Researchers at Johns Hopkins Medicine have announced that a newly funded study by the National Institutes of Health (NIH) is providing critical insights into a novel approach for treating Alzheimer’s disease. The research centers on a specific protein in the brain responsible for producing a small but biologically essential gas: hydrogen sulfide. While hydrogen sulfide is most commonly recognized by the public for its distinctive "rotten egg" odor, this study highlights its indispensable role as a signaling molecule, or gasotransmitter, that is vital for maintaining cognitive function and protecting against neurodegeneration.
The protein at the heart of this discovery is Cystathionine γ-lyase, commonly abbreviated as CSE. According to the study’s lead author, Bindu Paul, M.S., Ph.D., an associate professor of pharmacology, psychiatry, and neuroscience at the Johns Hopkins University School of Medicine, CSE appears to be a primary regulator of how memories are formed and retained. The findings, recently published in the Proceedings of the National Academy of Sciences (PNAS), suggest that disruptions in CSE activity may be a precursor to the cellular damage and cognitive decline seen in Alzheimer’s patients.
The Biological Significance of Hydrogen Sulfide
For decades, hydrogen sulfide ($H_2S$) was viewed primarily as a toxic environmental pollutant. However, in recent years, the scientific community has begun to recognize it as a member of a unique class of signaling molecules known as gasotransmitters, alongside nitric oxide and carbon monoxide. In the human brain, $H_2S$ is produced in extremely small quantities, where it acts as a protective agent for neurons.
Previous research has demonstrated that hydrogen sulfide can mitigate oxidative stress and prevent cellular apoptosis (programmed cell death). However, the challenge for medical researchers lies in the gas’s inherent toxicity. In large concentrations, hydrogen sulfide is lethal, meaning it cannot be administered directly as a traditional drug. The Johns Hopkins study instead focuses on the "upstream" mechanism—the CSE protein—as a way to safely maintain the delicate, naturally occurring levels of the gas within the brain’s neural pathways.
The researchers found that when the CSE enzyme is absent or malfunctioning, the brain loses its primary defense against oxidative stress. This leads to a cascade of cellular failures, including DNA damage and the weakening of the blood-brain barrier, all of which are hallmarks of Alzheimer’s disease and other forms of dementia.
A Chronology of Discovery: From Blood Pressure to Neurodegeneration
The current findings are the result of nearly two decades of research into the CSE protein, much of it led by Solomon Snyder, M.D., D.Sc., D.Phil., a professor emeritus at Johns Hopkins and a pioneer in the field of neuroscience. The timeline of this research illustrates a steady progression from general physiology to specific neurodegenerative pathologies:
- 2008: Researchers first developed a strain of genetically engineered mice that lacked the CSE protein. Initial studies focused on the cardiovascular system, linking CSE to the regulation of blood vessel function and blood pressure.
- 2014: Snyder’s team expanded their scope to the brain, reporting in Nature that CSE played a supportive role in brain health for models of Huntington’s disease. They observed that the loss of CSE accelerated motor decline and brain atrophy.
- 2021: The group turned its attention to Alzheimer’s disease, discovering that CSE levels were significantly depleted in the brains of mice with Alzheimer’s-like symptoms. Preliminary tests showed that micro-injections of hydrogen sulfide could temporarily restore some brain function.
- 2024: The latest research isolates the role of CSE itself, moving beyond previous models that included multiple genetic mutations. By studying mice where the only variable was the absence of CSE, the team proved that the loss of this single protein is sufficient to trigger Alzheimer’s-like cognitive decline.
Dr. Snyder, who retired from the Johns Hopkins faculty in 2023 but remains a co-corresponding author on the study, emphasized the significance of these findings. "This most recent work indicates that CSE alone is a major player in cognitive function and could provide a new avenue for treatment pathways in Alzheimer’s disease," Snyder stated.
