The landscape of neurodegenerative disease research has reached a pivotal turning point as scientists move beyond symptom management toward therapies that alter the biological progression of Alzheimer’s disease. For decades, the medical community struggled to find interventions that could effectively clear the toxic protein accumulations associated with cognitive decline. While the recent FDA approval of monoclonal antibodies has provided the first evidence that clearing amyloid beta can slow clinical decline, these treatments come with significant logistical and physiological burdens. In a groundbreaking study published on March 5 in the journal Science, researchers at Washington University School of Medicine in St. Louis have unveiled a novel cellular immunotherapy that leverages the brain’s own architecture to combat the disease. By genetically engineering astrocytes—the most abundant support cells in the central nervous system—to act as specialized "scavengers," the team has demonstrated a method that could potentially replace frequent, lifelong infusions with a single targeted injection.
The Evolution of Alzheimer’s Therapeutics and the Amyloid Hypothesis
Alzheimer’s disease is a progressive neurological disorder characterized by the accumulation of amyloid beta proteins, which aggregate into "plaques" between neurons. These plaques are thought to trigger a secondary cascade of neurofibrillary tangles composed of tau protein, leading to widespread inflammation, synaptic loss, and eventually, the death of brain cells. For years, the "amyloid hypothesis" was debated within the scientific community, but the recent success of monoclonal antibodies like lecanemab and donanemab has largely validated the theory. These drugs have shown that by reducing the "amyloid burden" in the brain, patients can retain their cognitive independence for approximately 10 to 12 months longer than those on a placebo.
However, these current gold-standard treatments are not without their drawbacks. Monoclonal antibodies require patients to undergo intravenous infusions once or twice a month at specialized clinics. Furthermore, these drugs often struggle to cross the blood-brain barrier effectively, necessitating high doses that increase the risk of Amyloid-Related Imaging Abnormalities (ARIA), such as brain swelling or microhemorrhages. The search for a more sustainable, "one-and-done" delivery system led the Washington University team to look toward the field of oncology for inspiration.
From Cancer Treatment to Neurology: The CAR-T Inspiration
The new strategy developed at Washington University is an adaptation of Chimeric Antigen Receptor (CAR) T-cell therapy, a revolutionary treatment that has transformed the prognosis for certain blood cancers. In traditional CAR-T therapy, a patient’s immune T-cells are extracted, genetically modified to express a receptor that recognizes a specific protein on cancer cells, and then re-introduced into the body. These "living drugs" then seek out and destroy the malignancy.
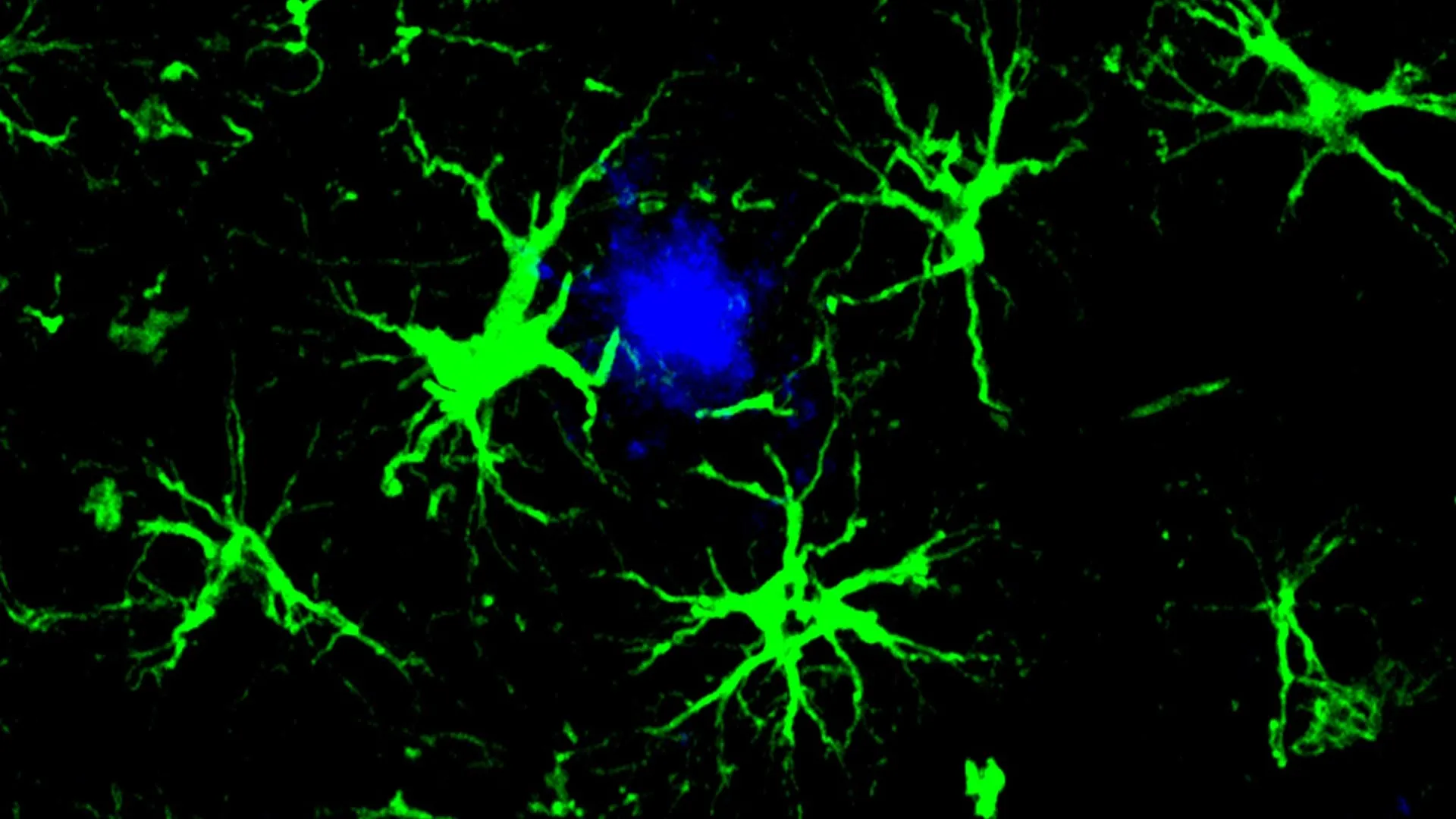
