The landscape of clinical trials is undergoing a significant transformation, driven by increasingly complex protocols and the escalating demands placed upon research sites. A critical bottleneck in this evolution is the protracted and often error-prone process of manual data entry, a practice that has become a substantial impediment to efficiency and timely insight generation. Paradigm Health’s innovative eSource Casebook solution is poised to address these pressing issues by integrating Electronic Health Records (EHRs) directly into the clinical trial workflow, thereby streamlining data capture, reducing site burden, and accelerating the path to critical research outcomes.
The traditional model of clinical trial data management relies heavily on manual transcription from patient records into Electronic Case Report Forms (eCRFs). This process is not only time-consuming but also inherently susceptible to human error. Industry data reveals a stark reality: it can take an average of eight days from a patient’s visit to the completion of data entry. This delay has a cascading effect, slowing down trial progress, inflating operational costs, and potentially delaying the availability of life-saving therapies to patients. Over the past decade, the sheer volume of data entry required has reportedly tripled, placing an unsustainable strain on already stretched research site resources.
While the concept of "eSource" – capturing data directly at the source of its creation – has been recognized for its potential to alleviate these burdens, its widespread adoption has been hampered by fragmented systems and a lack of seamless integration. Sponsors and research sites alike have struggled to leverage the full benefits of eSource due to the existence of data silos, where information remains locked within disparate platforms, hindering efficient data flow and cross-validation. This situation creates a pressing need for purpose-built solutions that can effectively bridge these gaps, enhance data quality, reduce the workload on research personnel, and foster greater collaboration without introducing additional operational overhead or jeopardizing trial timelines.
Reducing the Data Capture Burden to Boost Efficiency and Research Capacity
Paradigm Health’s eSource Casebook emerges as a direct response to this industry imperative. This innovative EHR-integrated application is designed to revolutionize how clinical trial data is collected and managed. By intelligently extracting trial-relevant information from both structured and unstructured data within EHRs, the eSource Casebook automates the auto-population of eCRFs. Furthermore, it facilitates the seamless transmission of this data to sponsor Electronic Data Capture (EDC) systems.
At its core, the eSource Casebook functions as an extension of the sponsor’s EDC, but with a crucial site-facing interface. It consolidates the entire data entry process into a single, intuitive application, which is designed to integrate effortlessly into the daily workflows of research teams. This eliminates the need for redundant data entry and the constant switching between multiple systems, a common source of frustration and inefficiency for clinical research coordinators (CRCs) and other site personnel.
The application’s intuitive import workflow, complete with citations for rapid verification, empowers CRCs and research teams to enter data significantly faster. This substantial reduction in time spent on data management translates directly into enhanced research capacity. Sites can therefore dedicate more resources to patient care and recruitment, take on a larger number of studies with existing personnel, and ultimately ensure a smoother, more patient-centric trial experience. This increased efficiency can also lead to a reduction in protocol deviations and data queries, further streamlining the overall trial process.

Efficient and Accurate Data Management: A Foundation for Reliable Research
The efficacy of clinical trials hinges on the accuracy and integrity of the data collected. The eSource Casebook directly tackles this challenge by minimizing manual intervention, a primary source of data errors. By directly interfacing with EHRs, the system leverages standardized data formats and robust extraction capabilities to ensure that data entered into eCRFs is as accurate as possible.
The ability to extract data from both structured fields (such as lab results, vital signs, and medication lists) and unstructured sources (like physician notes and narrative reports) is a key differentiator. This comprehensive data capture ensures that all relevant patient information is considered, leading to a more complete and nuanced understanding of treatment effects and patient responses. The use of advanced technologies, such as Natural Language Processing (NLP) powered by Large Language Models (LLMs), allows the eSource Casebook to interpret and extract valuable insights from free-text clinical notes, which often contain critical information that might otherwise be missed.