Testing Cognitive Decline: The Barnes Maze Experiments
To quantify the impact of CSE on memory, the research team employed the Barnes maze, a standard tool in behavioral neuroscience used to measure spatial learning and memory in rodents. The maze consists of a circular platform with multiple holes around the perimeter, only one of which leads to a dark escape tunnel. Because mice are naturally averse to bright lights and open spaces, they are motivated to find the tunnel.
The study compared "wild-type" (normal) mice with the genetically engineered CSE-deficient mice. At two months of age, both groups performed equally well, locating the escape route in roughly three minutes. However, as the mice aged, a stark divergence occurred. By the six-month mark, the normal mice continued to navigate the maze efficiently, while the CSE-deficient mice struggled significantly, often failing to find the escape route entirely.
"The decline in spatial memory indicates a progressive onset of neurodegenerative disease that we can attribute to CSE loss," said Suwarna Chakraborty, a researcher in Paul’s lab and the study’s first author. This age-dependent decline mimics the progression of Alzheimer’s in humans, which typically manifests in later stages of life after years of accumulated cellular stress.
Cellular and Structural Damage in the Brain
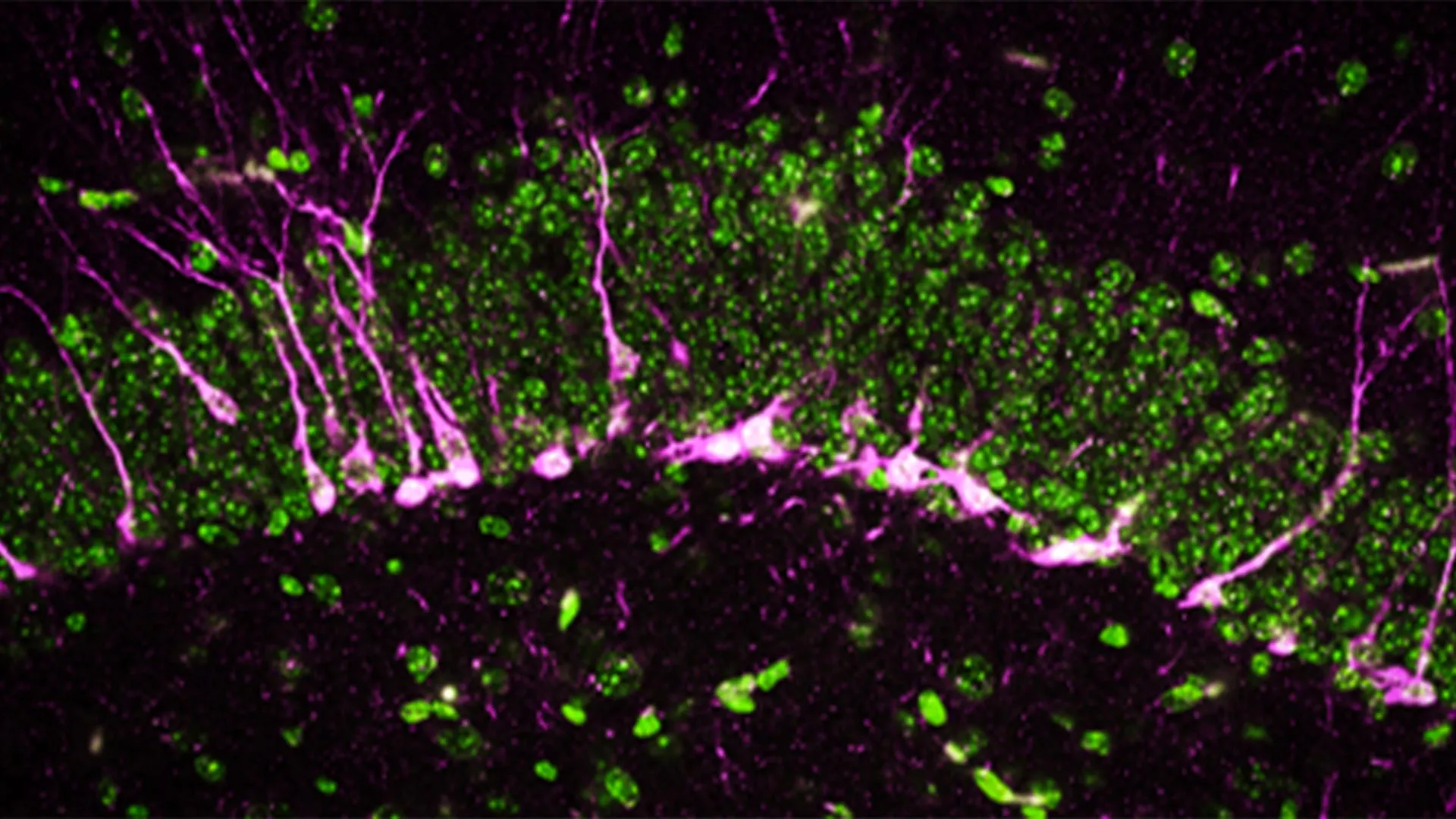
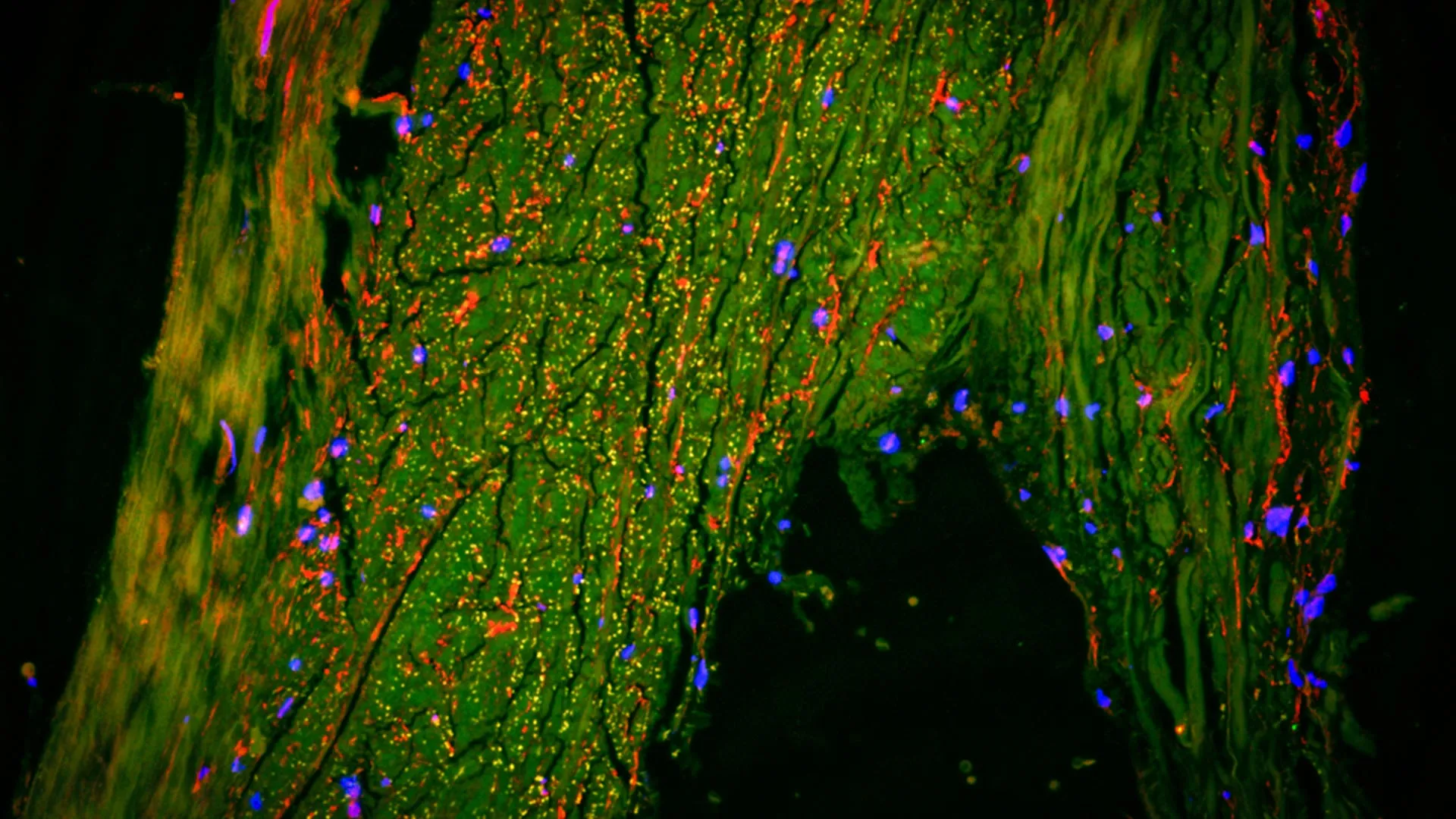
Beyond behavioral testing, the researchers utilized high-powered electron microscopy and biochemical analysis to examine the physical state of the mice’s brains. They focused on the hippocampus, the region of the brain primarily responsible for memory formation and spatial navigation.
