In the intricate world of cellular biology, understanding the fundamental processes that govern life’s earliest stages is paramount. For plants, this critical window of development hinges on a sophisticated metabolic strategy that relies on stored energy reserves before the advent of photosynthesis. Central to this process is a cellular organelle known as the peroxisome, a versatile membrane-bound compartment that plays a vital role in breaking down fatty acids. New research, spearheaded by scientists at Rice University, has illuminated a key protein, PEX11, and its surprising dual function in regulating peroxisome size during this crucial seedling phase, with implications that extend far beyond the plant kingdom.
The Peroxisome: A Tiny Organelle with a Big Job
Plants, throughout most of their existence, are masters of harnessing solar energy through photosynthesis. However, for a brief but vital period after a seed germinates, this capacity is absent. During this nascent stage, young plants are entirely dependent on stored fatty acids for sustenance. The breakdown of these fatty acids is a complex biochemical process that occurs within specialized cellular compartments: peroxisomes. These organelles, present not only in plants but also in human cells, are indispensable for a variety of metabolic functions, including the detoxification of harmful substances and the synthesis of essential molecules.
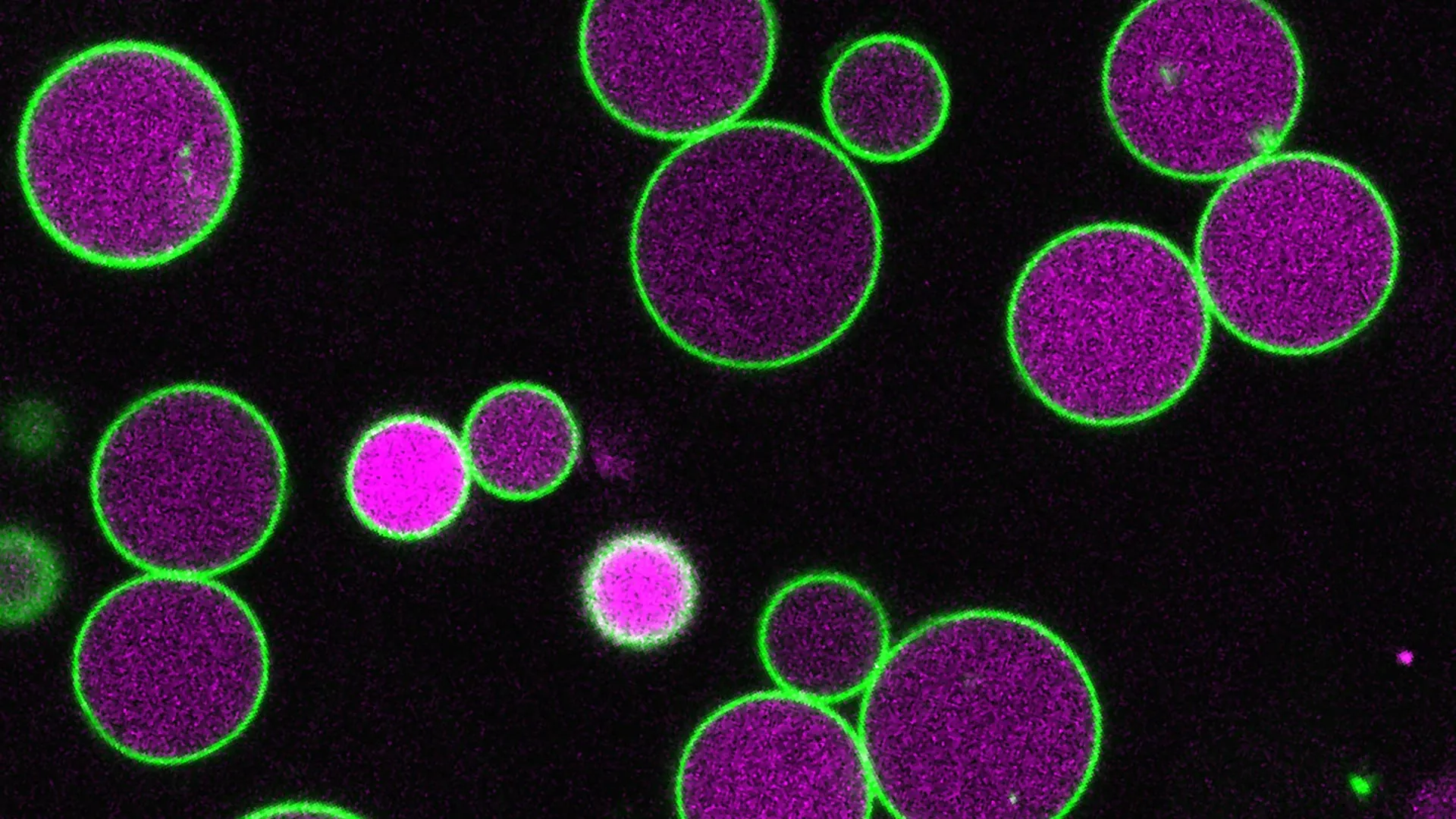
The sheer size and visibility of peroxisomes in certain plant models have made them invaluable for scientific inquiry. Arabidopsis thaliana, a small flowering plant widely used in plant biology research, is particularly well-suited for such studies. "The plant we use, Arabidopsis, has large cells and peroxisomes so large that we can see inside them with a light microscope," explained Bonnie Bartel, the Ralph and Dorothy Looney Professor of Biosciences at Rice University. "The peroxisome gets even larger during the seed to seedling stage, when the plant is relying on fatty acids for energy, before shrinking back down to its normal size once the plant can photosynthesize." This dynamic change in peroxisome size during early development hints at a sophisticated regulatory mechanism at play.
PEX11: More Than Just a Division Promoter
For years, scientists have recognized the protein PEX11 as a key player in peroxisome biology, primarily associating it with the process of peroxisome division – how these organelles multiply to meet cellular demands. However, the groundbreaking research from Bartel’s team, recently published in the prestigious journal Nature Communications, reveals a more profound and complex role for PEX11. Their findings indicate that this protein is not only involved in peroxisome division but also critically controls the organelle’s expansion and contraction during the crucial seed-to-seedling transition.
