The digital health company MEDVi, recently spotlighted in an April 2 New York Times profile that depicted CEO Matthew Gallagher as a "vibe-coded unicorn," has rapidly become the subject of intense journalistic and regulatory blowback. While initial scrutiny focused on the company’s GLP-1 weight-loss drug offerings, a new investigation reveals significant concerns surrounding QUAD, a compounded erectile dysfunction (ED) product marketed by MEDVi that includes apomorphine, a drug primarily approved in the U.S. for Parkinson’s disease and not for ED. This revelation adds another layer of complexity to the mounting questions about MEDVi’s operational practices, regulatory compliance, and patient safety protocols.
The Genesis of Scrutiny: From Unicorn Status to Regulatory Red Flags
MEDVi’s ascent to prominence, fueled by its aggressive use of AI in advertising and a direct-to-consumer telehealth model, seemed to position it as a disruptor in the health tech space. The New York Times article aimed to capture this innovative spirit, highlighting Gallagher’s vision and the company’s rapid growth. However, the glowing portrayal quickly unraveled as a series of investigative reports from prominent outlets challenged the narrative, exposing a foundation riddled with regulatory warnings, legal challenges, and questionable marketing tactics.
Futurism, Forrester, Drug Discovery & Development, and Business Insider were among the first to publish critical reports in the days following the NYT piece. Their findings collectively revealed a February 2026 FDA warning letter issued to MEDVi LLC for misbranding compounded GLP-1 drugs, a string of lawsuits challenging the company’s practices, and a pattern of using Meta advertisements featuring apparently AI-generated "doctor" personas. Additionally, concerns emerged regarding the oversight of MEDVi’s extensive affiliate marketing network, which often operates with varying degrees of transparency and adherence to medical advertising standards. This rapid shift from media darling to a company under the microscope underscored the precarious balance between tech-driven innovation and the stringent regulatory environment governing pharmaceuticals.

Unpacking QUAD: A Four-Drug Cocktail with Unapproved Components
Amidst the broader controversies, MEDVi’s compounded erectile dysfunction product, QUAD, has emerged as a distinct and particularly concerning area of inquiry. QUAD is marketed as a "fast 4-in-1 ED formula" delivered as a single sublingual dose. Its composition is a potent combination: three phosphodiesterase-5 (PDE5) inhibitors—sildenafil (commonly known as Viagra), tadalafil (Cialis), and vardenafil (Levitra)—alongside apomorphine.
The inclusion of apomorphine is the most striking aspect, given its primary U.S. FDA approval for managing motor fluctuations (OFF episodes) in adults with advanced Parkinson’s disease. Its use in veterinary medicine is even more divergent, where it is employed to induce vomiting in dogs. Crucially, apomorphine is not FDA-approved in the UnitedS. for the treatment of erectile dysfunction. This immediately flags QUAD as a compounded drug incorporating an off-label component for a condition for which it lacks U.S. regulatory clearance.
The Perilous History of Apomorphine for ED
The history of apomorphine’s use for erectile dysfunction is fraught with challenges and regulatory setbacks, a context largely absent from MEDVi’s marketing. In Europe, apomorphine was indeed marketed for ED beginning in May 2001. However, its authorization by the European Medicines Agency (EMA) expired in May 2006, with its then-developer, Abbott Laboratories, opting not to renew it for commercial reasons. This decision followed widespread reports of its limited efficacy and notable side effects. A UK study involving 11,185 patients found that a significant two-thirds discontinued its use, citing a lack of perceived effectiveness. Nausea was a commonly reported adverse event. While a 2020 systematic review and meta-analysis in Archives of Sexual Behavior offered a more positive assessment, it noted that sublingual apomorphine was generally well-tolerated only at lower doses (2 mg and 3 mg), with discontinuation due to adverse events climbing significantly at higher doses.