The findings revealed several critical failures at the cellular level:
- Impaired Neurogenesis: The team found that proteins essential for the creation of new neurons (neurogenesis) were either reduced or entirely absent in mice lacking CSE.
- Blood-Brain Barrier Integrity: Electron microscope images showed large breaks in the blood vessels within the brain. A compromised blood-brain barrier allows toxins to enter the brain tissue, further accelerating the progression of Alzheimer’s.
- Neuronal Migration Issues: Newly formed neurons in CSE-deficient mice were unable to reach their intended destinations in the hippocampus, effectively stalling the brain’s ability to repair itself or form new memory circuits.
Co-first author Sunil Jamuna Tripathi noted that the mice lacking CSE were "compromised at multiple levels," creating a biological profile that closely mirrors the pathology observed in human Alzheimer’s patients.
Broader Implications for Alzheimer’s Treatment
The search for effective Alzheimer’s treatments has been fraught with challenges. For decades, the pharmaceutical industry focused almost exclusively on the "amyloid hypothesis"—the idea that clearing amyloid-beta plaques from the brain would stop the disease. However, many drugs targeting amyloid have failed in clinical trials, or offered only modest benefits, leading researchers to seek alternative targets.
The Johns Hopkins study suggests that targeting the CSE/hydrogen sulfide pathway could represent a "metabolic" approach to treatment. Rather than just clearing out protein aggregates, this approach aims to bolster the brain’s internal defense mechanisms and maintain the integrity of its vascular system.
According to the U.S. Centers for Disease Control and Prevention (CDC), more than 6 million Americans are currently living with Alzheimer’s, a number projected to nearly triple by 2050. The economic and social burden of the disease is immense, with annual costs in the hundreds of billions of dollars. If therapies can be developed to boost CSE activity or safely mimic the effects of hydrogen sulfide, it could offer a way to slow or even halt the disease’s progression before irreversible damage occurs.
Collaborative Research and Future Directions
The study was a massive collaborative effort, involving researchers from Johns Hopkins University, Case Western Reserve University, the Leibniz Institute for Analytical Sciences in Germany, Hollings Cancer Center, and West Virginia University School of Medicine.
Funding for the research was provided by a wide array of institutions, including the National Institutes of Health, the Department of Defense, and the American Heart Association. The breadth of funding reflects the cross-disciplinary importance of CSE research, which touches on everything from cardiovascular health to neuropsychiatry.
The next phase of research will likely involve screening for small-molecule compounds that can enhance the activity of the CSE protein. Researchers are also looking into the role of diet and lifestyle, as certain sulfur-containing amino acids are precursors to $H_2S$ production in the body. While human clinical trials are still in the distance, the isolation of CSE as a primary driver of cognitive health provides a clear and promising roadmap for future drug development.
As the scientific community continues to move toward a more multifaceted understanding of Alzheimer’s, the "rotten egg gas" once dismissed as a mere byproduct of decay may prove to be one of the most vital components of the living brain’s survival kit.









Leave a Reply