In a landmark achievement for regenerative medicine and hematology, researchers at the University of Basel and University Hospital Basel have successfully engineered a functional, three-dimensional model of human bone marrow using exclusively human cells. This breakthrough, recently detailed in the journal Cell Stem Cell, represents the first time scientists have been able to recreate the intricate "blood factory" of the human body in a laboratory setting without relying on animal-derived components or simplified, non-representative cell cultures. The achievement marks a significant shift in how scientists study blood formation, test new pharmaceuticals, and approach the treatment of complex blood-related pathologies, including leukemia and multiple myeloma.
The bone marrow is an exceptionally complex organ, often described as the body’s primary hematopoietic site. It is a highly specialized tissue network comprising bone cells, intricate nerve endings, a dense web of blood vessels, and a variety of immune cells. For decades, this environment has remained one of the most difficult biological systems to replicate in vitro due to its structural density and the precise chemical signaling required to maintain blood cell production. By successfully mirroring this complexity, the Basel-based team has provided a new lens through which the mechanics of human biology can be observed with unprecedented clarity.
The Architecture of the Human Blood Factory
Bone marrow operates as a silent but vital engine, producing billions of new blood cells every day. It remains largely invisible to the average person until its function is compromised. In cases of blood cancer or bone marrow failure, the production of red blood cells, white blood cells, and platelets is disrupted, leading to life-threatening complications. Understanding why this process breaks down requires a deep dive into the "niches"—the specific microenvironments within the marrow that regulate cell behavior.
The research team, led by Professor Ivan Martin and Dr. Andrés García García from the Department of Biomedicine, focused their efforts on the "endosteal niche." This specific region is located near the inner surface of the bone and is of particular interest to oncologists. It is widely recognized as a sanctuary for cancer cells; in many forms of leukemia, malignant cells retreat to the endosteal niche to escape the effects of chemotherapy, leading to eventual relapse. Until now, the lack of a human-specific model that included all components of this niche—bone cells, vessels, and nerves—forced researchers to rely on mouse models, which frequently fail to replicate human-specific drug responses or disease progressions.
