A collaborative team of researchers from the University of Michigan and Hokkaido University has finally resolved one of the most enduring mysteries in the field of geology, a conundrum famously known as the Dolomite Problem. For more than 200 years, scientists have struggled to explain why the mineral dolomite is so prevalent in ancient rock formations yet remains almost impossible to grow in a modern laboratory setting under natural conditions. The solution, recently published in the journal Science, utilized cutting-edge atomic simulations and electron microscopy to demonstrate that dolomite growth requires periodic fluctuations in its environment to strip away structural defects.
This breakthrough not only settles a geological debate dating back to the late 18th century but also provides a revolutionary framework for manufacturing high-tech materials. By understanding the mechanism that allows dolomite to crystallize, engineers may be able to develop new techniques for producing defect-free semiconductors, solar cells, and battery components at a much faster rate than previously thought possible.
The Historical Context of the Dolomite Problem
Dolomite, a calcium magnesium carbonate mineral, was first described in 1791 by the French naturalist and geologist Déodat de Dolomieu. He identified the mineral in the mountain range in the northern Italian Alps that now bears his name—the Dolomites. These mountains, recognized as a UNESCO World Heritage site, are composed of massive layers of the mineral, as are other iconic landmarks such as the Niagara Falls cliffs and the Hoodoos of Bryce Canyon National Park.
Despite its abundance in the Earth’s crust—accounting for approximately 2% of all sedimentary rocks—dolomite has remained an anomaly. Geologists noted that while rocks older than 100 million years are rich in dolomite, the mineral is conspicuously absent in more recent geological formations. For decades, researchers attempted to replicate the mineral’s growth in laboratory settings to understand this discrepancy, but they consistently failed to produce any significant amount of dolomite at room temperature and pressure. This inability to grow the mineral under conditions that seemingly mimic nature led to the "Dolomite Problem," a staple of geological textbooks for generations.
The Atomic Barrier to Growth
The research team, led by Wenhao Sun, the Dow Early Career Professor of Materials Science and Engineering at the University of Michigan, focused on the atomic-level interactions that occur during crystallization. Unlike calcite, which is a simple calcium carbonate, dolomite consists of highly ordered, alternating layers of calcium and magnesium.
In an aqueous environment, atoms from the surrounding water attach themselves to the surface of a growing crystal. For dolomite to form correctly, calcium and magnesium ions must arrange themselves in a specific, alternating sequence. However, the researchers discovered that the chemical similarity between calcium and magnesium ions leads to frequent "misplacements." As the mineral grows, magnesium ions often land on sites intended for calcium, and vice versa.
These misplaced atoms create structural defects that prevent the next layer of the crystal from attaching properly. In a stable environment, these defects become permanent "stumbling blocks" that grind the growth process to a halt. The researchers calculated that under constant conditions, it would take roughly 10 million years to grow a single well-ordered layer of dolomite. This staggering timescale explains why the mineral is rarely seen forming in modern, stable environments.
Simulating Geological Time in Milliseconds
To uncover how nature overcomes this barrier, the University of Michigan team utilized the Predictive Structure Materials Science (PRISMS) Center’s computational power. Simulating crystal growth at the atomic level is notoriously difficult because it requires calculating the energy states of countless electron-atom interactions.
"Each atomic step would normally take over 5,000 CPU hours on a supercomputer," noted Joonsoo Kim, a doctoral student at U-M and the study’s first author. "Now, we can do the same calculation in two milliseconds on a desktop."
The team developed a new software capability that leveraged the symmetry of the crystal structure to extrapolate energy predictions. This allowed them to simulate the growth of dolomite over timescales that reflect real-world geological processes. The simulations revealed a critical insight: the structural defects that block growth are less stable than the correctly aligned atoms. When the environment surrounding the crystal changes—specifically, when the concentration of minerals in the water drops—the unstable, misplaced atoms are the first to dissolve.
Nature’s Reset: The Role of Environmental Fluctuations
The study proposes that dolomite growth in nature is not a continuous process but a series of "growth and reset" cycles. In environments such as coastal lagoons or tidal flats, the mineral is subjected to frequent changes in salinity and water concentration due to rainfall, evaporation, and tidal cycles.