In the context of the brain, however, T-cells are not the ideal candidates for long-term residence due to the risk of excessive inflammation within the confined space of the skull. Instead, the research team, led by senior author Marco Colonna, MD, and first author Yun Chen, PhD, focused on astrocytes. Unlike T-cells, astrocytes are native to the brain and are responsible for maintaining the blood-brain barrier, providing nutrients to neurons, and regulating synaptic activity. By equipping these cells with a "CAR homing device," the researchers effectively turned the brain’s own maintenance crew into a specialized cleaning force capable of identifying and engulfing amyloid plaques.
The Biological Mechanism: Why Microglia Fail and Astrocytes Succeed
Under normal physiological conditions, the brain possesses an innate immune system led by microglia. These cells act as the primary macrophages of the central nervous system, constantly patrolling the environment for cellular debris and pathogens. In the early stages of Alzheimer’s, microglia are effective at clearing small amounts of amyloid. However, as the disease progresses, the sheer volume of plaque becomes overwhelming. Microglia can become "exhausted" or enter a state of chronic inflammation, where they no longer clear plaques efficiently and instead contribute to the damage of surrounding healthy tissue.
Astrocytes, while not primary immune cells, have a latent ability to perform phagocytosis—the process of engulfing and digesting large particles. The Washington University study capitalized on this. By introducing a specific gene via a harmless viral vector, the researchers programmed the astrocytes to express a receptor specifically tuned to amyloid beta. This modification allows the astrocytes to bypass the "exhaustion" phase seen in microglia. Because astrocytes are physically anchored in the brain tissue and exist in massive numbers, they provide a stable, long-term platform for plaque clearance that does not rely on the constant recruitment of new immune cells from the bloodstream.
Experimental Methodology and Findings in Murine Models
The research was conducted using mice genetically predisposed to develop Alzheimer’s-like pathology. These mice typically begin to show significant amyloid buildup by six months of age, mirroring the early clinical stages of the human disease. The study utilized a two-pronged experimental design to test both the preventative and therapeutic potential of the CAR-astrocytes.
In the first group, the engineered treatment was administered to young mice before any plaques had formed. Using a single injection of a viral vector carrying the CAR gene, the researchers observed the mice over several months. By the time the mice reached the six-month mark—a point where untreated mice would typically have brains saturated with amyloid—the treated mice showed virtually no detectable plaque formation. This suggests that CAR-astrocytes could serve as a powerful prophylactic if administered during the "preclinical" stage of Alzheimer’s, where biomarkers indicate future risk but symptoms have not yet appeared.
