In a landmark study that bridges the gap between mechanical physics and molecular biology, researchers from the Department of Medicine at the LKS Faculty of Medicine, University of Hong Kong (HKUMed), have successfully decoded the biological mechanism that translates physical movement into bone strength. The research team has identified a specific protein, Piezo1, which functions as the body’s internal "exercise sensor" within the bone marrow. This discovery provides a long-sought-after explanation for why weight-bearing exercise is essential for skeletal health and, more importantly, offers a blueprint for developing pharmacological "exercise mimetics." These potential medications could replicate the physiological benefits of physical activity for individuals who are physically unable to move, such as the elderly, the bedridden, or those suffering from chronic paralytic conditions.
The findings, recently published in the prestigious international journal Signal Transduction and Targeted Therapy, arrive at a critical juncture as global populations age at an unprecedented rate. By isolating the molecular pathways that govern bone density, the HKUMed team has moved the medical community one step closer to treating osteoporosis not just by slowing bone loss, but by actively simulating the restorative power of movement at the cellular level.
The Global Crisis of Osteoporosis and Skeletal Decay
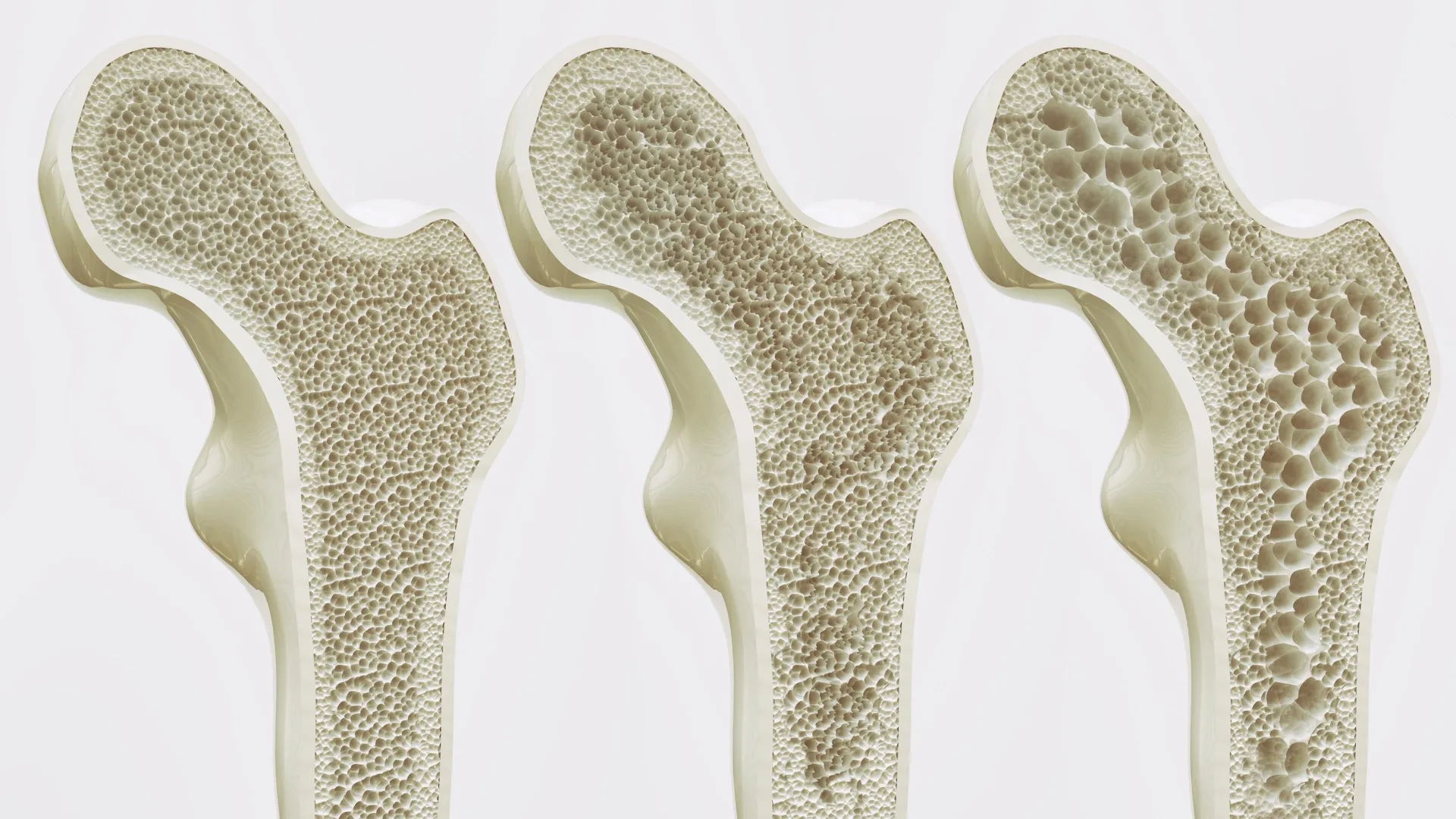
Osteoporosis is frequently described by clinicians as a "silent epidemic" because bone loss occurs without symptoms until a fracture takes place. The scale of the problem is vast; data from the World Health Organization (WHO) indicates that approximately one in three women and one in five men over the age of 50 will suffer an osteoporotic fracture in their remaining lifetime. These injuries are not merely mechanical failures but are associated with high rates of morbidity and mortality. For instance, hip fractures in the elderly often lead to a permanent loss of independence, with nearly 20% of patients requiring long-term nursing home care following the injury.
In Hong Kong, the situation is particularly acute. The city’s rapidly aging demographic has seen a surge in bone-related ailments. Statistics show that osteoporosis currently affects 45% of women and 13% of men aged 65 and older in the territory. The economic burden on the Hong Kong healthcare system is substantial, involving surgical costs, long-term rehabilitation, and the management of secondary complications arising from immobility.