The Technical Blueprint: Hydroxyapatite and Pluripotent Stem Cells
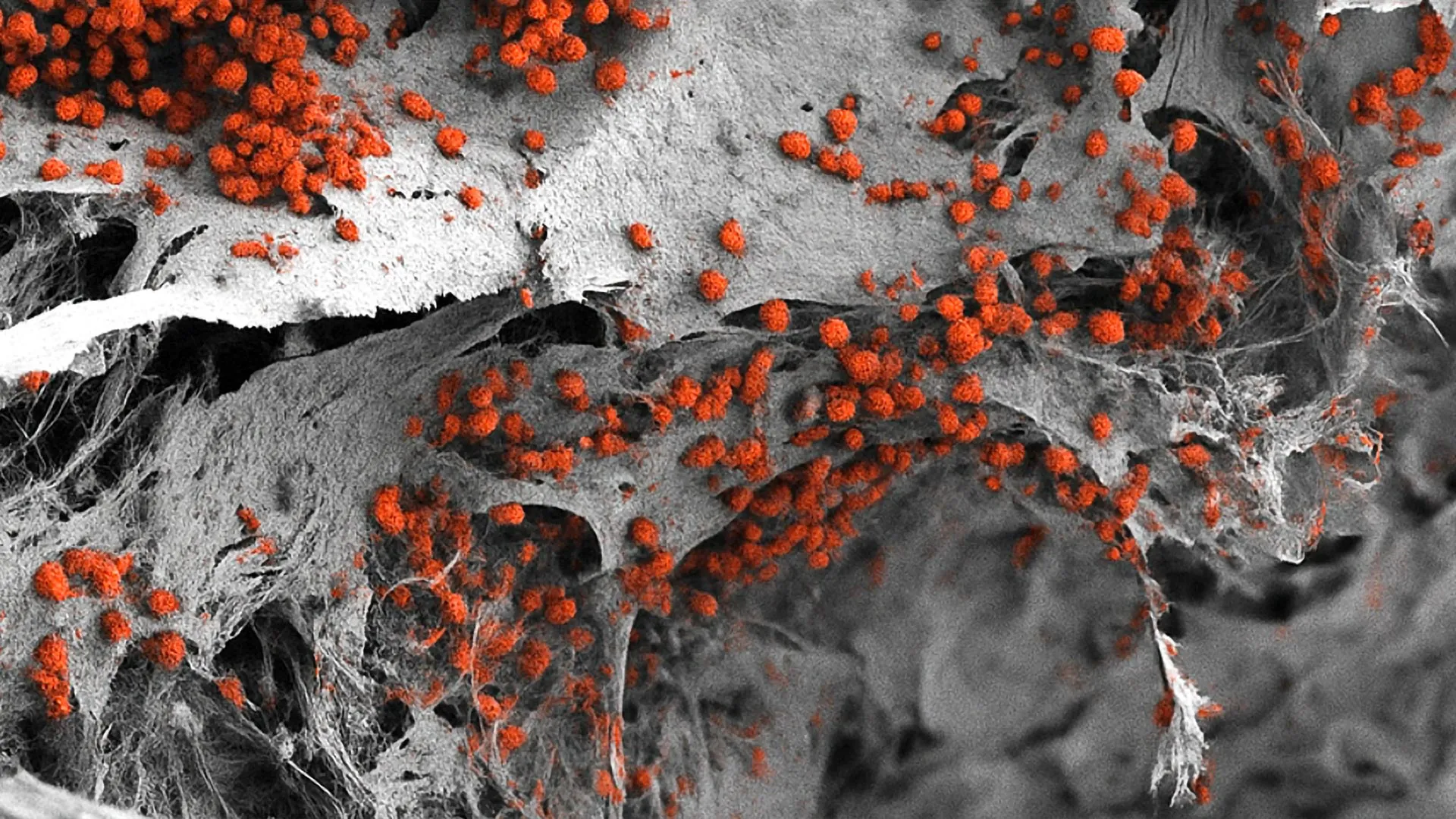
The construction of this model was a multi-stage engineering feat that combined materials science with advanced stem cell biology. The process began with the creation of an artificial scaffold made of hydroxyapatite. Hydroxyapatite is a naturally occurring mineral form of calcium apatite that constitutes the primary inorganic component of human teeth and bones. By using this material, the researchers provided a biocompatible framework that mimicked the physical and chemical properties of the skeletal environment.
Into this mineral scaffold, the team introduced human induced pluripotent stem cells (iPSCs). These are adult cells—often skin or blood cells—that have been molecularly reprogrammed back into an embryonic-like state. Because iPSCs are "pluripotent," they possess the unique ability to differentiate into any cell type in the human body when provided with the correct biological cues.
The researchers meticulously guided these stem cells through a series of developmental stages. By manipulating the signaling molecules and environmental conditions within the hydroxyapatite framework, they encouraged the stem cells to evolve into the diverse array of cells found in the marrow, including osteoblasts (bone-forming cells), endothelial cells (which form blood vessels), and various stromal and neural components. The result was a living, 3D structure that reached dimensions of eight millimeters in diameter and four millimeters in thickness—a substantial size for a lab-grown tissue model.
Supporting Data and Functional Success
The success of the model was measured by its ability to perform the core function of bone marrow: hematopoiesis. The analysis conducted by the Basel team confirmed that the lab-grown endosteal niche was not merely a structural replica but a functional one. For a period of several weeks, the system successfully maintained the formation and maturation of human blood cells.
Quantitative analysis showed that the cellular composition of the model closely mirrored the ratios found in natural human bone marrow. Furthermore, the researchers observed that the various cell types within the model were interacting as they would in the body, with the vascular network providing necessary nutrients and the bone cells providing the structural and chemical signals required for stem cell maintenance. This longevity and functional stability are critical for long-term studies of disease progression and the effects of chronic drug exposure.