"Peroxisomes are implicated in some human diseases and used in bioengineering," commented Nathan Tharp, the lead author of the study and a Rice graduate student. "They can, however, be rather tricky to study." The challenge in studying PEX11 stemmed from the fact that its function was not easily dissectible. In Arabidopsis, the PEX11 protein is encoded by not one, but five distinct genes. This genetic redundancy presented a significant hurdle for researchers. Disrupting a single PEX11 gene had minimal observable impact on the plant, while attempting to eliminate all five proved lethal, making it impossible to isolate the specific functions of the protein.
Leveraging CRISPR for Precision Genetic Engineering
To overcome the challenge posed by the multiple PEX11 genes, Tharp employed cutting-edge CRISPR-Cas9 gene-editing technology. This revolutionary tool allows scientists to make precise modifications to DNA sequences, enabling targeted gene inactivation. "I was able to use recent advances in CRISPR to go in and break specific combinations of the five genes," Tharp stated. "It was only then that we were able to see that PEX11 is clearly involved in controlling the growth of the peroxisome during the seed to seedling stage." This strategic approach allowed the researchers to meticulously investigate the effects of altering different combinations of PEX11 genes, paving the way for a deeper understanding of the protein’s multifaceted role.
Giant Peroxisomes: A Window into Growth Regulation
The experimental manipulation of PEX11 genes yielded remarkable results. Tharp engineered two distinct lines of mutant Arabidopsis plants, each with specific combinations of PEX11 genes disabled. As anticipated, peroxisomes in these mutant plants initially enlarged during the seed-to-seedling transition, mirroring the process in wild-type plants. However, instead of returning to their normal size, these peroxisomes continued to grow unchecked, reaching unprecedented dimensions. In some extreme cases, the peroxisomes expanded to such an extent that they occupied nearly the entire volume of the cell, stretching from one end to the other.
Accompanying this dramatic enlargement of peroxisomes was a striking absence of intracellular vesicles. These small, membrane-bound sacs typically bud off from the peroxisome and play a role in its metabolic functions, including the processing of fatty acids. Under normal circumstances, the formation and budding of these vesicles appear to be intrinsically linked to the regulation of peroxisome size, effectively acting as a mechanism to shed excess membrane and control growth. In the PEX11 mutant plants, however, these crucial vesicles were either absent or abnormally small and infrequent.
