In a landmark study published in the journal Nature on April 15, 2026, a multi-institutional research team led by the Dalian Institute of Chemical Physics (DICP) of the Chinese Academy of Sciences (CAS) announced a breakthrough that fundamentally alters the scientific understanding of heterogeneous catalysis. For the first time in the history of material science, researchers have directly tracked the movement of oxygen atoms within the interior, or "bulk," of a catalyst support. This discovery, centered on ruthenium supported by rutile titanium dioxide (Ru/r-TiO2), challenges the long-held dogma that catalytic spillover is a phenomenon restricted almost exclusively to the two-dimensional surfaces of materials.
The collaborative effort, headed by Professor Tao Zhang and Professor Yanqiang Huang at DICP, alongside Professor Wei Liu of DICP and Professor Yanggang Wang of the Southern University of Science and Technology, utilized advanced environmental transmission electron microscopy (ETEM) to witness "bulk oxygen spillover." This observation reveals that the inner layers of a catalyst support, previously dismissed as inert or "useless" in the context of active reactions, can serve as a reservoir and transport channel for reactive species, potentially doubling or tripling the efficiency of industrial chemical processes.
The Evolution of Catalytic Theory: From Surface to Bulk
To appreciate the magnitude of this discovery, one must understand the traditional framework of heterogeneous catalysis. For over a century, the field has operated on the principle that chemical reactions occur at the "active sites" located on the surface of a solid catalyst. Because reactants—such as hydrogen, carbon monoxide, or oxygen—cannot penetrate the dense crystalline structure of most solid supports, the interior of the catalyst particle was seen merely as a structural scaffold.
"Spillover" is a critical sub-process in these reactions. It occurs when an atom, such as oxygen or hydrogen, is adsorbed and dissociated on a metal particle (like ruthenium or platinum) and then migrates onto the surface of the supporting material (like titanium dioxide). This migration expands the reactive zone beyond the metal itself. Until now, the scientific consensus was that this migration happened strictly along the surface "skin" of the support.
The findings presented by the DICP team prove that this migration is not just a surface-level event. By demonstrating that oxygen atoms can dive into the bulk of the rutile titanium dioxide and emerge at the metal interface from layers deep within the crystal, the researchers have effectively opened a third dimension for catalytic engineering.
Experimental Design and the Choice of Rutile TiO2
The selection of ruthenium (Ru) supported on rutile titanium dioxide (r-TiO2) was a strategic decision based on the unique thermodynamic properties of these materials. Titanium dioxide is one of the most studied supports in catalysis due to its "reducible" nature—it can easily lose or gain oxygen atoms, changing its oxidation state in response to the environment.
Rutile is one of the primary crystalline forms of TiO2. The researchers focused on this specific phase because its well-defined lattice structure allowed for precise tracking of atomic movement. Using Environmental Transmission Electron Microscopy (ETEM), the team created a controlled "laboratory under the lens," where they could introduce gases and vary temperatures while maintaining atomic-scale resolution.
