In a landmark announcement on April 1, the U.S. Food and Drug Administration (FDA) granted approval to Foundayo (orforglipron), Eli Lilly’s pioneering once-daily oral GLP-1 receptor agonist designed for the management of obesity. This approval marks a significant milestone as Foundayo becomes the first non-peptide oral GLP-1 specifically indicated for weight management, and notably, represents the fastest new molecular entity approval since 2002. The drug was fast-tracked under the FDA’s Commissioner’s National Priority Voucher program, receiving clearance just 50 days after its filing, a remarkable nearly ten months ahead of its anticipated review date. This expedited approval underscores the critical unmet need in obesity treatment and the innovative nature of Foundayo’s therapeutic profile.
The timing of this regulatory triumph is particularly poignant for Eli Lilly, as the pharmaceutical giant has concurrently ascended to an unprecedented position, surpassing all other global pharmaceutical companies in terms of revenue. Despite this dual achievement – a pivotal drug approval and a top-tier financial performance – the company’s stock has experienced a notable decline of nearly 14% since January 1, presenting a perplexing paradox to industry observers and investors alike. This dichotomy between operational success and market skepticism highlights the complex dynamics currently at play within the biopharmaceutical sector, especially concerning highly anticipated, high-value therapeutic areas like metabolic diseases.
Eli Lilly’s Ascendance to Industry Leader
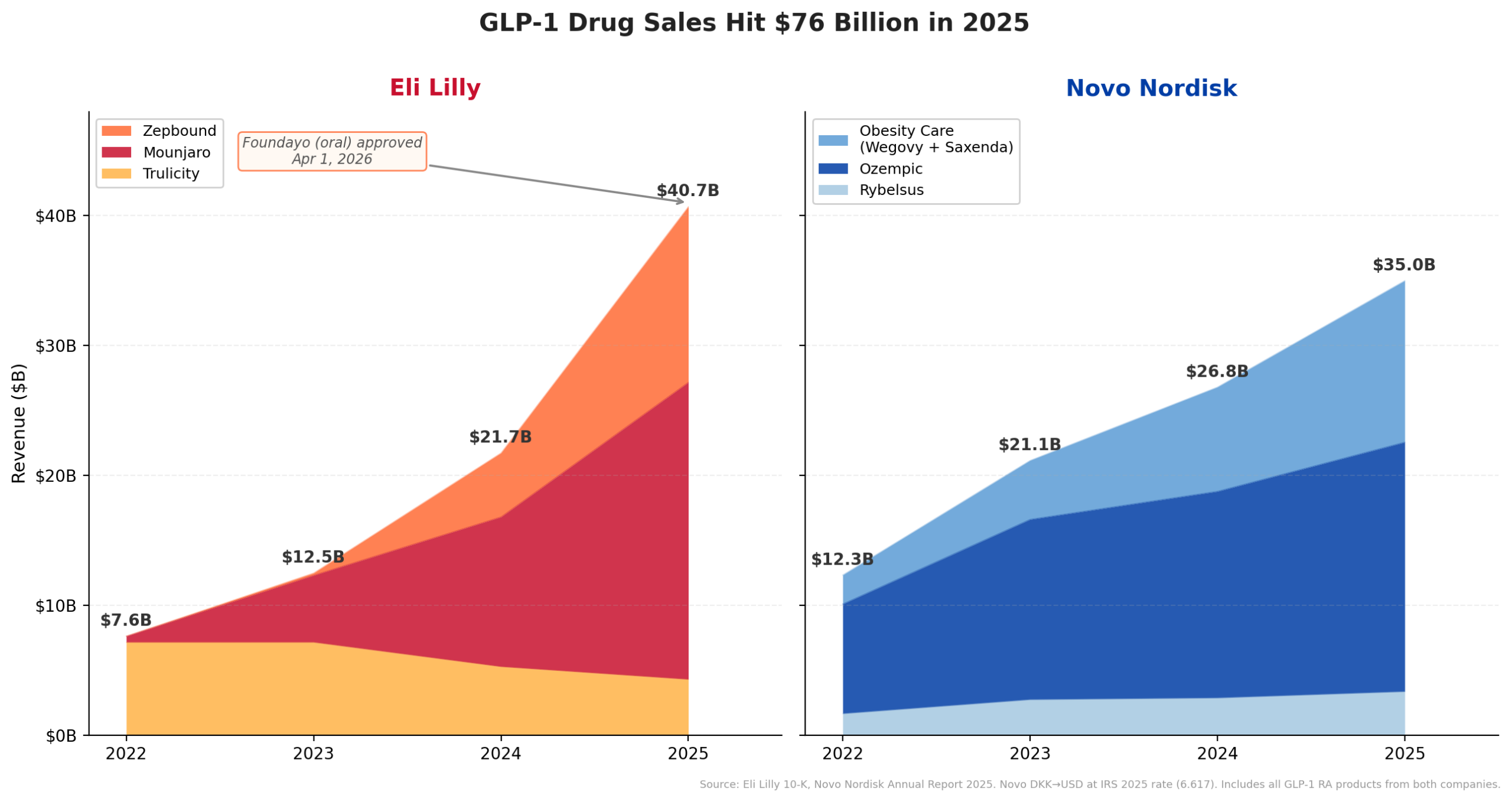
Eli Lilly’s financial performance in fiscal year 2025 cemented its new status as the pharmaceutical industry’s revenue leader. The company reported an impressive $65.18 billion in revenue, reflecting a substantial 44.7% year-over-year growth. This growth rate not only stands as the highest among the top 20 pharmaceutical companies globally but also propelled Lilly past long-standing titans such as Merck & Co. ($65.01B), Pfizer ($62.58B), and AbbVie ($61.16B) to claim the coveted number one spot for the first time in its history. While the margin over Merck was a narrow $170 million, the disparity in growth trajectories was profound: Lilly’s robust expansion dwarfed Merck’s modest 1.3% growth. This remarkable surge is a testament to Lilly’s strategic focus and successful execution in bringing transformative therapies to market.