"The vesicles taking pieces of membrane as they form may help control the peroxisome’s growth," Tharp elaborated. "In our PEX11 mutants, these vesicles either don’t form or are abnormally small and rare, and so we see these massive peroxisomes, way larger than normal." This observation strongly suggests that PEX11’s function extends beyond its known role in division to actively orchestrate the process of peroxisome shrinkage, likely through its influence on vesicle formation and membrane turnover.
A Conserved Mechanism: From Plants to Humans
The implications of these findings are not confined to the realm of plant science. Recognizing the fundamental nature of cellular processes, Tharp sought to determine if the observed growth control mechanism involving PEX11 was conserved across different species. To test this hypothesis, he introduced the yeast version of the PEX11 protein, known as Pex11, into the genetically engineered mutant plant cells. The results were striking: the yeast Pex11 protein was able to functionally complement the plant’s deficiency, restoring the peroxisomes to their normal size.
"We put yeast Pex11 into our mutant plant cells to see if it could return the peroxisomes back to normal," Tharp reported. "And it did." This remarkable cross-species compatibility provides compelling evidence that the fundamental mechanism by which PEX11 regulates peroxisome size has been remarkably conserved throughout evolution.
Professor Bartel emphasized the significance of this conserved function: "Finding that this protein fills the same role in yeast and plant cells suggests that it may be a highly conserved protein. Our findings in plants, in this relatively easy-to-study model, may thus be applicable to human cells and cells used for bioengineering." This discovery opens exciting avenues for research into human peroxisome-related disorders, many of which involve peroxisome dysfunction. Understanding how PEX11 regulates peroxisome size in humans could lead to novel therapeutic strategies. Furthermore, the application of peroxisomes in bioengineering, such as for the production of biofuels or pharmaceuticals, could also benefit from a deeper comprehension of their growth control mechanisms.
Timeline of Discovery and Future Directions
The research journey leading to these pivotal discoveries can be traced through several key stages:
- Early Observations (Pre-2000s): Scientists identify peroxisomes and recognize their crucial role in fatty acid metabolism and early plant development. The protein PEX11 is initially identified for its role in peroxisome division.
- Genetic Challenges (Early 2010s): Researchers encounter difficulties in studying PEX11’s full function due to the presence of multiple PEX11 genes in Arabidopsis, where disrupting individual genes has little effect and disrupting all is lethal.
- Technological Advancement (Mid-2010s): The advent of advanced CRISPR-Cas9 gene-editing techniques provides a powerful tool for precisely manipulating multiple genes simultaneously.
- Targeted Genetic Engineering (Late 2010s – Early 2020s): Nathan Tharp, under the guidance of Professor Bonnie Bartel, utilizes CRISPR to selectively disable combinations of PEX11 genes, enabling the isolation of PEX11’s growth-regulating function.
- Experimental Validation (2020-2022): The creation and analysis of mutant plants with excessively large peroxisomes, revealing the link between PEX11, vesicle formation, and peroxisome size control. Cross-species complementation experiments with yeast Pex11.
- Publication and Dissemination (2023): The findings are published in Nature Communications, bringing this significant breakthrough to the wider scientific community.
The implications of this research are far-reaching. By elucidating the precise molecular mechanisms that control peroxisome size during a critical developmental phase, scientists have gained invaluable insights into plant growth and adaptation. The conserved nature of the PEX11 protein suggests that these findings will have a profound impact on our understanding of human health and disease. Conditions such as Zellweger syndrome, a severe genetic disorder affecting peroxisome biogenesis, could potentially be better understood and perhaps even treated by targeting the PEX11 pathway. In the field of biotechnology, a more refined control over peroxisome size could optimize their function in engineered organisms for various industrial applications.
Future research will likely focus on further dissecting the precise molecular interactions between PEX11, the proteins involved in vesicle formation, and the peroxisome membrane. Investigating the upstream signals that regulate PEX11 activity during different developmental stages will also be crucial. Moreover, exploring the therapeutic potential of manipulating PEX11 in human cells and investigating its role in other organisms will undoubtedly be key areas of future scientific endeavor, building upon the solid foundation laid by this comprehensive study. The humble peroxisome, once a cellular enigma, is steadily revealing its secrets, thanks to the persistent efforts of researchers like those at Rice University.















Leave a Reply