In a landmark development for the field of inorganic chemistry, a team of scientists at King’s College London has announced the discovery of a novel molecular form of aluminum that demonstrates the potential to replace expensive and scarce transition metals in industrial chemical processes. Led by Dr. Clare Bakewell, a Senior Lecturer in the Department of Chemistry, the research team has successfully synthesized highly reactive aluminum molecules capable of breaking some of the most resilient chemical bonds known to science. The findings, recently published in the prestigious journal Nature Communications, represent a significant leap forward in the quest for "earth-abundant catalysis," a movement aimed at reducing the global manufacturing sector’s reliance on "conflict minerals" and high-cost precious metals like platinum, palladium, and rhodium.
The discovery centers on a unique molecular architecture: a cyclotrialumane. This compound consists of three aluminum atoms arranged in a precise triangular, or trimeric, structure. While aluminum is the most abundant metal in the Earth’s crust, it has historically been viewed as less versatile than transition metals for complex chemical synthesis. However, the King’s College team has demonstrated that by manipulating the structural arrangement of aluminum atoms, they can induce levels of reactivity that were previously thought impossible for main-group elements. This breakthrough could fundamentally alter the economics of chemical production, offering a path toward greener, cheaper, and more resilient supply chains.
The Science of the Cyclotrialumane: A Structural Breakthrough
The core of the team’s research lies in the creation of the first known stable example of a cyclotrialumane. In the world of molecular chemistry, the geometry of a molecule often dictates its function. By arranging three aluminum atoms in a triangle, the researchers created a high-energy, strained system that is eager to react with other substances to reach a more stable state.
One of the most remarkable features of this new compound is its stability in solution. Often, highly reactive molecules are transient, existing for only fractions of a second before breaking down or reacting prematurely. The cyclotrialumane developed by Dr. Bakewell’s team remains intact when dissolved in various chemical solvents, providing a stable platform for controlled industrial reactions. This stability is a prerequisite for any material intended to serve as a catalyst or reagent in large-scale manufacturing.
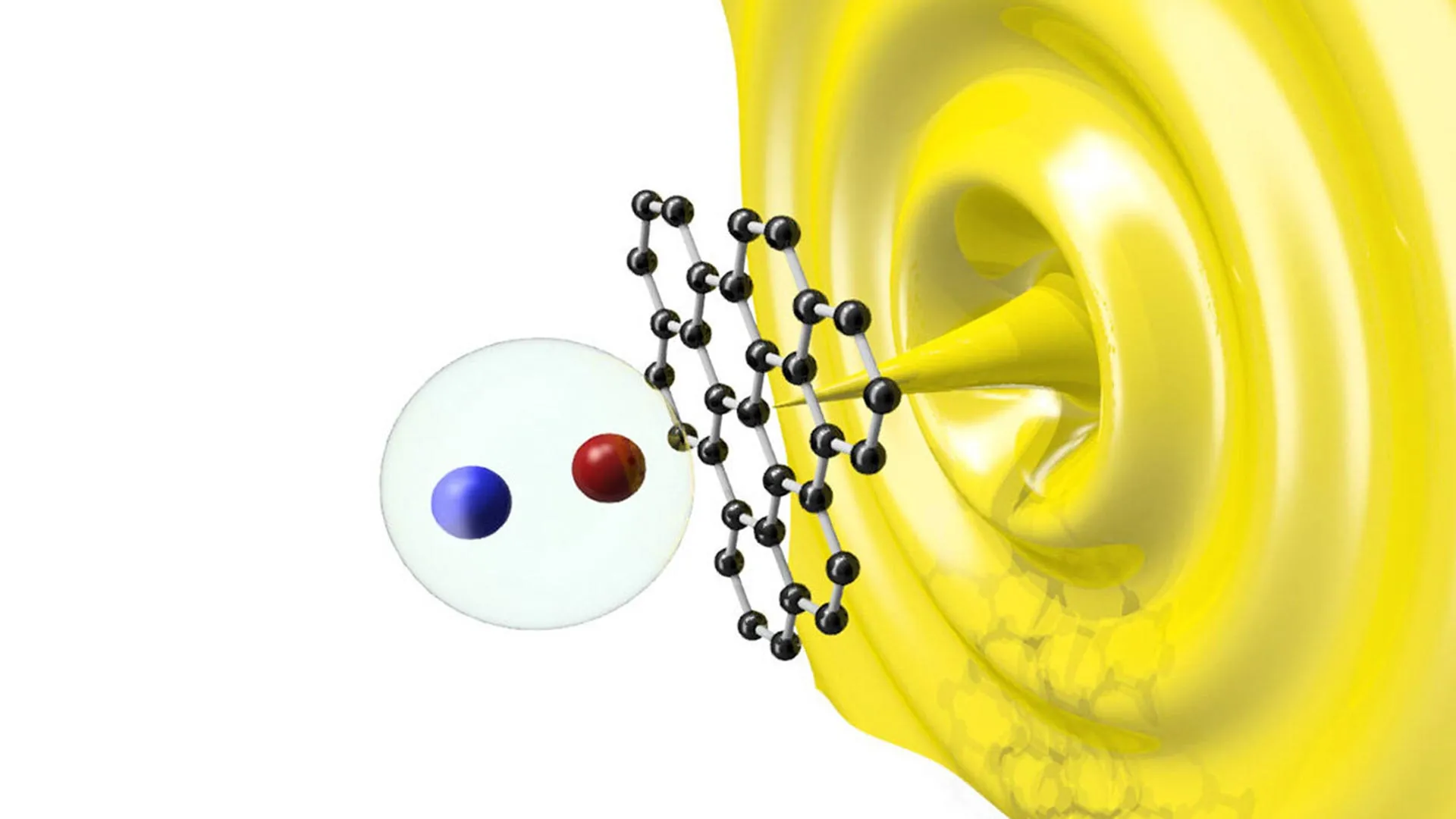