In the second group, the treatment was administered to older mice that already possessed a high "plaque load." This scenario more closely resembles the typical human patient who receives a diagnosis after cognitive symptoms are already present. The results were equally significant: within three months of the single injection, the levels of amyloid in the brain were reduced by approximately 50 percent compared to the control group. This reduction is comparable to, or in some cases exceeds, the clearance rates seen with monthly antibody infusions, but achieved with a significantly lower frequency of intervention.
Comparative Analysis: Single Injection vs. Chronic Infusions
The implications of a "single injection" therapy are profound for both healthcare infrastructure and patient quality of life. Current monoclonal antibody treatments require significant resources, including infusion center staffing, regular MRI monitoring for ARIA, and the logistical burden on caregivers to transport elderly patients to appointments every two weeks.
"Consistent with the antibody drug treatments, this new CAR-astrocyte immunotherapy is more effective when given in the earlier stages of the disease," noted David M. Holtzman, MD, a co-author of the study and a renowned figure in Alzheimer’s research. "But where it differs, and where it could make a difference in clinical care, is in the single injection that successfully reduced the amount of harmful brain proteins."
From a pharmacoeconomic perspective, a one-time gene-therapy-based approach could potentially reduce the long-term costs of Alzheimer’s care, provided the initial treatment is priced within reach of national health systems. Furthermore, by localized the "drug production" to the astrocytes within the brain, the therapy avoids the systemic side effects often associated with high-dose intravenous medications.
Safety Considerations and the Path to Human Trials
While the results in mice are promising, the transition to human clinical trials requires rigorous optimization. One of the primary concerns with any CAR-based therapy is "off-target" effects—the possibility that the engineered cells might attack healthy tissue or interfere with the essential functions of normal astrocytes. Astrocytes play a critical role in recycling neurotransmitters like glutamate; if the CAR modification disrupts this process, it could lead to excitotoxicity or seizures.
Dr. Colonna emphasized that the team is currently working to refine the precision of the CAR homing device. "Although more work needs to be done to optimize the approach and address potential side effects, these results open up an exciting new opportunity," he stated. Future research will likely focus on "kill switches" or inducible promoters—genetic tools that would allow doctors to turn the CAR-astrocyte activity "on" or "off" depending on the patient’s needs or the presence of adverse reactions.
Broader Impact: A Platform for Neurodegenerative Disease
The success of CAR-astrocytes in targeting amyloid beta suggests that this technology could be a "platform" for treating a wide variety of central nervous system disorders. By simply changing the target of the CAR homing device, researchers could potentially program astrocytes to clear other toxic proteins, such as:
- Tau Protein: The secondary hallmark of Alzheimer’s, which is more closely linked to actual cognitive decline than amyloid.
- Alpha-synuclein: The protein responsible for Lewy bodies in Parkinson’s disease and Lewy Body Dementia.
- TDP-43: A protein associated with Amyotrophic Lateral Sclerosis (ALS) and Frontotemporal Dementia.
Furthermore, the researchers identified a potential application in neuro-oncology. Glioblastomas and other brain tumors are notoriously difficult to treat because the blood-brain barrier prevents many chemotherapies from reaching the site. By engineering astrocytes to recognize tumor-specific markers, scientists could turn the brain’s own environment into a hostile territory for cancer cells, potentially offering a new line of defense against some of the most aggressive forms of brain cancer.
Chronology of Progress in Alzheimer’s Research
The Washington University study represents the latest milestone in a timeline of accelerating discovery:
- 1906: Dr. Alois Alzheimer first describes the plaques and tangles in the brain of a deceased patient.
- 1984: The amyloid beta protein is identified and sequenced.
- 2000s: The first attempts at amyloid vaccines (AN-1792) are tested but halted due to brain inflammation in human subjects.
- 2021-2023: The FDA grants accelerated and full approval to the first amyloid-clearing monoclonal antibodies.
- 2024: The Washington University team publishes the first successful use of CAR-astrocytes to clear plaques in vivo, signaling a shift toward cellular and gene-based therapies.
Conclusion
The findings published in Science mark a paradigm shift in how the medical community approaches the "unsolvable" problem of neurodegeneration. By moving away from the "outside-in" approach of intravenous antibodies and toward an "inside-out" strategy of reprogramming native brain cells, the Washington University researchers have provided a blueprint for the next generation of Alzheimer’s care. While human applications remain years away, the prospect of a single-injection therapy that can both prevent and reverse the accumulation of toxic proteins offers a new beacon of hope for the millions of families affected by Alzheimer’s disease worldwide. The ability to harness the brain’s own cells to maintain its health could eventually transform Alzheimer’s from a terminal diagnosis into a manageable, or even preventable, condition.














Leave a Reply