. Finally, the system appears to have come under the regulatory control of an additional protein system, further refining its function.
This intricate sequence of evolutionary changes illustrates a powerful principle: the repurposing of existing biological machinery. The CorMR system serves as a compelling example of how evolution can take an ancient tool, originally designed for one purpose, and endow it with entirely new and critical functions, profoundly impacting the organism’s morphology and, in this case, contributing to the evolutionary success of multicellular life.
Broader Implications for Understanding Life’s Development
The discovery of the CorMR system in cyanobacteria has far-reaching implications for our understanding of fundamental biological processes. It provides a tangible example of how evolutionary innovation can occur through the adaptation and repurposing of existing genetic and protein systems. This principle is not unique to cyanobacteria; it is a fundamental driver of evolutionary change across all domains of life.
The findings also shed light on the early evolution of multicellularity. By understanding how single-celled organisms developed internal structures and mechanisms to coordinate and maintain cellular form, we gain deeper insights into the developmental pathways that eventually led to the complex multicellular organisms we see today. The resemblance of the CorMR system’s filaments to the cytoskeleton of eukaryotic cells suggests a potential evolutionary convergence or an ancient shared ancestry for such structural components.
Furthermore, the research highlights the ecological importance of cyanobacteria. Their ability to adapt and evolve, as exemplified by the CorMR system, has allowed them to thrive in diverse environments and continue to play a vital role in global biogeochemical cycles. Future research may explore whether similar repurposing of DNA segregation systems has occurred in other bacterial lineages, potentially revealing broader patterns of evolutionary adaptation. The ongoing study of these ancient organisms continues to unlock secrets about the very origins of life and the intricate mechanisms that have shaped our planet’s biosphere.












Leave a Reply