Researchers from the Department of Medicine at the School of Clinical Medicine, LKS Faculty of Medicine, University of Hong Kong (HKUMed) have reached a significant milestone in musculoskeletal biology by identifying a specific protein that functions as the body’s internal "exercise sensor." This discovery provides a long-sought-after molecular explanation for how physical movement translates into skeletal strength and offers a potential pharmacological roadmap for treating osteoporosis. The study, published in the prestigious international journal Signal Transduction and Targeted Therapy, highlights the role of a protein called Piezo1 in regulating the balance between bone formation and fat accumulation within the bone marrow. This breakthrough is particularly significant for aging populations and individuals who are physically unable to engage in weight-bearing exercise, as it suggests that the benefits of physical activity could eventually be delivered through targeted medication.
The Global Burden of Osteoporosis and Bone Fragility
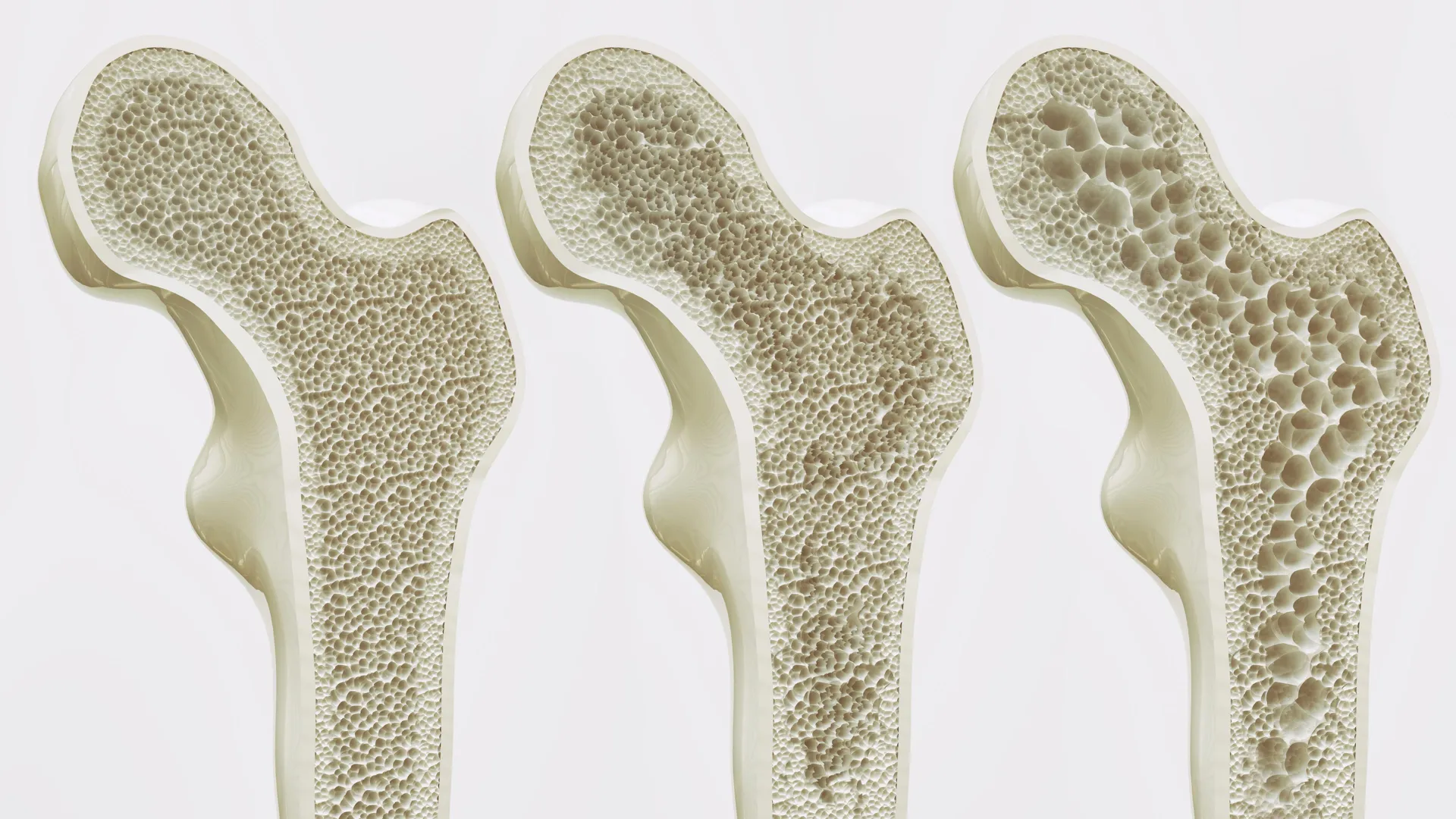
Osteoporosis is a systemic skeletal disorder characterized by low bone mass and micro-architectural deterioration of bone tissue, leading to increased bone fragility and a consequent increase in fracture risk. It is often referred to as a "silent disease" because bone loss occurs without symptoms until a sudden strain, bump, or fall causes a fracture. According to data from the World Health Organization (WHO), osteoporosis affects hundreds of millions of people worldwide. Statistics indicate that approximately one in three women and one in five men over the age of 50 will suffer an osteoporotic fracture in their remaining lifetime.
