The LKS Faculty of Medicine at the University of Hong Kong (HKUMed) has announced a landmark discovery in the field of musculoskeletal health, identifying a specific biological mechanism that decodes how physical movement translates into bone density. Led by researchers from the Department of Medicine at the School of Clinical Medicine, the study has pinpointed a protein known as Piezo1 which serves as the body’s internal "exercise sensor." This protein allows bone marrow cells to detect mechanical strain and respond by generating new bone tissue rather than fat. The findings, recently published in the prestigious international journal Signal Transduction and Targeted Therapy, provide a potential molecular roadmap for developing "exercise mimetics"—pharmacological treatments that could replicate the skeletal benefits of physical activity for individuals who are physically unable to exercise.
The Global Crisis of Osteoporosis and Age-Related Bone Loss
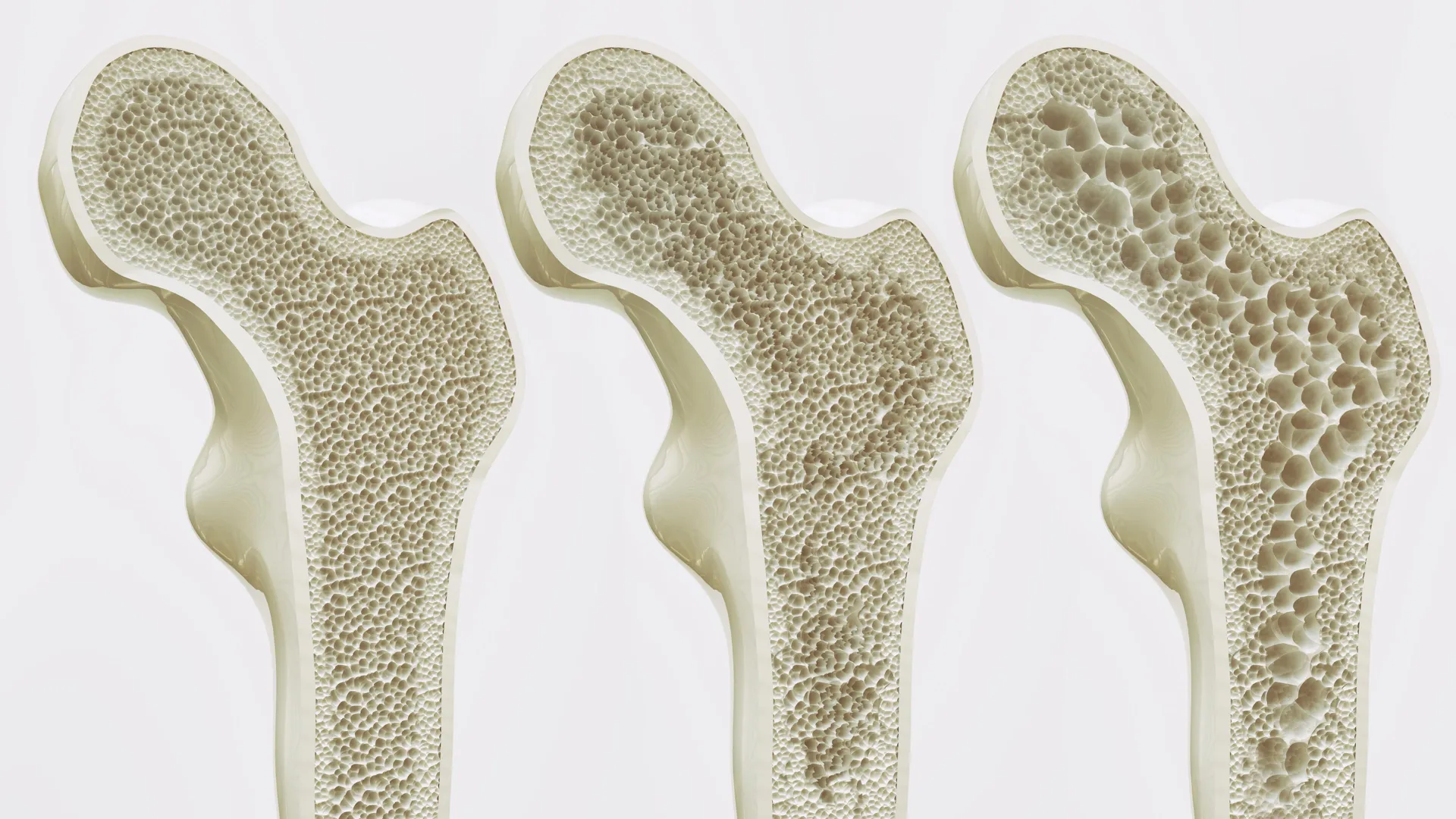
Osteoporosis is frequently referred to by medical professionals as a "silent epidemic" because bone loss occurs without symptoms until a fracture takes place. As the global population ages, the prevalence of this condition is reaching critical levels. According to data from the World Health Organization (WHO), osteoporosis currently affects an estimated 200 million people worldwide. The clinical implications are severe: approximately one in three women and one in five men over the age of 50 will suffer an osteoporotic fracture in their lifetime.