The fundamental challenge in treating these populations has always been the "exercise paradox." While physical activity is the most effective natural way to maintain bone density, the very people who need it most—the frail and the elderly—are often the least capable of performing it. This creates a downward spiral where inactivity leads to bone loss, which leads to increased frailty, further preventing activity.
Understanding the Cellular Tug-of-War: Bone vs. Fat
To understand the HKUMed discovery, one must look at the micro-environment of the bone marrow. Within the marrow reside mesenchymal stem cells (MSCs), which are essentially the body’s skeletal "building blocks." These stem cells are multipotent, meaning they have the potential to differentiate into several different types of tissue. In a healthy, active individual, these cells primarily develop into osteoblasts (bone-forming cells).
However, as the body ages or remains sedentary, a pathological shift occurs. Instead of becoming bone, these stem cells increasingly differentiate into adipocytes (fat cells). This process, known as marrow adiposity, leads to a "fatty marrow" that crowds out healthy bone tissue and weakens the skeletal structure from the inside out. Until now, the exact "switch" that told a stem cell to become bone instead of fat in response to movement remained a mystery.
The Role of Piezo1: The Molecular Exercise Sensor
The HKUMed study, led by Professor Xu Aimin, Director of the State Key Laboratory of Pharmaceutical Biotechnology, utilized a combination of mouse models and human stem cell analysis to identify the protein Piezo1 as the critical sensor in this process. Piezo1 is a mechanosensitive ion channel located on the surface of mesenchymal stem cells. It acts like a biological transducer, sensing the mechanical pressure and fluid shear stress generated when a person walks, runs, or lifts weights.
The research demonstrated that when Piezo1 is activated by physical force, it triggers a signaling cascade that promotes osteogenesis (bone formation) while simultaneously suppressing adipogenesis (fat formation). In experimental models where Piezo1 was removed or "knocked out," the results were dramatic: even with physical stimulus, the bones failed to strengthen. Instead, the stem cells defaulted to fat production, and the bone marrow became riddled with fat deposits, leading to rapid bone thinning and increased fragility.