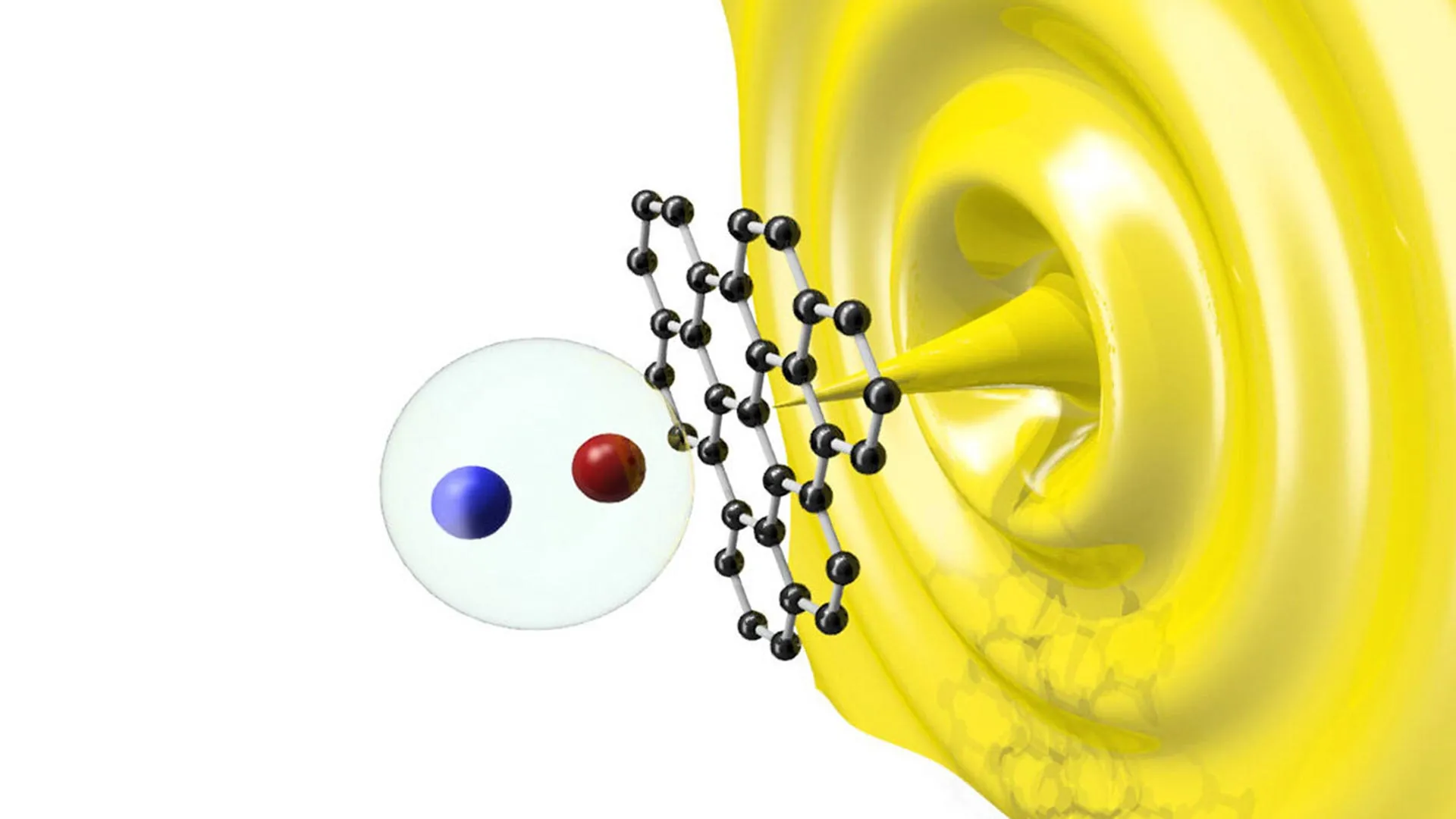
During the observation, the team focused on the Ru/r-TiO2 interface. They noticed that the oxygen chemical potential—the force that drives atoms to move from areas of high concentration to low concentration—created a "suction" effect. This gradient pulled oxygen atoms from three to five atomic layers beneath the surface of the TiO2, moving them through the bulk and toward the ruthenium metal particles.
The "Atomic Guard" and the Metal-Support Interface
One of the most significant takeaways from the study is the role of the metal-support interface. Professor Wei Liu described this interface as an "atomic scale guard." The research showed that the interface acts as a gatekeeper, regulating the flow of oxygen between the bulk of the support and the metal active site.
This discovery expands upon the theory of Strong Metal-Support Interaction (SMSI), a concept first introduced by S. J. Tauster in 1978. For nearly 50 years, SMSI has described how metal particles become encapsulated by a thin layer of the oxide support under certain conditions, which can drastically change the catalyst’s reactivity. However, Tauster’s theory focused on the surface encapsulation. The new research by the DICP team suggests that the interaction goes much deeper, involving a dynamic exchange of mass between the interior of the support and the metal surface.