During periods of high concentration, a layer of dolomite begins to form, but it is riddled with defects. When it rains or the tide changes, the surrounding water becomes less saturated, causing the disordered, unstable defects to dissolve away. This "washing" process leaves behind a clean, stable, and correctly ordered surface. When the concentration rises again, a new layer can form on top of the perfect template.
Over millions of years, these repeated cycles of growth and partial dissolution allow the mineral to accumulate into the massive formations seen in the geological record. This theory aligns perfectly with the locations where dolomite is still found forming today, such as the hypersaline lagoons of Brazil, which experience frequent fluctuations in water chemistry.
Experimental Validation via Electron Microscopy
To prove the theory, the Michigan team collaborated with Yuki Kimura, a professor of materials science at Hokkaido University, and postdoctoral researcher Tomoya Yamazaki. The challenge was to recreate the "growth and wash" cycle in a laboratory setting.
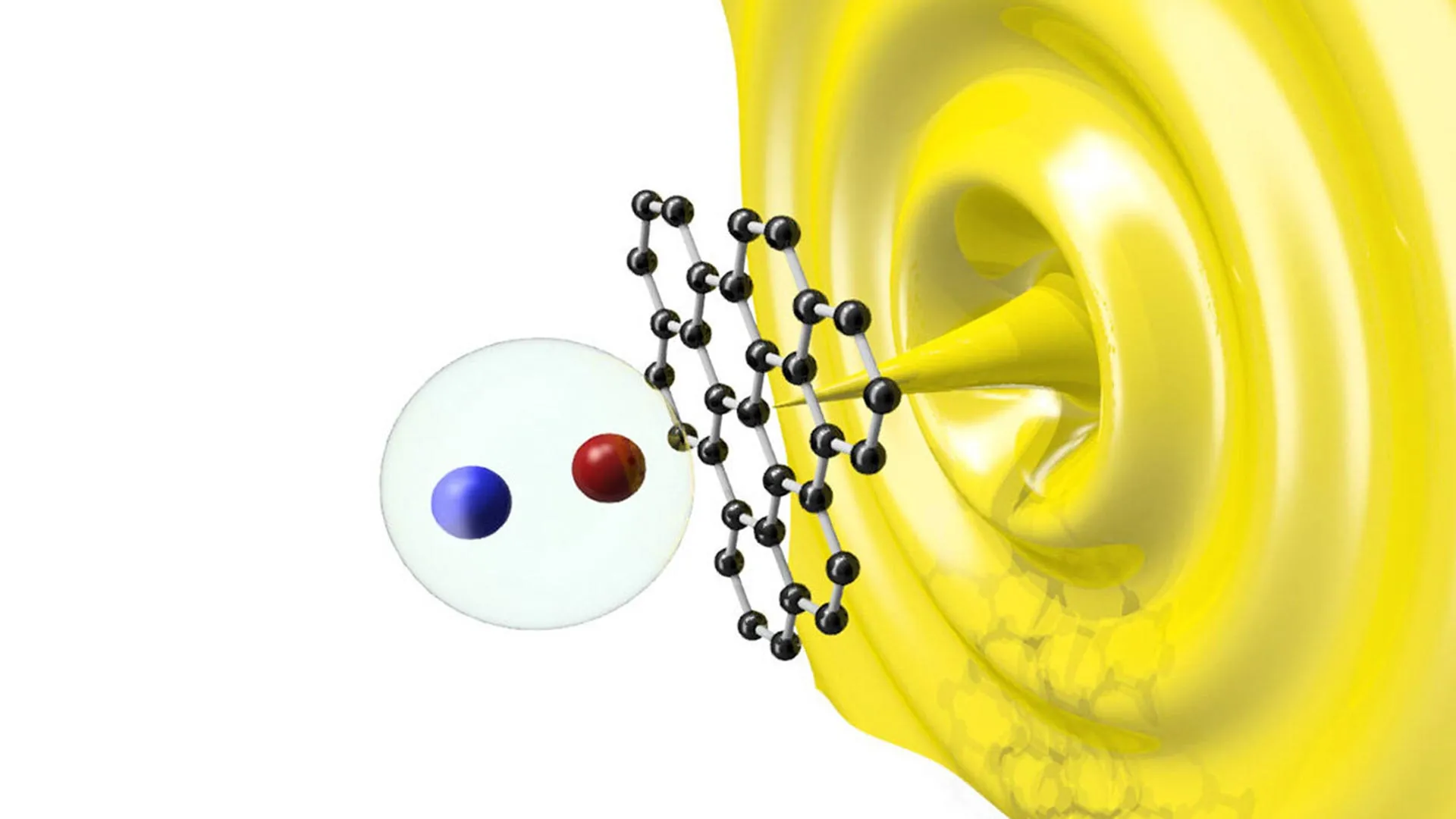
The researchers used a transmission electron microscope (TEM) equipped with a specialized liquid cell. In a unique twist, they utilized the microscope’s electron beam as a tool for manipulation rather than just imaging. By pulsing the electron beam, the team could split the water in the solution, creating a mild acid. This acid served to dissolve the nascent dolomite crystal’s surface defects.
