The research, spearheaded by Professor Mingxin Huang, marks a pivotal moment in materials science. By utilizing a "sequential dual-passivation" strategy, the team has created a steel that does not merely resist corrosion through traditional means but evolves its own protective shield as electrical potential increases. This discovery, detailed in the journal Materials Today, could fundamentally alter the economics of renewable energy by making direct seawater electrolysis a commercially viable reality.
The Economic Imperative for Green Hydrogen
Green hydrogen is widely regarded as the "holy grail" of the clean energy transition. Unlike "grey hydrogen," which is derived from natural gas and releases significant carbon dioxide, green hydrogen is produced through electrolysis—using renewable electricity to split water molecules into hydrogen and oxygen. However, the scalability of this technology has long been hindered by two primary factors: the scarcity of freshwater and the prohibitive cost of electrolyzer hardware.
Seawater is an abundant and ideal feedstock for hydrogen production, particularly for coastal industrial hubs. Yet, the chemical composition of seawater presents a nightmare for engineering. Chloride ions are notoriously aggressive, causing rapid pitting and structural failure in conventional metals. To combat this, the industry has historically relied on titanium structural components coated with noble metals like platinum or gold. While effective, these materials are prohibitively expensive for the multi-gigawatt deployments required to meet global decarbonization targets.
According to data provided by the HKU research team, the structural components of a 10-megawatt Proton Exchange Membrane (PEM) electrolysis system can account for more than 50% of the total system cost. At the time of their report, a 10 MW system was estimated to cost approximately HK$17.8 million, with structural materials alone exceeding HK$9.4 million. The introduction of SS-H2 could reduce the cost of these structural materials by a factor of 40, representing a massive shift in the capital expenditure (CAPEX) required for green hydrogen infrastructure.