In Hong Kong, the public health challenge is particularly acute due to one of the fastest-aging populations in the world. Local data suggests that osteoporosis affects approximately 45% of women and 13% of men aged 65 and older. These fractures—most commonly of the hip, spine, and wrist—are associated with high morbidity and mortality. Hip fractures, in particular, are devastating; they often result in a permanent loss of independence, with many patients requiring long-term nursing home care. Furthermore, the mortality rate in the year following a hip fracture can be as high as 20% to 24%. The economic strain on the Hong Kong healthcare system is substantial, involving not only acute surgical costs but also long-term rehabilitation and social services.
The Biological Mechanism of Bone Loss and Aging
To understand the HKUMed discovery, one must first look at the cellular environment within the bone marrow. The marrow contains mesenchymal stem cells (MSCs), which are multipotent cells capable of differentiating into various cell types, including osteoblasts (cells that build bone) and adipocytes (cells that store fat). In a healthy, active individual, there is a delicate equilibrium between these two pathways. Physical activity creates mechanical stress and pressure on the skeleton, which signals these stem cells to prioritize bone formation.
However, as the body ages, this biological "switch" begins to malfunction. The balance shifts, and mesenchymal stem cells increasingly differentiate into fat cells rather than bone-forming cells. This process leads to what researchers call "fatty marrow." As fat accumulates inside the bone cavity, it displaces healthy bone tissue and creates a pro-inflammatory environment that further accelerates bone resorption. For patients who are bedridden or have limited mobility due to chronic illness or injury, this lack of mechanical stimulation causes rapid bone loss, creating a cycle of deterioration that current medications—which primarily focus on slowing bone breakdown rather than stimulating formation via mechanical pathways—struggle to fully reverse.
Identifying Piezo1: The Molecular Switch for Movement
The research team, led by Professor Xu Aimin, Director of the State Key Laboratory of Pharmaceutical Biotechnology and Chair Professor in the Department of Medicine at HKUMed, utilized mouse models and human stem cell cultures to pinpoint the exact mechanism that detects physical force. They identified Piezo1, a mechanically activated ion channel protein located on the surface of mesenchymal stem cells.
Piezo1 acts as a gatekeeper for mechanical signals. When a person moves, runs, or lifts weights, the physical pressure deforms the cell membrane, causing the Piezo1 channel to open. This allows ions to flow into the cell, triggering a cascade of biochemical signals that promote the expression of bone-building genes. The HKUMed study demonstrated that Piezo1 is essential for this process. In experimental models where Piezo1 was removed or inactivated, the bones became significantly weaker and more porous, even when the subjects were active.
Conversely, the study found that when Piezo1 is functioning correctly, it actively suppresses the formation of fat cells. It does this by inhibiting specific inflammatory signals, namely Ccl2 and lipocalin-2 (Lcn2). When Piezo1 is absent or inactive, these inflammatory markers surge, pushing the stem cells toward fat production and stalling bone growth. By blocking these inflammatory signals, the researchers were able to partially restore bone health, confirming that Piezo1 is the primary regulator of this bone-fat balance.