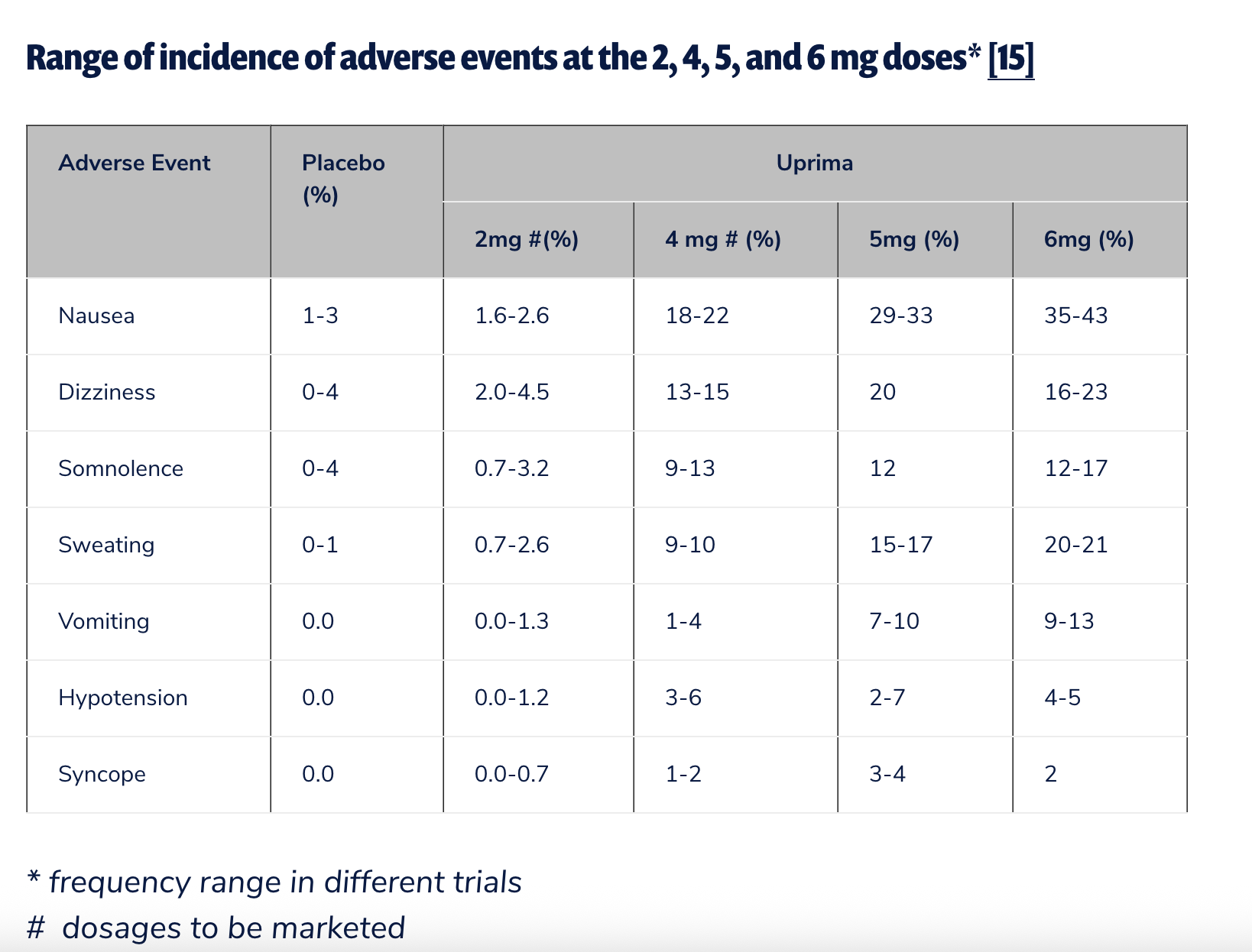
The U.S. experience with apomorphine for ED was even more conclusive. In the early 2000s, a joint venture between Abbott and Takeda, TAP Pharmaceuticals, sought FDA approval for a sublingual apomorphine product under the brand name Uprima. However, the application was withdrawn in June 2000 after an FDA advisory committee raised serious concerns about risks of hypotension (low blood pressure) and syncope (fainting). Public Citizen, a consumer advocacy organization, played a pivotal role in opposing Uprima’s approval. Sidney Wolfe, M.D., then director of Public Citizen’s Health Research Group, penned a blistering letter to Dr. Janet Woodcock, then director of the FDA’s Center for Drug Evaluation and Research (CDER), urging outright rejection. Dr. Wolfe famously warned, "If the FDA approves this drug, it is certain that it will have to be banned within a short time after marketing begins because of the high probability of further occurrence of the serious injuries it has already been found to cause." FDA approval for ED in the U.S. never materialized, cementing apomorphine’s unapproved status for this indication.