In the specific context of Hong Kong, the challenge is amplified by one of the world’s fastest-aging populations. Statistics indicate that osteoporosis affects roughly 45% of women and 13% of men aged 65 and older in the region. These fractures, particularly of the hip and spine, are associated with high mortality rates, chronic pain, and a significant loss of independence. For many elderly patients, a single fall can lead to a permanent transition into long-term nursing care, placing an immense socioeconomic burden on the healthcare infrastructure.
The fundamental biological problem underlying this crisis is the gradual loss of bone mineral density (BMD). In a healthy, young body, bone is a dynamic tissue that constantly remodels itself. However, as humans age, the balance of this remodeling process shifts. The HKUMed study addresses the root cause of this shift, focusing on why the body stops "choosing" to build bone and starts "choosing" to store fat within the skeletal framework.
The Biological Switch: Bone versus Fat in the Marrow
To understand the HKUMed discovery, one must look at the behavior of mesenchymal stem cells (MSCs) located within the bone marrow. These versatile cells are the progenitors for several different types of tissue; they have the potential to differentiate into either osteoblasts (cells that form bone) or adipocytes (cells that store fat).
Under normal conditions in active individuals, physical forces—such as the impact of walking, running, or weight-bearing exercise—signal these stem cells to become bone-forming osteoblasts. This keeps the skeleton strong and resilient. However, the aging process and prolonged inactivity disrupt this signaling pathway. In sedentary or elderly populations, MSCs increasingly differentiate into adipocytes. This leads to an accumulation of "marrow fat," which does not contribute to structural integrity and actually secretes factors that further weaken the surrounding bone matrix.
"Osteoporosis and age-related bone loss affect millions worldwide, often leaving elderly and bedridden patients vulnerable to fractures and loss of independence," explained Professor Xu Aimin, Director of the State Key Laboratory of Pharmaceutical Biotechnology and Chair Professor in the Department of Medicine at HKUMed. Professor Xu, who led the research, noted that while exercise is the most effective natural defense against this process, it is a "catch-22" for many patients. Those who need the benefits of exercise the most—the frail, the injured, and the chronically ill—are often the least capable of performing it.
Identifying Piezo1: The Molecular Exercise Sensor
The breakthrough achieved by the HKUMed team involves the identification of the Piezo1 protein as the primary mechanosensor in bone marrow mesenchymal stem cells. Through a series of sophisticated experiments involving both mouse models and human cell cultures, the researchers demonstrated that Piezo1 sits on the surface of these stem cells, acting like a microscopic "pressure switch."
When a person moves, the mechanical stress exerted on the bones activates Piezo1. This activation triggers a cascade of intracellular signals that promote the expression of bone-forming genes while simultaneously suppressing the pathways that lead to fat formation. The research team utilized advanced genetic engineering to create mouse models where Piezo1 was specifically "knocked out" or removed from the MSCs. The results were definitive: even when these mice remained active, their bones weakened rapidly. Without the Piezo1 "sensor," their bodies could no longer "feel" the exercise, causing their stem cells to default into fat production.
Furthermore, the study revealed that the absence of Piezo1 activity triggers a secondary harmful effect: the release of inflammatory signals, specifically Ccl2 and lipocalin-2. These pro-inflammatory markers create a toxic microenvironment in the bone marrow, further accelerating bone loss and hindering the repair of existing bone tissue. By blocking these specific inflammatory signals in their experimental models, the researchers were able to partially restore bone health, suggesting a multi-layered approach to future treatments.
The Promise of Exercise Mimetics
The most significant implication of this research is the potential to develop "exercise mimetics." This term refers to a new class of drugs designed to trigger the same physiological responses as physical activity without the requirement for actual movement.
"We have essentially decoded how the body converts movement into stronger bones," said Professor Xu Aimin. "By activating the Piezo1 pathway, we can mimic the benefits of exercise, effectively tricking the body into thinking it is exercising, even in the absence of movement."