A Chronology of Innovation: The Super Steel Legacy
The development of SS-H2 is not an isolated event but the latest milestone in Professor Huang’s long-running "Super Steel" Project. This research program has a distinguished history of producing high-performance alloys that solve specific societal and industrial challenges.
In 2017 and 2020, the team gained international recognition for developing "Super Steel" alloys that broke the traditional trade-off between strength and ductility, creating materials that were both ultra-strong and ultra-tough for automotive and aerospace applications. In 2021, at the height of the global pandemic, the project pivoted to public health, engineering an anti-COVID-19 stainless steel capable of inactivating the SARS-CoV-2 virus on its surface within hours.
The shift toward hydrogen production began roughly six years ago. The team recognized that while stainless steel has been a staple of industrial engineering for over a century, its fundamental electrochemical limits were preventing its use in the next generation of energy technology. The journey from the initial laboratory observation of the "dual-passivation" phenomenon to the recent publication and industrial scaling reflects a rigorous period of scientific validation and engineering refinement.
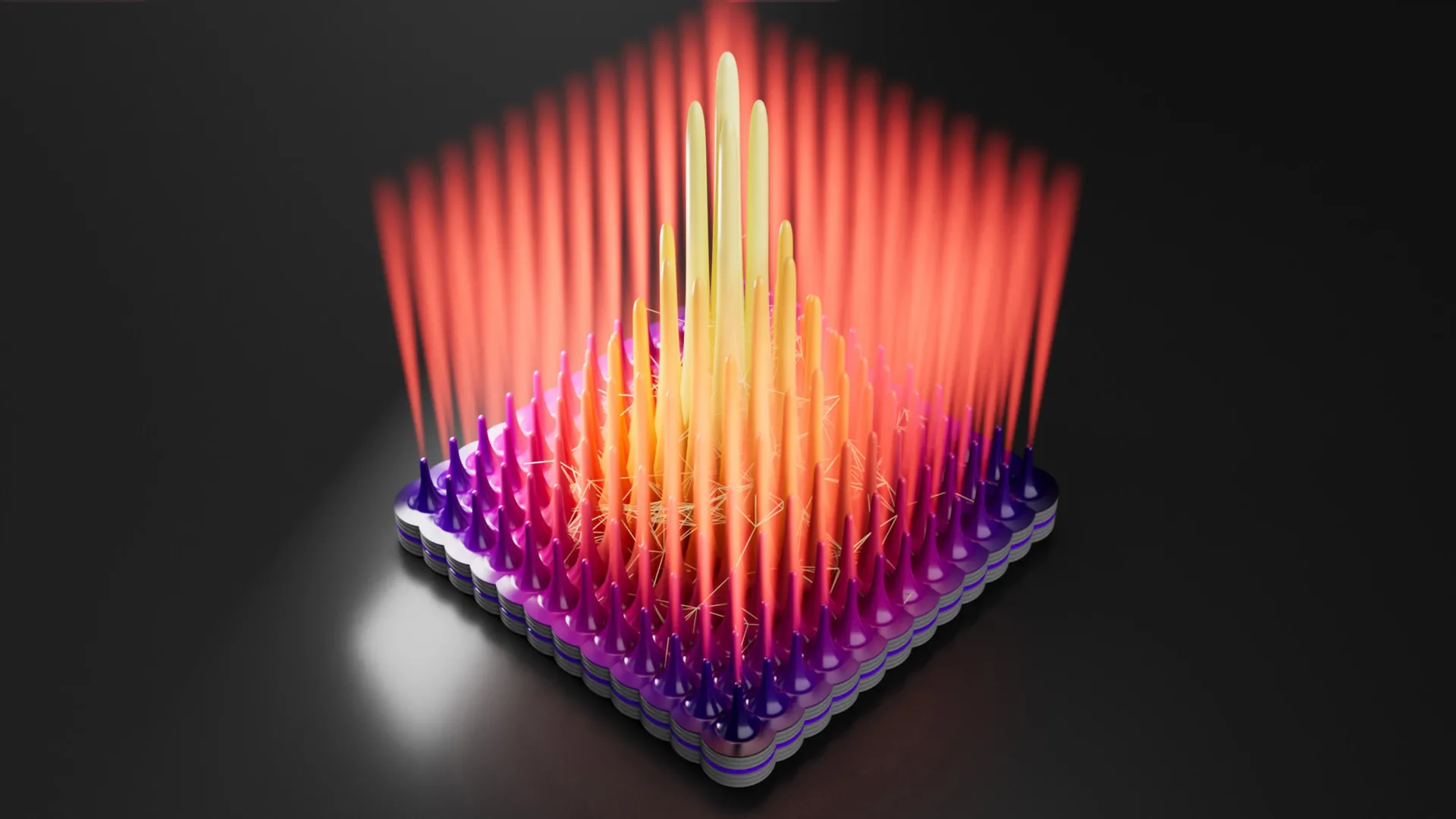
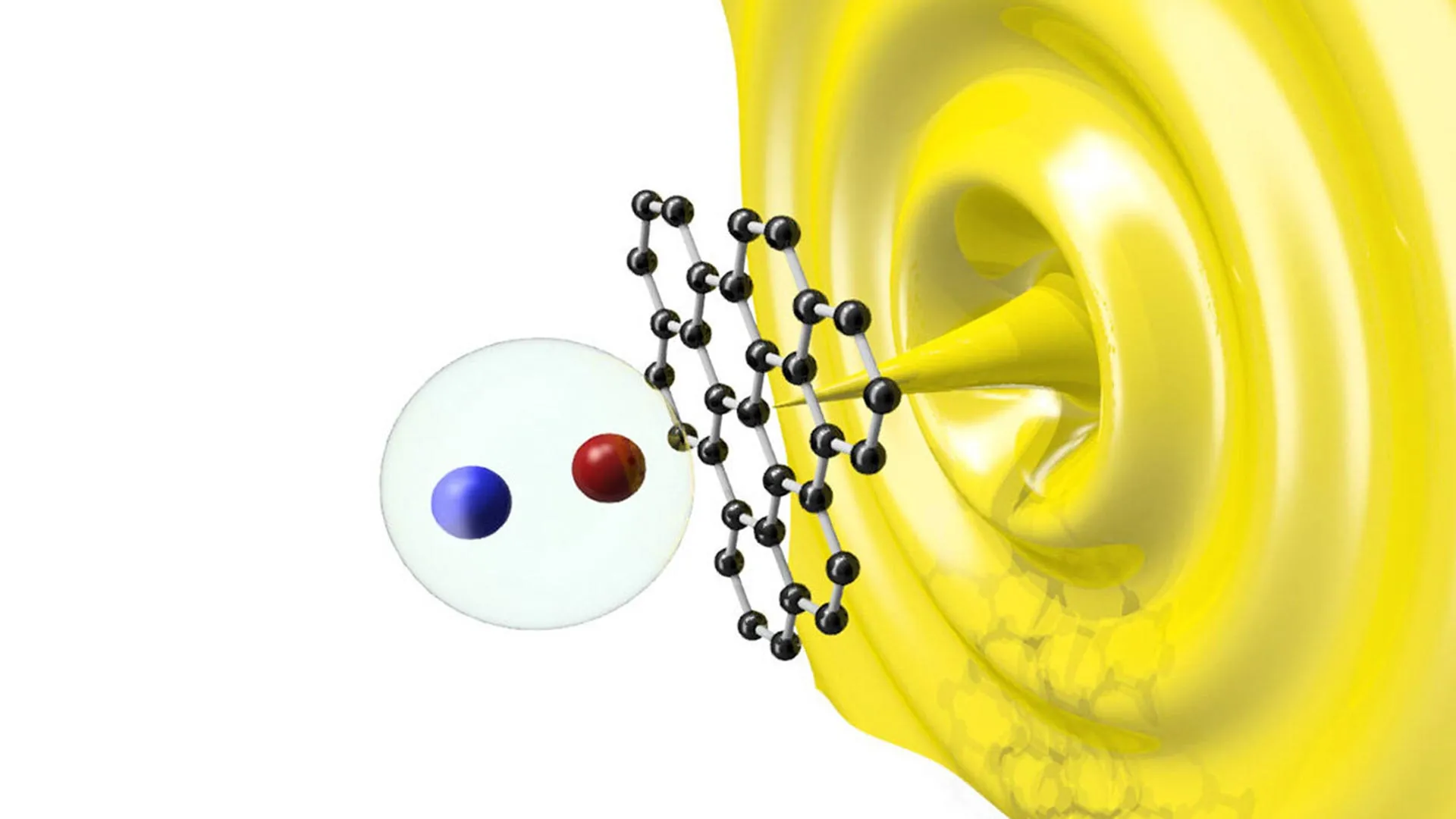
The Science of Dual-Passivation: Overcoming the Chromium Ceiling
To understand why SS-H2 is a breakthrough, one must look at the limitations of standard stainless steel. Conventional stainless steel relies on chromium to provide corrosion resistance. When exposed to oxygen, chromium forms a thin, stable layer of chromium oxide (Cr2O3) on the surface of the steel. This "passive film" protects the underlying metal from rust.