The primary catalyst for this extraordinary financial surge has been Eli Lilly’s formidable portfolio of injectable GLP-1 receptor agonists. Mounjaro (tirzepatide), approved for type 2 diabetes and increasingly used off-label for weight loss, generated an astounding $9.0 billion in the first half of 2025 alone, marking a 90% increase year-over-year. Complementing this, Zepbound (tirzepatide), the specific obesity-indication version of the drug, contributed an additional $5.7 billion in H1 2025. Zepbound’s rapid market penetration is particularly noteworthy given its launch only in late 2023, now further enhanced by its availability in a multi-dose KwikPen, simplifying patient administration. Collectively, Lilly’s GLP-1 franchise is on track to exceed an annualized revenue of $30 billion, underscoring its unprecedented commercial success and profound impact on the company’s financial profile.
The Strategic Significance of Foundayo: A New Era for Oral GLP-1s
The approval of Foundayo heralds a significant shift in the landscape of GLP-1 therapies, particularly for obesity management. Until recently, the market for blockbuster GLP-1 drugs, including Novo Nordisk’s Ozempic and Wegovy, and Lilly’s Mounjaro and Zepbound, was dominated by injectable formulations. While Novo Nordisk did introduce an oral version of semaglutide, Rybelsus, for type 2 diabetes and later an oral Wegovy for obesity in December 2025, these are peptide-based formulations that carry specific administration requirements, such as taking them on an empty stomach with restricted water intake.
Foundayo distinguishes itself as a small molecule GLP-1 receptor agonist. This chemical characteristic offers a substantial advantage: it can be taken at any time of day, with or without food or water restrictions, significantly enhancing convenience and potentially improving patient adherence. This ease of use addresses a critical barrier for many patients who are hesitant or unable to manage injectable medications. Eli Lilly CEO Dave Ricks emphasized this point, noting that while Foundayo may "not be more effective" than the company’s injectable Zepbound in terms of raw weight loss percentages, "it’s more accessible, it’s easier to fit into your daily routine." Clinical trials, specifically the ATTAIN-1 program, demonstrated that patients on the highest dose of Foundayo achieved an average weight loss of 12.4% over 72 weeks. While this is less than the 20%-plus weight loss typically observed with injectable tirzepatide, Ricks positioned Foundayo as a complementary, rather than competitive, offering within Lilly’s expanding portfolio. The strategic goal, as articulated by Ricks, is to ensure patients have access to the medication that best aligns with their health goals, regardless of formulation, as long as "it has Lilly on the box."
Expanding the Addressable Market and Global Implications
The introduction of an oral GLP-1 option for obesity has profound implications for market expansion. The GLP-1 market has witnessed explosive growth, skyrocketing from under $12 billion in 2022 to an estimated annualized run rate of approximately $65 billion by mid-2025. An oral, convenient option like Foundayo is expected to significantly broaden the addressable patient population, reaching individuals who prefer pills over injections, or who might have previously been hesitant to start GLP-1 therapy due to the injectable format. This includes a vast segment of the population living with obesity who might not yet be candidates for or comfortable with more aggressive injectable treatments.
Lilly’s ambition for Foundayo extends beyond the U.S. borders, with the company having already submitted orforglipron for approval in over 40 countries worldwide. This global strategy underscores the perceived universal demand for a convenient and effective oral obesity treatment. The foundational work for orforglipron began in 2018 when Lilly licensed the compound from Japanese drugmaker Chugai in a deal valued at $50 million upfront, a strategic investment that has clearly paid significant dividends.