The Dangers of "Stacking" PDE5 Inhibitors and Apomorphine
Beyond the individual concerns surrounding apomorphine, the combination of three PDE5 inhibitors with apomorphine in a single dose raises substantial safety questions. PDE5 inhibitors, while effective for ED, are known to cause a decrease in blood pressure and are contraindicated with certain medications, such as nitrates, due to the risk of dangerous hypotension. They also carry warnings regarding interactions with alpha-blockers and some blood pressure medications.
The FDA has explicitly addressed the issue of combining PDE5 inhibitors. Labeling for sildenafil, for instance, notes that combinations of PDE5 inhibitors "have not been studied" and "may further lower blood pressure," concluding unequivocally that "the use of such combinations is not recommended."
Professional drug-interaction databases widely used by U.S. pharmacies and electronic health records echo these warnings. For example, Drugs.com classifies the combination of tadalafil and apomorphine as a "Major" interaction, its highest tier. This classification indicates a high risk of additive blood-pressure lowering, potentially leading to dizziness, fainting, and dangerous changes in heart rate. These databases also flag hypotension as a "Major" disease contraindication for tadalafil, specifically cautioning against its use in patients with resting blood pressure below 90/50 mmHg. Furthermore, they trigger a therapeutic-duplication warning when more than one erectile dysfunction agent is prescribed simultaneously, stating that the recommended maximum is one. QUAD, containing four such agents, dramatically exceeds this recommended limit. This "stacking" approach, while potentially appealing to those seeking stronger or faster effects, introduces a compounded and largely unstudied risk profile that deviates significantly from established medical guidelines and regulatory precautions.
The Compounding Conundrum: Regulatory Loopholes and Patient Safety
MEDVi’s reliance on compounded drugs lies at the heart of many of these controversies. Compounded drugs are custom-prepared medications made by a licensed pharmacist to meet the specific needs of an individual patient, typically when an FDA-approved drug is not available or suitable. Unlike FDA-approved drugs, compounded drugs are not subject to premarket review for safety, effectiveness, or quality by the FDA. This means their manufacturing processes, purity, potency, and stability are not verified by the agency.
While traditional compounding serves a vital niche in healthcare, the model employed by companies like MEDVi—large-scale production and aggressive direct-to-consumer marketing of compounded formulations—blurs the lines between compounding and manufacturing. This practice often operates in a regulatory gray area, where the volume and scope of distribution resemble those of a pharmaceutical manufacturer, but without adhering to the rigorous safety and efficacy standards required for FDA approval. The FDA has consistently warned against such practices, particularly when compounded drugs are marketed with unsubstantiated claims or contain unapproved active pharmaceutical ingredients for the specified indication. The agency’s February 2026 warning letter to MEDVi for its GLP-1 products specifically cited misbranding, highlighting the FDA’s concern over how these compounded drugs are being presented and sold to the public.
Flawed Onboarding and Misleading Success Probabilities
Drug Discovery & Development‘s test runs on April 7, 2026, exposed significant flaws in MEDVi’s patient onboarding process for QUAD, further amplifying concerns about patient safety. The initial intake questionnaire, designed to determine eligibility for treatment, appeared to accept free-form responses that did not directly answer the posed questions, suggesting a lack of robust screening mechanisms.
More critically, when asked about blood pressure, the system presented a question: "diagnosed with high blood pressure (hypertension) or low blood pressure (hypotension)." However, the available answer cards only referred to high blood pressure. Selecting "Yes, I have high blood pressure" correctly halted the intake with an "Eligibility Status" message indicating that the assessment could not be completed. Disturbingly, there was no corresponding option or off-ramp for patients who might have low blood pressure (hypotension), a condition particularly vulnerable to the hypotensive effects of QUAD’s drug components. This glaring omission suggests a failure in the gating logic, potentially allowing individuals at higher risk of adverse events to proceed through the intake process unchecked.