The auto-population of eCRFs, pre-filled with verified EHR data, significantly reduces the cognitive load on research staff. Instead of transcribing information from scratch, CRCs can quickly review and confirm pre-populated fields, ensuring accuracy and adherence to protocol requirements. This streamlined approach not only accelerates data entry but also minimizes the risk of transcription errors, data duplication, and omissions.
Empowered and Productive Research Teams: Reclaiming Time and Focus
The impact of the eSource Casebook extends beyond mere data processing; it fundamentally aims to empower research teams and enhance their productivity. By automating many of the most time-consuming and repetitive tasks associated with data management, the solution frees up valuable time for CRCs and investigators to focus on what truly matters: patient care and scientific inquiry.
The reduction in manual data entry directly alleviates the administrative burden that often leads to burnout among research staff. When data collection is more efficient and less prone to errors, the need for extensive data reconciliation and query resolution diminishes. This not only saves time but also reduces stress and improves job satisfaction for clinical research professionals.
Moreover, by providing a single, integrated platform for data management, the eSource Casebook fosters a more collaborative and efficient working environment. Research teams can access and manage patient data with greater ease, leading to better communication and coordination among team members. This can be particularly beneficial in multi-site trials where consistent data management practices are crucial for maintaining data integrity across different locations. The enhanced efficiency can also enable sites to manage more complex studies or increase their overall research capacity without necessarily increasing headcount, a critical consideration in today’s competitive research environment.
Scalable and Dynamic Infrastructure: Adapting to Evolving Needs
The complexities of clinical trials are not static; they evolve with advancements in therapeutic areas and regulatory requirements. The eSource Casebook is built on a scalable and dynamic infrastructure designed to adapt to these changing needs. Its modular architecture allows for easy integration with various EHR systems and sponsor EDC platforms, ensuring compatibility and future-proofing the solution.

The ability to connect to EHRs via standardized protocols like FHIR (Fast Healthcare Interoperability Resources) is crucial for ensuring interoperability and data exchange. FHIR is an open standard that enables different health information systems to exchange data in a consistent and efficient manner. By adhering to such standards, the eSource Casebook can seamlessly integrate with a wide range of EHR vendors, making it a versatile solution for diverse research settings.
Furthermore, the application’s reliance on advanced technologies like LLMs allows it to remain at the forefront of data extraction and analysis. As NLP capabilities continue to advance, the eSource Casebook can be updated to leverage these improvements, further enhancing its ability to extract meaningful data from complex clinical narratives. This dynamic infrastructure ensures that the solution remains relevant and effective as the clinical research landscape continues to evolve.
Connecting to the EHR and Integrating into Existing Workflows
The seamless integration of the eSource Casebook into existing clinical workflows is a cornerstone of its design philosophy. The application is not intended to be an add-on that disrupts established practices but rather a tool that enhances and streamlines them. This is achieved through several key features.