The Paradox of Wall Street’s Reaction
Despite these monumental achievements, Wall Street’s reaction, characterized by a nearly 14% decline in Lilly’s stock year-to-date, presents a puzzle. Several factors could contribute to this apparent disconnect between corporate success and investor sentiment.
One potential explanation is that the market may have already "priced in" much of the anticipated success of Lilly’s GLP-1 pipeline, including the approval of an oral variant. Investor expectations for Lilly have been exceptionally high, driven by the unprecedented demand for Mounjaro and Zepbound. When highly anticipated news, even positive news, aligns with or slightly underperforms these elevated expectations, a "sell the news" phenomenon can occur.
Another factor could be concerns surrounding the competitive landscape. While Lilly now has an oral GLP-1, it faces fierce competition from Novo Nordisk, which has its own oral semaglutide (Rybelsus and oral Wegovy). The market may be scrutinizing the long-term competitive dynamics, particularly regarding efficacy, pricing, and market share capture in a rapidly evolving and highly competitive space. Furthermore, the 12.4% weight loss seen with Foundayo, while clinically significant, is lower than the 20%+ achieved with Lilly’s own injectable tirzepatide. Investors might be weighing the potential cannibalization of their higher-efficacy, higher-priced injectable products or projecting a lower revenue ceiling for Foundayo compared to its injectable counterparts.
Manufacturing and supply chain considerations also loom large. The explosive demand for GLP-1s has strained manufacturing capabilities across the industry, leading to intermittent shortages. While Foundayo, as a small molecule, might be easier to produce at scale than peptide-based injectables, the sheer volume required to meet global demand for an oral obesity drug could still present significant logistical challenges. Investors might be factoring in the potential for slower-than-expected ramp-up in production or future supply constraints.
Finally, broader market trends and valuation concerns could be at play. Eli Lilly’s stock has seen a meteoric rise over the past few years, leading to a high valuation. Some investors might be taking profits or re-evaluating whether the current valuation fully accounts for future risks, including increased R&D costs, potential future competition, or changes in healthcare policy regarding obesity treatments. The overall economic climate and interest rate environment can also influence investor appetite for high-growth, high-valuation stocks.
Broader Impact and Future Outlook
Eli Lilly’s dual achievement of becoming the top-revenue pharma company and securing approval for Foundayo carries significant implications for the pharmaceutical industry, public health, and the treatment of obesity.
For the pharmaceutical industry, Lilly’s rise to the top signals a shift in market leadership, emphasizing the strategic importance of innovation in high-demand therapeutic areas. It underscores the immense commercial potential of effective treatments for chronic conditions like obesity and diabetes, which affect hundreds of millions globally. This success will undoubtedly spur increased investment and R&D efforts across the industry into metabolic disorders, potentially leading to a new wave of therapeutic advancements.
From a public health perspective, the availability of an accessible oral GLP-1 like Foundayo is a game-changer. Obesity is a complex, chronic disease associated with numerous comorbidities, including type 2 diabetes, heart disease, certain cancers, and musculoskeletal issues. By offering a convenient oral option, Lilly is poised to significantly increase access to effective weight management, potentially leading to improved health outcomes for a much larger patient population. The ease of administration could also improve adherence rates, which is a critical factor in the long-term success of chronic disease management. Patient advocacy groups are likely to welcome this development, as it removes a significant barrier for many individuals seeking treatment.
The competitive dynamics between Eli Lilly and Novo Nordisk are set to intensify further. Both companies are at the forefront of the GLP-1 revolution, and their strategic moves in terms of formulations, indications, and global reach will define the future of this multi-billion-dollar market. Lilly’s "complementary" strategy of offering both highly efficacious injectables and convenient orals aims to capture the broadest possible patient demographic, suggesting a nuanced approach to market segmentation.
In conclusion, while the immediate reaction from Wall Street might appear counterintuitive, Eli Lilly’s recent achievements mark a transformative period for the company and the pharmaceutical industry at large. The approval of Foundayo, coupled with its unprecedented revenue growth driven by its injectable GLP-1s, firmly positions Lilly as a dominant force in metabolic health. The long-term implications of these developments — for patient care, market competition, and the future trajectory of pharmaceutical innovation — are profound and will continue to unfold in the years to come. The initial investor skepticism, while notable, may simply reflect the market’s attempt to recalibrate expectations in the face of extraordinary success and the inherent complexities of a rapidly evolving, high-stakes therapeutic landscape.











Leave a Reply