Furthermore, the team discovered that a lack of Piezo1 activity triggers the release of specific inflammatory signals, namely Ccl2 and lipocalin-2. These proteins act as chemical messengers that further accelerate the conversion of stem cells into fat and create a "toxic" environment for bone growth. By blocking these inflammatory signals in the laboratory, the researchers were able to partially mitigate the bone loss, confirming that Piezo1 sits at the top of a complex regulatory hierarchy.
Collaborative Research and Methodology
The study was a multi-year, international collaboration involving the State Key Laboratory of Pharmaceutical Biotechnology at HKUMed and the Institute of Molecular and Cellular Pharmacology at the French National Centre for Scientific Research (CNRS). The team employed advanced genetic engineering to create mice that lacked Piezo1 specifically in their bone marrow stem cells, allowing them to isolate the protein’s function without affecting other organs.
Professor Eric Honoré, a co-leader of the research from the University Côte d’Azur in France, noted that the study utilized high-resolution imaging and transcriptomic profiling to map the changes in the bone environment. This rigorous methodology allowed the team to see exactly how the "mechanical" signal of movement was converted into the "chemical" signal of gene expression.
Expert Perspectives on Clinical Applications
The identification of Piezo1 has been hailed by the scientific community as a breakthrough for regenerative medicine. Professor Xu Aimin emphasized that the goal is not to replace exercise for healthy individuals but to provide a biological lifeline for those for whom exercise is an impossibility.
"Current treatments for osteoporosis, such as bisphosphonates, primarily work by preventing the breakdown of old bone," Professor Xu explained. "However, they do not necessarily stimulate the creation of new, high-quality bone in the same way that physical activity does. By targeting the Piezo1 pathway, we are looking at a way to trick the body into thinking it is exercising. This could revolutionize care for bedridden patients who currently have very few options to prevent the rapid skeletal decay that comes with immobility."
Dr. Wang Baile, Research Assistant Professor at HKUMed and co-leader of the study, highlighted the potential for "exercise mimetics." These are compounds that could be delivered via medication to chemically activate the Piezo1 sensor. "For a patient recovering from a stroke or a spinal cord injury, the inability to move often leads to secondary bone loss that complicates their recovery. A drug that activates Piezo1 could maintain their bone mass during the healing process, ensuring that when they are ready to walk again, their skeleton is strong enough to support them."
Broader Implications and Future Trajectory
The implications of this research extend beyond the elderly. The discovery is of significant interest to space agencies like NASA and the CNSA (China National Space Administration). Astronauts in microgravity environments experience accelerated bone loss—often losing as much bone mass in a month as an elderly person does in a year—because the lack of gravity removes the mechanical load on Piezo1. "Exercise mimetics" could be a standard part of future long-duration space missions to Mars or the Moon.
Furthermore, the research provides a new lens through which to view the relationship between obesity and bone health. Since the Piezo1 pathway regulates the balance between fat and bone, it suggests that metabolic health and skeletal health are more deeply intertwined than previously thought.
The HKUMed team is now moving toward the next phase of research: drug screening. They are searching for small-molecule compounds that can safely and effectively activate Piezo1 in humans. This process involves ensuring that the activation is specific to the bone marrow to avoid potential side effects in other tissues where Piezo1 might be present, such as the lungs or blood vessels.
Conclusion and Funding Acknowledgments
The research was supported by a diverse range of prestigious grants, reflecting the global importance of the work. Contributors included the Research Grants Council of Hong Kong, the National Natural Science Foundation of China, the Human Frontier Science Program, and various French national research agencies.
As the world faces a "silver tsunami" of aging populations, the discovery of the Piezo1 exercise sensor offers a beacon of hope. By deconstructing the molecular essence of movement, HKUMed researchers have provided the foundation for a future where the benefits of a workout can be delivered in a pill, preserving the independence and health of millions who are currently sidelined by the limitations of their own bodies. The transition from bench to bedside will take time, but the map for the journey is now clearly drawn.














Leave a Reply