Bridging the Gap Between Animal Models and Human Biology
For nearly a century, animal models—particularly mice—have been the gold standard for medical research. While these models have yielded invaluable insights into the basic mechanics of biology, they possess significant limitations when applied to human pathology. Mice and humans differ in their immune responses, metabolic rates, and the specific genetic markers of blood cancers.
"We have learned a great deal about how bone marrow works from mouse studies," noted Professor Ivan Martin in a statement following the publication. "However, our model brings us closer to the biology of the human organism."
The development of this all-human model aligns with the global scientific movement toward the "3Rs": the Replacement, Reduction, and Refinement of animal experiments. By providing a more accurate human-centric platform, the University of Basel aims to decrease the number of animals required for early-stage research and drug discovery. In many cases, drugs that appear successful in mice fail during human clinical trials because the animal’s biology does not perfectly match the human response. A human-cell-based model could identify these failures much earlier in the development process, saving time and resources while improving patient safety.
Implications for Hematological Oncology and Drug Resistance
One of the most promising applications of this technology is in the field of oncology. Blood cancers such as leukemia, lymphoma, and multiple myeloma are notoriously difficult to treat because of the protective environment the bone marrow provides to malignant cells.
The endosteal niche model allows researchers to observe, in real-time, how cancer cells interact with bone and vascular tissue. By introducing cancer cells into the lab-grown marrow, scientists can study the mechanisms of "minimal residual disease"—the small number of cancer cells that survive treatment and later cause the disease to return. This could lead to the discovery of new drugs that specifically target the "hiding places" of cancer cells, making traditional treatments like chemotherapy or radiation more effective.
Furthermore, the model serves as a sophisticated platform for drug testing. Pharmaceutical companies can use these 3D structures to screen thousands of potential compounds for toxicity and efficacy before moving to human trials. Dr. Andrés García García pointed out, however, that the current size of the model—while impressive for biological accuracy—presents a challenge for high-throughput screening. "For this specific purpose, the size of our bone marrow model might be too large," he explained. The next phase of research will likely involve miniaturizing the system to allow for the simultaneous testing of numerous drug doses and combinations.
Toward Personalized Medicine: The Future of Patient-Specific Care
Looking beyond general research, the Basel team envisions a future where this technology is used for personalized medicine. In this scenario, a patient diagnosed with a blood cancer could have their own cells used to create a "personalized bone marrow model" in the lab. Doctors could then test various treatment regimens on the patient’s own lab-grown tissue to see which specific drug or combination of therapies is most effective at eradicating the cancer cells while sparing healthy tissue.
This approach would move oncology away from a "one-size-fits-all" methodology and toward a tailored strategy that accounts for the unique genetic and cellular profile of each individual’s disease. While the researchers acknowledge that further refinements are necessary to make this process fast and cost-effective enough for clinical use, the current study provides the essential proof-of-concept.
Broader Impact and Scientific Reactions
The scientific community has reacted with cautious optimism to the Basel study. Experts in bioengineering have praised the team for their ability to integrate neural and vascular components into a mineralized scaffold, a feat that has eluded researchers for years. The inclusion of these elements is vital, as the nervous system plays a documented role in regulating the release of blood cells into the circulation.
The implications of this research extend beyond cancer. The model could also be used to study rare genetic blood disorders, the effects of aging on the immune system, and the impact of environmental toxins on blood production. By providing a controlled, observable environment that mimics the human body, the University of Basel has opened a new chapter in hematological research.
As the project moves forward, the researchers plan to integrate even more components into the system, such as a more complex immune cell presence and a circulating "blood" flow to better simulate the systemic environment. The ultimate goal is to create a "marrow-on-a-chip" device that can be easily integrated into existing laboratory workflows worldwide.
In summary, the creation of a fully human, 3D bone marrow model is a significant milestone in biotechnology. It offers a more ethical, accurate, and potentially life-saving alternative to traditional research methods. By bridging the gap between the laboratory bench and the patient’s bedside, Professor Martin, Dr. García García, and their team have set the stage for a new era of medical discovery where the complexities of the human "blood factory" are no longer a mystery, but a tool for healing.














Leave a Reply