The Concept of Exercise Mimetics: Medicine for the Immobile
The most revolutionary implication of this research is the development of "exercise mimetics." These are a new class of drugs designed to replicate the physiological effects of physical exercise at the molecular level. While exercise remains the "gold standard" for maintaining health, it is a therapy that many of the most vulnerable patients cannot access.
"Current treatments rely heavily on physical activity, which many patients simply cannot perform," explained Professor Xu Aimin. "We need to understand how our bones get stronger when we move or exercise before we can find a way to replicate the benefits of exercise at the molecular level. This study is a critical step towards that goal."
By targeting the Piezo1 pathway, scientists hope to develop medications that "trick" the mesenchymal stem cells into thinking the body is undergoing physical exertion. This would stimulate bone formation and prevent marrow fat accumulation even in a sedentary state. Such a breakthrough would be life-changing for several patient groups:
- The Frail Elderly: Those who are too weak to perform weight-bearing exercises safely.
- Bedridden Patients: Individuals recovering from major surgery, strokes, or spinal cord injuries who experience rapid bone density loss during immobilization.
- Chronic Disease Sufferers: Those with conditions like severe arthritis or cardiovascular disease that limit physical activity.
- Astronauts: Bone loss due to a lack of mechanical loading is a major hurdle for long-duration space flight; Piezo1-based therapies could potentially mitigate this risk.
Collaborative Research and Institutional Support
The study was a high-level international collaboration, reflecting the global importance of the findings. Alongside Professor Xu Aimin, the project was co-led by Dr. Wang Baile, Research Assistant Professor at HKUMed, and Professor Eric Honoré from the Institute of Molecular and Cellular Pharmacology at the French National Centre for Scientific Research (CNRS).
Professor Honoré emphasized that this research moves beyond traditional physical therapy. "This offers a promising strategy beyond traditional physical therapy," Honoré stated. "In the future, we could potentially provide the biological benefits of exercise through targeted treatments, thereby slowing bone loss in vulnerable groups such as the bedridden patients or those with limited mobility, and substantially reducing their risk of fractures."
The research was supported by a wide array of prestigious funding bodies, including the Research Grants Council of Hong Kong, the Health Bureau of the HKSAR Government, the National Key R&D Program of China, and the National Natural Science Foundation of China. International support came from the Human Frontier Science Program and several French medical foundations, underscoring the scientific community’s consensus on the urgency of finding new solutions for age-related bone decay.
Analysis of Future Implications and Clinical Translation
While the identification of Piezo1 is a major scientific victory, the path to a widely available "exercise pill" for bone health involves several more steps. The HKUMed team is currently focused on translating these laboratory findings into clinical applications. This involves screening for chemical compounds that can safely and effectively activate the Piezo1 pathway in humans without causing adverse side effects in other tissues where Piezo proteins might be present.
From a public health perspective, the success of such a treatment could fundamentally alter the trajectory of geriatric care. Currently, the management of osteoporosis is largely reactive—treating the bone density after it has already dropped or repairing a fracture after it has occurred. A Piezo1-based therapy could represent a shift toward proactive, regenerative medicine. By maintaining the "youthful" signaling of the bone marrow, healthcare providers could prevent the skeletal decay that often precedes the loss of independence in the elderly.
Furthermore, this research contributes to the growing field of mechanobiology, which explores how physical forces influence cell behavior. The discovery that a single protein can dictate the fate of a stem cell—choosing between bone and fat—suggests that similar mechanisms may exist in other tissues, such as muscle or cartilage. This opens the door for broader research into how "exercise sensors" might be used to treat sarcopenia (muscle loss) or osteoarthritis.
Conclusion
The HKUMed discovery of the Piezo1 protein’s role as an exercise sensor marks a turning point in the fight against osteoporosis. By decoding the molecular language of movement, the research team has provided a blueprint for future therapies that could protect the most vulnerable members of society from the debilitating effects of bone loss. As the world continues to grapple with the challenges of an aging population, the ability to chemically mimic the benefits of exercise stands as one of the most promising frontiers in modern medical science. The focus now shifts to clinical development, with the hope that the "biological benefits of exercise" will one day be accessible to all, regardless of their physical mobility.














Leave a Reply