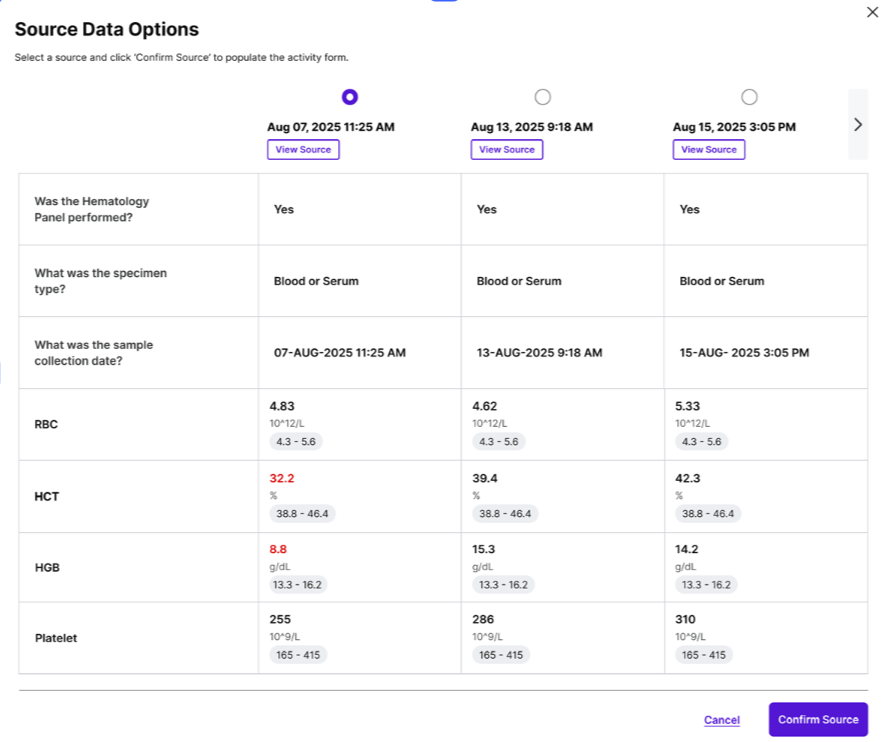
The direct import of structured EHR data via FHIR means that essential patient information can be pulled into the system with minimal manual effort. This eliminates the need for CRCs to manually search for and enter data points such as demographics, diagnoses, medications, allergies, and laboratory results. The system intelligently maps and validates this data against the specific requirements of the clinical trial protocol.
The power of LLMs is harnessed to tackle the challenge of unstructured data. Clinical notes, often containing rich qualitative information, can be processed by the eSource Casebook to extract key findings, adverse events, or other relevant details. This transforms previously difficult-to-access information into structured data that can be readily used for eCRF population.
The user interface is designed to be highly intuitive, presenting EHR-like views that are pre-filtered for trial relevance. This allows research staff to quickly review and select the appropriate data without having to navigate complex EHR systems or switch between multiple applications. The system then auto-populates the eCRFs, presenting the data in a format that is easily understandable and directly translatable for submission to the sponsor’s EDC. This significantly reduces the mental overhead associated with data entry, allowing researchers to focus on the scientific integrity of the data rather than the mechanics of its entry.
The seamless submission of data to the sponsor EDC is the final step in this efficient process. The eSource Casebook ensures that data is formatted correctly and transmitted securely, minimizing the potential for delays or errors during the data transfer phase. This end-to-end integration creates a streamlined data management pathway, from the point of care to the sponsor’s central database.
Broader Implications for the Clinical Research Ecosystem
The widespread adoption of solutions like Paradigm Health’s eSource Casebook has the potential to catalyze significant positive changes across the entire clinical research ecosystem. For sponsors, this means faster trial timelines, reduced operational costs associated with data management, and higher quality data, leading to more robust and reliable trial results. This can accelerate the drug development process, bringing new therapies to patients more quickly.
For research sites, the benefits are equally profound. Reduced administrative burden, improved efficiency, and enhanced data quality can lead to greater profitability and sustainability. It can also make research participation more attractive to healthcare providers, thereby increasing the availability of clinical trial opportunities for patients.
For patients, the ultimate beneficiaries of these advancements, the impact is multifaceted. A smoother and more efficient trial process can lead to a better patient experience, with less time spent on administrative tasks and more focus on their health and well-being. Faster trial completion also means quicker access to potentially life-changing treatments.
The shift towards integrated eSource solutions represents a critical step in modernizing clinical trials. By leveraging technology to automate data capture, reduce manual errors, and streamline workflows, the industry can overcome long-standing challenges and accelerate the delivery of innovative medical solutions. Paradigm Health’s eSource Casebook stands as a testament to this progress, offering a comprehensive and effective solution for the evolving demands of clinical research.
Ready to accelerate data entry, reduce auditing costs, and ensure faster, more accurate data flow?
Contact Paradigm Health at paradigm.inc/solutions/conduct-esource-casebook to learn more about how the eSource Casebook can transform your clinical trial operations.
















Leave a Reply