The reactivity of this aluminum trimer was put to the test against dihydrogen (H2) and ethene (C2H4). The molecule successfully split dihydrogen bonds—a process typically reserved for expensive transition metal catalysts like platinum—and facilitated the step-by-step insertion and chain growth of ethene. Ethene is a fundamental building block in the petrochemical industry, used to create everything from plastics to antifreeze. The ability of an aluminum-based compound to manage these reactions suggests that it could eventually replace the heavy metals currently used in polymer science and fuel production.
Economic Implications: Aluminum vs. Precious Metals
The motivation behind the search for aluminum-based alternatives is rooted in stark economic reality. Modern industry is heavily dependent on transition metals, particularly those in the Platinum Group Metals (PGMs). These elements are essential for catalytic converters in automobiles, the production of fertilizers, and the synthesis of pharmaceuticals. However, their scarcity and the geographic concentration of their reserves create significant market volatility.
According to current market data, platinum often trades at prices exceeding $900 to $1,000 per ounce, while palladium can reach even higher peaks depending on supply chain disruptions. In contrast, aluminum is priced by the ton, typically costing around $2,200 to $2,500 per metric ton. As Dr. Bakewell noted, aluminum is approximately 20,000 times less expensive than the precious metals it seeks to mimic.
Beyond the raw cost of the material, there is the issue of supply chain security. Most of the world’s transition metals are extracted from a small handful of regions, including South Africa and Russia. Political instability or trade sanctions in these areas can lead to immediate spikes in the cost of essential chemicals. Aluminum, being the third most abundant element in the Earth’s crust after oxygen and silicon, is available globally. Shifting toward aluminum-based chemistry would democratize chemical production and shield industries from the geopolitical tensions associated with rare earth and precious metal mining.
Chronology of the Discovery and Research Context
The journey toward this discovery began several years ago as part of a broader academic trend focusing on "Main Group Chemistry." For decades, the chemical industry relied on the d-orbitals of transition metals to facilitate complex electron transfers. Main-group elements like aluminum (found in the p-block of the periodic table) were generally considered "boring" by comparison, usually appearing in stable +3 oxidation states that offered little catalytic utility.
However, over the last decade, researchers began exploring "low-valent" main-group chemistry—trying to force elements like aluminum, magnesium, and silicon into unusual oxidation states or geometries to mimic the behavior of transition metals. The Bakewell Group at King’s College London focused specifically on the potential of aluminum clusters.