Dr. Wang Baile, Research Assistant Professor at HKUMed and co-leader of the study, emphasized that this is not about replacing exercise for the healthy, but providing a lifeline for the vulnerable. "This discovery is especially meaningful for older individuals and patients who cannot exercise due to frailty, injury, or chronic illness. Our findings open the door to developing drugs that chemically activate the Piezo1 pathway to help maintain bone mass and support independence."
The concept of exercise mimetics is gaining traction in various fields of medicine, including metabolic health and muscle atrophy. However, the HKUMed study is one of the first to provide a clear, actionable target for bone tissue. For a bedridden patient recovering from surgery or an elderly individual with severe osteoarthritis, a Piezo1 activator could potentially prevent the rapid bone degradation that typically occurs during periods of immobility.
International Collaboration and Research Support
The study was a highly collaborative effort, reflecting the global nature of modern medical breakthroughs. The HKUMed team worked closely with Professor Eric Honoré, a Team Leader at the Institute of Molecular and Cellular Pharmacology of the French National Centre for Scientific Research (CNRS). Professor Honoré, an expert in mechanobiology, highlighted the broader scientific impact of the discovery.
"This offers a promising strategy beyond traditional physical therapy," Professor Honoré stated. "In the future, we could potentially provide the biological benefits of exercise through targeted treatments, thereby slowing bone loss in vulnerable groups such as bedridden patients or those with limited mobility, and substantially reducing their risk of fractures."
The research was supported by a diverse array of funding bodies, including the Research Grants Council of Hong Kong, the Health and Medical Research Fund under the Health Bureau of the HKSAR, and the National Natural Science Foundation of China. International support came from the Human Frontier Science Program and various French medical research foundations, underscoring the high level of interest in solving the global problem of age-related bone deterioration.
Timeline and Future Clinical Outlook
The identification of Piezo1 marks the conclusion of a rigorous basic research phase and the beginning of a complex translational phase. The timeline for moving from laboratory discovery to a clinical drug typically spans several years, involving multiple stages:
- Compound Discovery: Identifying or synthesizing chemical compounds that can safely and effectively activate Piezo1 in humans.
- Pre-clinical Testing: Extensive testing in animal models to ensure that systemic activation of Piezo1 does not have adverse effects on other organs, such as the heart or lungs, where mechanical sensing is also vital.
- Phase I Clinical Trials: Testing the safety and dosage of "exercise mimetic" candidates in a small group of human volunteers.
- Phase II and III Trials: Evaluating the efficacy of the drug in preventing bone loss and reducing fracture rates in large populations of at-risk patients.
While the path to a commercial medication is long, the HKUMed team is already moving forward with the next steps. Their current focus is on refining the delivery mechanisms for potential therapies to ensure they specifically target the bone marrow environment, thereby minimizing potential side effects.
Analysis of Broader Societal and Healthcare Implications
The successful development of a Piezo1-based therapy would have profound implications for public health policy and healthcare economics. Currently, the cost of treating osteoporotic fractures—including surgery, hospitalization, and long-term rehabilitation—runs into the billions of dollars annually. In Hong Kong alone, the direct medical cost of hip fractures is projected to rise significantly as the "silver tsunami" of aging residents peaks over the next two decades.
By shifting the focus from reactive treatment (fixing a broken bone) to proactive molecular maintenance (preventing the bone-to-fat shift), healthcare systems could save significant resources. Moreover, the benefits extend beyond bone health. Because bone marrow health is intrinsically linked to the production of blood cells and the regulation of the immune system, maintaining a healthy marrow environment via Piezo1 activation could have systemic benefits for the aging population.
Furthermore, this research has potential applications in the field of aerospace medicine. Astronauts in microgravity environments suffer from rapid bone loss because their skeletons are no longer subjected to the mechanical load of Earth’s gravity. A pharmacological activator of the Piezo1 "exercise sensor" could be a vital tool for maintaining the health of crews on long-duration space missions, such as a journey to Mars.
Conclusion
The discovery of Piezo1 as the bone’s primary exercise sensor by the HKUMed research team represents a major leap forward in our understanding of mechanobiology and geriatric medicine. By bridging the gap between physical movement and molecular signaling, Professor Xu Aimin and his colleagues have provided a new target for the treatment of osteoporosis that bypasses the need for physical exertion. As the research moves toward clinical application, it offers a new sense of hope for millions of people worldwide who face the prospect of bone loss and the resulting loss of mobility. The vision of "exercise in a pill" for bone health is no longer a matter of science fiction, but a tangible goal that could redefine the aging process and enhance the quality of life for the elderly and infirm.












Leave a Reply