The experiment involved placing a tiny dolomite seed crystal in a solution of calcium and magnesium. They pulsed the beam 4,000 times over a two-hour period. The result was unprecedented: the crystal grew by approximately 100 nanometers, representing roughly 300 layers of new dolomite. Prior to this experiment, the most successful laboratory attempts had yielded no more than five layers. This successful growth provided the "smoking gun" evidence that periodic dissolution is the key to dolomite formation.
Broader Implications for Materials Science and Technology
The resolution of the Dolomite Problem has significant implications far beyond the realm of geology. The "growth through dissolution" strategy offers a new paradigm for materials science. Traditionally, the industry has operated under the assumption that to create a perfect, defect-free crystal, one must grow it very slowly to allow atoms to find their correct positions.
"Our theory shows that you can grow defect-free materials quickly, if you periodically dissolve the defects away during growth," said Wenhao Sun.
This insight could be transformative for several high-tech industries:
- Semiconductors: The production of silicon wafers and other semiconducting materials requires near-perfect crystalline structures. Implementing pulsed growth cycles could reduce manufacturing time while increasing yield and performance.
- Renewable Energy: High-efficiency solar panels rely on the precise arrangement of atoms in perovskite and other crystalline materials. Controlled dissolution could help eliminate the defects that currently limit the lifespan and efficiency of these cells.
- Energy Storage: The next generation of solid-state batteries depends on the integrity of crystalline electrolytes. Faster, more reliable methods for growing these materials could accelerate the transition to electric vehicles.
- Pharmaceuticals: Many life-saving drugs are delivered in crystalline form. Controlling the growth process could lead to more stable and effective medications.
Chronology of the Discovery
- 1791: Déodat de Dolomieu identifies dolomite in the Alps.
- 1800s-1900s: Geologists identify the "Dolomite Problem" as they fail to grow the mineral in labs.
- Early 2000s: Advances in computational materials science allow for more detailed atomic modeling.
- 2020: The University of Michigan’s PRISMS Center begins developing the software necessary to simulate complex crystal growth.
- 2022: The U-M team develops the theory of "defect-mediated growth inhibition" and the necessity of dissolution cycles.
- 2023: Hokkaido University conducts the TEM pulsed-beam experiments, successfully growing 300 layers of dolomite.
- November 2023: The findings are published in the journal Science, officially providing a solution to the 232-year-old mystery.
Conclusion
The study, titled "Dissolution enables dolomite crystal growth near ambient conditions," was supported by several major institutions, including the American Chemical Society, the U.S. Department of Energy, and the Japanese Society for the Promotion of Science. By bridging the gap between theoretical physics, computational chemistry, and experimental mineralogy, the research team has not only solved a historical enigma but has also opened a new door for the future of material engineering.
As geologists look back at the massive cliffs of the Dolomites, they now see more than just ancient stone; they see a record of a planet in constant flux, where the simple act of washing away imperfection allowed for the creation of some of the world’s most spectacular landscapes. For the scientific community, the message is clear: sometimes, the secret to building something perfect is knowing when to let parts of it melt away.













Leave a Reply