The researchers identified a specific channel within the TiO2 lattice that facilitates this bulk spillover. When the chemical potential is correctly tuned, the interior of the catalyst becomes an active participant in the mass transfer process. This means that the "dead volume" of industrial catalysts can now be recruited to participate in the reaction, providing a steady supply of reactive atoms to the metal sites even when the surface is crowded or blocked.
Technical Data and Key Observations
The team’s data, derived from real-time in situ imaging, provided several quantifiable insights:
- Depth of Participation: Oxygen atoms were observed migrating from depths of 0.9 to 1.5 nanometers (approximately 3-5 atomic layers) below the surface.
- Driving Force: The movement was confirmed to be driven by oxygen chemical potential gradients, which can be manipulated by adjusting the temperature and the partial pressure of surrounding gases.
- Structural Integrity: Unlike surface-only spillover, which can sometimes lead to the degradation of the catalyst surface, bulk spillover appeared to utilize the stable crystalline lattice of the rutile structure, suggesting higher long-term stability for catalysts designed with this pathway in mind.
Professor Yanqiang Huang emphasized the importance of this mechanism: "This unique oxygen spillover enables the bulk of a catalyst, which is otherwise inaccessible to reactants, to contribute to mass transfer. It underscores the critical importance of interface engineering."
Global Implications for Industry and Green Energy
The ability to utilize the bulk of a catalyst has profound implications for a wide range of industrial applications. Catalysis is a cornerstone of the global economy, involved in the production of 90% of all commercially produced chemical products, including fertilizers, plastics, and fuels.
1. Hydrogen Energy and Fuel Cells:
The findings could lead to more efficient catalysts for the Oxygen Evolution Reaction (OER) and Oxygen Reduction Reaction (ORR), which are central to hydrogen fuel cells and water electrolysis. By tapping into the bulk oxygen of supports, scientists can design electrodes that are more resistant to "poisoning" and have higher turnover rates.
2. Carbon Capture and Utilization:
Converting CO2 into useful chemicals like methanol requires highly efficient oxygen management within the catalyst. Bulk spillover provides a new mechanism to move oxygen away from the reaction site, preventing the catalyst from becoming "clogged" and allowing for continuous operation at lower temperatures.
3. Emission Control:
Automotive catalytic converters rely on the storage and release of oxygen to neutralize toxic exhaust gases. Understanding bulk spillover allows engineers to design supports that can store significantly more oxygen in a smaller volume, leading to smaller and more effective converters.
Future Directions: From Discovery to Application
The research team is already looking toward the next phase of their work. Professor Tao Zhang, a leading figure in Chinese catalysis research, noted that the goal is to move from observation to "rational design."
"Taking this excellent opportunity, we can improve the architecture of catalysis from two-dimensional surface reactions to the three-dimensional ‘surface-interface-bulk’ synergy," Professor Zhang stated. "The next goal is to develop practical catalysts that utilize the bulk to directly contribute to chemical reactions."
This shift toward "3D Catalysis" will likely involve the development of new synthesis techniques that create "oxygen highways" within the bulk of support materials. By doping the interior of oxides or creating specific lattice defects, scientists may be able to pre-program the path that atoms take through the material.
Conclusion: A New Era of Interfacial Engineering
The study by the DICP and its partners represents a paradigm shift in the chemical sciences. By proving that the interior of a catalyst is not merely a passive observer but a dynamic participant, the team has provided a new set of tools for tackling some of the world’s most pressing energy and environmental challenges.
As the scientific community digests these findings, the focus will undoubtedly turn to other metal-oxide combinations. If bulk spillover is possible in titanium dioxide, it may also occur in other common supports like alumina, zirconia, or ceria. The "atomic scale guard" has been identified; the next challenge is learning how to command it to open the gates for a new generation of high-performance materials.
The work of Zhang, Huang, Liu, and Wang stands as a testament to the power of modern imaging technology and the persistence of researchers in questioning long-standing scientific "truths." In the quest for more sustainable and efficient chemistry, the path forward apparently lies not just on the surface, but deep within the heart of the atom.














Leave a Reply