However, this protection has a functional ceiling known as the transpassive potential. In highly oxidative environments—such as those found in an electrolyzer—the electrical potential can rise to levels where the stable chromium oxide layer is further oxidized into soluble chromate (Cr(VI)) species. Once this happens, the protective film dissolves, and the steel undergoes rapid, catastrophic corrosion. For most stainless steels, this breakdown occurs at around 1000 millivolts (mV). Water oxidation, the process necessary to produce hydrogen, requires potentials of approximately 1600 mV. This gap has historically made stainless steel unsuitable for direct use in water-splitting anodes.
The HKU team’s "sequential dual-passivation" strategy introduces a second line of defense. The SS-H2 alloy incorporates manganese (Mn) in a way that defies traditional metallurgical expectations. Historically, manganese has been viewed as an element that impairs the corrosion resistance of stainless steel. However, the HKU researchers discovered that at a potential of approximately 720 mV, a manganese-based passive layer begins to form on top of the initial chromium-based layer.
This secondary shield remains stable and protective up to an ultra-high potential of 1700 mV. By creating a "shield on a shield," the team has extended the functional range of stainless steel far beyond its previous limits, allowing it to survive the extreme electrochemical environment of a seawater electrolyzer without the need for expensive coatings.
Reactions and Industrial Implementation
The discovery initially met with skepticism within the research group due to the counter-intuitive nature of using manganese for corrosion resistance. Dr. Kaiping Yu, the lead author of the study and a member of Professor Huang’s lab, noted that the prevailing scientific consensus suggested manganese would weaken the material. It was only after extensive atomic-level analysis and repeated testing that the team confirmed the mechanism behind the manganese-based passivation.
"Beyond being surprised, we cannot wait to exploit the mechanism," Dr. Yu stated, highlighting the excitement surrounding the material’s potential to disrupt current engineering paradigms.
Moving beyond the laboratory, the HKU team has taken significant steps toward the industrialization of SS-H2. Patents have been filed in multiple jurisdictions, with several already granted. Perhaps more importantly, the team has transitioned from small-batch laboratory samples to industrial-scale production. In collaboration with a manufacturing partner in Mainland China, the team has successfully produced tons of SS-H2-based wire. This wire can be processed into meshes and foams, the high-surface-area structures required for efficient electrodes and structural supports in industrial electrolyzer tanks.
Professor Huang emphasized that while the transition from experimental material to finished industrial product remains challenging, the progress made thus far is a "big step toward industrialization."
Broader Implications for the Global Energy Landscape
The relevance of the SS-H2 breakthrough has only intensified since its initial reporting. As of 2025, the global focus on seawater electrolysis has grown, as freshwater scarcity becomes an increasingly urgent issue in the context of climate change. Recent reviews in major scientific journals, such as Nature Reviews Materials, continue to identify corrosion and the high cost of components as the primary barriers to the "hydrogen economy."
While other research groups are exploring various coatings and catalysts to protect standard steel, the HKU approach is unique because it alters the alloy itself. By making the base material inherently resistant to high-potential corrosion, the HKU team provides a more robust and simplified solution that reduces the risk of coating delamination or catalyst degradation.
The implications for energy security are also significant. By reducing reliance on expensive and geographically concentrated materials like titanium and platinum-group metals, countries can build out their hydrogen infrastructure using more common industrial commodities. This could democratize access to green hydrogen technology, allowing more nations to leverage their coastal geography for energy independence.
Conclusion: A Practical Path to Scale
The development of SS-H2 represents a rare alignment of fundamental materials science and practical economic necessity. While the engineering of full-scale seawater electrolysis plants remains a complex task involving many variables—including chlorine gas suppression and mineral precipitate management—the availability of a low-cost, corrosion-resistant structural material removes one of the most daunting hurdles.
As the world seeks to move away from fossil fuels, the ability to harvest energy from the wind and sun and store it in the form of hydrogen extracted from the sea is no longer a distant dream. Through the "Super Steel" Project’s latest innovation, the University of Hong Kong has provided a tangible tool for the industrial-scale production of clean energy. The dual-shielded steel developed by Professor Huang and his team may well become the backbone of the future green hydrogen infrastructure, proving that even a century-old material like stainless steel still has the potential to surprise and transform the world.













Leave a Reply