The timeline of the breakthrough can be traced through several key phases:
- Theoretical Modeling: Initial computational studies to determine if a triangular aluminum structure could remain stable while retaining high reactivity.
- Synthesis and Isolation: The team successfully synthesized the cyclotrialumane in the laboratory, using specialized ligands to "wrap" the aluminum core and prevent it from collapsing.
- Validation: Using X-ray crystallography and Nuclear Magnetic Resonance (NMR) spectroscopy, the team confirmed the triangular structure.
- Reactivity Testing: The final phase involved exposing the compound to various substrates, leading to the discovery of the 5- and 7-membered aluminum and carbon rings—structures that had never been observed in traditional chemistry.
The publication of their findings in Nature Communications serves as the culmination of this rigorous experimental cycle, moving the concept from a theoretical possibility to a proven molecular reality.
Environmental Impact and the Transition to "Green Chemistry"
The environmental benefits of this discovery extend beyond the avoidance of rare metal mining. The extraction of platinum and other PGMs is an energy-intensive process that often involves the use of toxic chemicals and results in significant carbon emissions and habitat destruction. Furthermore, the "catalyst footprint"—the environmental cost of producing the catalyst itself—is a major factor in the life-cycle analysis of chemical products.
Aluminum production, while energy-intensive, has seen significant strides in sustainability through recycling. Aluminum can be recycled infinitely with only 5% of the energy required for primary production. By developing catalysts based on an element that is already deeply integrated into circular economy infrastructures, the chemical industry could significantly lower its total environmental impact.
Moreover, the reactivity of the aluminum trimer allows for more efficient reaction pathways. In industrial chemistry, "efficiency" translates to lower temperatures and pressures required for a reaction to occur. If aluminum-based catalysts can facilitate these reactions under milder conditions than current transition metal-based systems, the energy savings across the global manufacturing sector could be monumental.
Expert Analysis: Pushing the Boundaries of Chemical Knowledge
The scientific community has reacted with cautious optimism to the King’s College findings. Independent analysts suggest that while the research is in its "exploratory phase," the discovery of 5- and 7-membered aluminum-carbon rings is a "frontier-pushing" event. These structures are not merely mimics of existing transition metal processes; they represent entirely new types of chemical behavior.
Dr. Bakewell emphasized this distinction in her statements, noting that the team is not just looking for a "cheaper version" of platinum. "What’s special about this work is that we’re pushing the boundaries of chemical knowledge," she explained. "We can use this aluminum trimer to build completely new compounds… these capabilities go beyond the transition metals we were originally trying to mimic."
The ability to create larger, more complex ring structures through the reaction with ethene suggests that this chemistry could lead to the development of entirely new materials. These might include specialized polymers with unique thermal or mechanical properties that cannot be produced using current catalytic methods.
Challenges and the Path Toward Industrial Integration
Despite the promise of the cyclotrialumane, several hurdles remain before it can be deployed in a factory setting.
- Scalability: The current synthesis of the aluminum trimer is conducted on a laboratory scale. Scaling this up to produce the quantities needed for industrial catalysis requires further engineering.
- Sensitivity: Like many highly reactive molecules, these aluminum compounds can be sensitive to air and moisture. Developing versions that are robust enough for use in standard industrial environments is a key goal for future research.
- Longevity: In industrial processes, a catalyst must be able to perform thousands of cycles before it is spent. The "turnover frequency" and "turnover number" of this new aluminum compound will need to be optimized to compete with the longevity of platinum-based systems.
Conclusion: A New Era for Earth-Abundant Materials
The work of the Bakewell Group at King’s College London marks a turning point in the field of sustainable chemistry. By unlocking the hidden potential of aluminum, the researchers have provided a blueprint for a future where the production of medicines, plastics, and fuels is no longer tethered to the high costs and environmental degradation associated with rare earth metals.
While the transition to a purely aluminum-based chemical economy may take years or even decades, the identification of the cyclotrialumane proves that the "workhorses" of the periodic table are more capable than previously imagined. As the global community continues to prioritize green energy and sustainable manufacturing, discoveries like this will be essential in bridging the gap between current industrial requirements and future environmental goals. The "exploratory phase" may just be beginning, but the foundations have been laid for a cleaner, greener, and more affordable chemical industry.














Leave a Reply