Adding to the skepticism, across multiple QUAD intake test runs, users who were not screened out were consistently presented with a "Your Medical Review" screen displaying a "Success Probability" of 94%. This identical 94% figure has also been observed in MEDVi’s GLP-1 weight-loss intake process, as documented in a screen recording filed as an exhibit in the federal class-action lawsuit Day v. OpenLoop Health. The unchanging nature of this "success probability" across different products and varied user inputs strongly suggests it is a generic, pre-programmed marketing claim rather than a personalized, evidence-based assessment. Such a practice could be highly misleading, creating a false sense of security and efficacy for potential patients.
Affiliate Marketing: Distancing from Scrutiny While Promoting Unapproved Products
MEDVi’s sprawling affiliate marketing network plays a crucial role in its direct-to-consumer strategy, yet it also highlights a complex relationship with regulatory scrutiny. An affiliate marketing page titled "MEDVi QUAD Reviews: Is This Fast 4-in-1 ED Formula Worth the Hype?" allows users to "check your eligibility for MEDVi QUAD in minutes." While it acknowledges that the therapy "may not be suitable for individuals with certain medical conditions or those taking specific medications" and requires a "clinician review," it concurrently promotes the product’s benefits.
Another affiliate page, posted on MedicalFoundationofNC.org, attempts to differentiate the regulatory issues, stating, "the FDA warning letter and the GLP-1 regulatory crackdown are about a different product line" and that the February 2026 warning letter "did not reference MEDVi’s QUAD ED product line." While technically true that the specific warning letter addressed GLP-1 products, this framing potentially downplays the broader pattern of regulatory concern surrounding MEDVi’s practices. The page includes disclaimers such as "ask hard questions before enrolling," "not medical advice," and explicitly states that MedicalFoundationofNC.org is "not a medical practice, pharmacy, or licensed healthcare provider" and "not affiliated with The Medical Foundation of North Carolina, Inc., UNC Health Foundation, UNC Health Care, the UNC School of Medicine, or the University of North Carolina at Chapel Hill." These disclaimers, while legally necessary, underscore the indirect and often arms-length nature of how these products are promoted and the potential for consumers to misunderstand the source and authority of the information they receive.
Broader Implications: Telehealth, Compounding, and Consumer Trust
The MEDVi saga, particularly concerning QUAD, encapsulates several critical issues facing the modern healthcare landscape. The rapid expansion of telehealth platforms and direct-to-consumer models, while offering convenience and accessibility, also presents challenges for robust medical oversight and regulatory enforcement. When combined with large-scale compounding practices that push the boundaries of traditional pharmacy, the potential for patient harm increases significantly.
The case of MEDVi highlights a growing tension between the venture capital-backed "move fast and break things" ethos of tech startups and the inherently cautious, patient-safety-first principles of pharmaceutical regulation. The "vibe-coded unicorn" narrative, while appealing to investors, can obscure fundamental questions about scientific rigor, clinical evidence, and ethical marketing.

For patients, the implications are profound. Lured by promises of rapid efficacy and tailored solutions, they may unwittingly expose themselves to unstudied drug combinations with potentially serious side effects, all while believing they are receiving legitimate medical care. The lack of transparent, individualized risk assessment during onboarding, coupled with generic "success probability" figures, erodes the trust essential for a healthy doctor-patient relationship and informed consent.
Official Silence and Ongoing Legal Battles
In response to inquiries regarding QUAD and the broader issues, Drug Discovery & Development reported that MEDVi and OpenLoop Health did not immediately provide a comment. This lack of public response from the companies directly involved leaves many questions unanswered, particularly concerning the rationale behind the QUAD formulation and the safety protocols in place. Nicholas Chimicles, lead counsel for the plaintiffs in Day v. OpenLoop Health, confirmed his firm’s awareness of the New York Times profile of MEDVi but declined to comment further on the ongoing litigation, which mentions MEDVi though it is not a direct defendant.
The silence from MEDVi and OpenLoop Health, coupled with the detailed investigative reports and the history of FDA warnings and consumer advocacy, paints a concerning picture. As the regulatory landscape continues to evolve to address the complexities of telehealth, AI in healthcare, and compounded medications, the MEDVi case stands as a stark reminder of the critical importance of rigorous oversight, scientific integrity, and unwavering commitment to patient safety in the pursuit of innovation. The future trajectory of MEDVi, and indeed the broader direct-to-consumer health industry, will undoubtedly be shaped by how these profound challenges are ultimately addressed.















Leave a Reply