Researchers at Oregon Health & Science University (OHSU) have unveiled a groundbreaking discovery: a previously unrecognized internal system within cells that functions akin to atmospheric "trade winds," facilitating the rapid and directed transport of essential proteins to the cell’s leading edge. This revelation fundamentally alters long-held scientific paradigms regarding cell motility, the mechanisms underlying cancer metastasis, and the intricate processes of wound healing. The findings, meticulously detailed in the esteemed journal Nature Communications, challenge the prevailing notion of intracellular protein movement as a primarily passive, diffusion-driven phenomenon.
For decades, the dominant model in cellular biology posited that proteins within the cell navigated their environments through a process akin to random drifting, known as diffusion. This textbook understanding suggested that proteins would eventually reach their intended destinations by chance. However, the OHSU study decisively refutes this passive model, demonstrating that cells actively generate directed fluid currents. These currents serve as internal conveyor belts, efficiently propelling critical proteins toward the cell’s dynamic front, the region responsible for extension, locomotion, and tissue repair.
The Serendipitous Spark: From Classroom Observation to Cellular Revelation
The genesis of this significant discovery can be traced back to an unexpected observation made during a routine neurobiology course at the prestigious Marine Biological Laboratory in Woods Hole, Massachusetts. Dr. Catherine (Cathy) Galbraith and Dr. James (Jim) Galbraith, co-corresponding authors of the study, were conducting a standard laboratory experiment with students when they encountered an anomaly that would ultimately reshape cellular biology.
"It actually started out as an unexpected finding," Dr. Cathy Galbraith recounted. "We were just conducting an experiment with students in class." The experiment involved employing a laser to temporarily render proteins invisible within a specific strip at the rear of a living cell. This technique, a common methodology for investigating intracellular transport mechanisms, allows researchers to meticulously track the movement of these proteins. During the course of the experiment, the researchers observed the emergence of an additional, distinct dark band appearing at the cell’s anterior edge – precisely the area that actively extends as a cell initiates movement.
"We kind of did it for fun and then realized this gave us a way of measuring something that wasn’t able to be measured before," Dr. Cathy Galbraith elaborated. This initial curiosity quickly transformed into a dedicated scientific pursuit.
Further in-depth investigation revealed that this newly observed dark band was not an artifact but represented a wave of soluble actin, a crucial protein integral to cell motility. This wave was being propelled forward with remarkable speed. Prior scientific consensus held that actin primarily reached this leading-edge region through the slow, random process of diffusion. The OHSU team’s findings, however, unveiled a fundamentally different and far more active mechanism at play.
"We realized the cartoon models in textbooks were missing a huge piece," Dr. Jim Galbraith remarked, underscoring the magnitude of the paradigm shift. "There had to be some kind of flow in the cell pushing things forward. Cells really do ‘go with the flow.’" This pithy observation encapsulates the essence of their discovery: cells are not passive recipients of molecular movement but active orchestrators of internal currents.
Directed Flows: The Engine of Intracellular Protein Transport
The Galbraiths, who joined OHSU in 2013 after significant research contributions at the National Institutes of Health (NIH), brought with them a wealth of experience. Their prior work included collaborations with Nobel Laureate Dr. Eric Betzig at the Howard Hughes Medical Institute’s Janelia Research Campus, where they honed their expertise in advanced imaging techniques. This collaboration proved instrumental in their subsequent groundbreaking research.
Leveraging sophisticated imaging tools, the OHSU team was able to conclusively demonstrate that cells actively generate directional fluid flows. The researchers aptly compare these internal cellular currents to atmospheric rivers – powerful, concentrated streams of moisture that influence global weather patterns. These cellular "rivers" efficiently transport actin and a diverse array of other proteins towards the cell’s front, achieving speeds far exceeding what passive diffusion could achieve alone.
"We found that the cell can actually squeeze at the back and target where it sends that material," Dr. Jim Galbraith explained, drawing a relatable analogy. "If you squeeze half a sponge, the water only goes on that half. That’s basically what the cell is doing." This active squeezing mechanism at the cell’s posterior appears to be the driving force behind the directed cytoplasmic streaming observed.
A critical characteristic of these newly identified flows is their nonspecific nature. This means they are not limited to transporting a single type of protein but can carry multiple protein types simultaneously. This inherent efficiency creates a robust system that robustly supports essential cellular functions such as protrusion (the extension of the cell membrane), adhesion (the attachment to surfaces), and rapid shape alterations. These processes are paramount for cell migration, the complex mechanisms of immune responses, and the critical task of tissue regeneration.
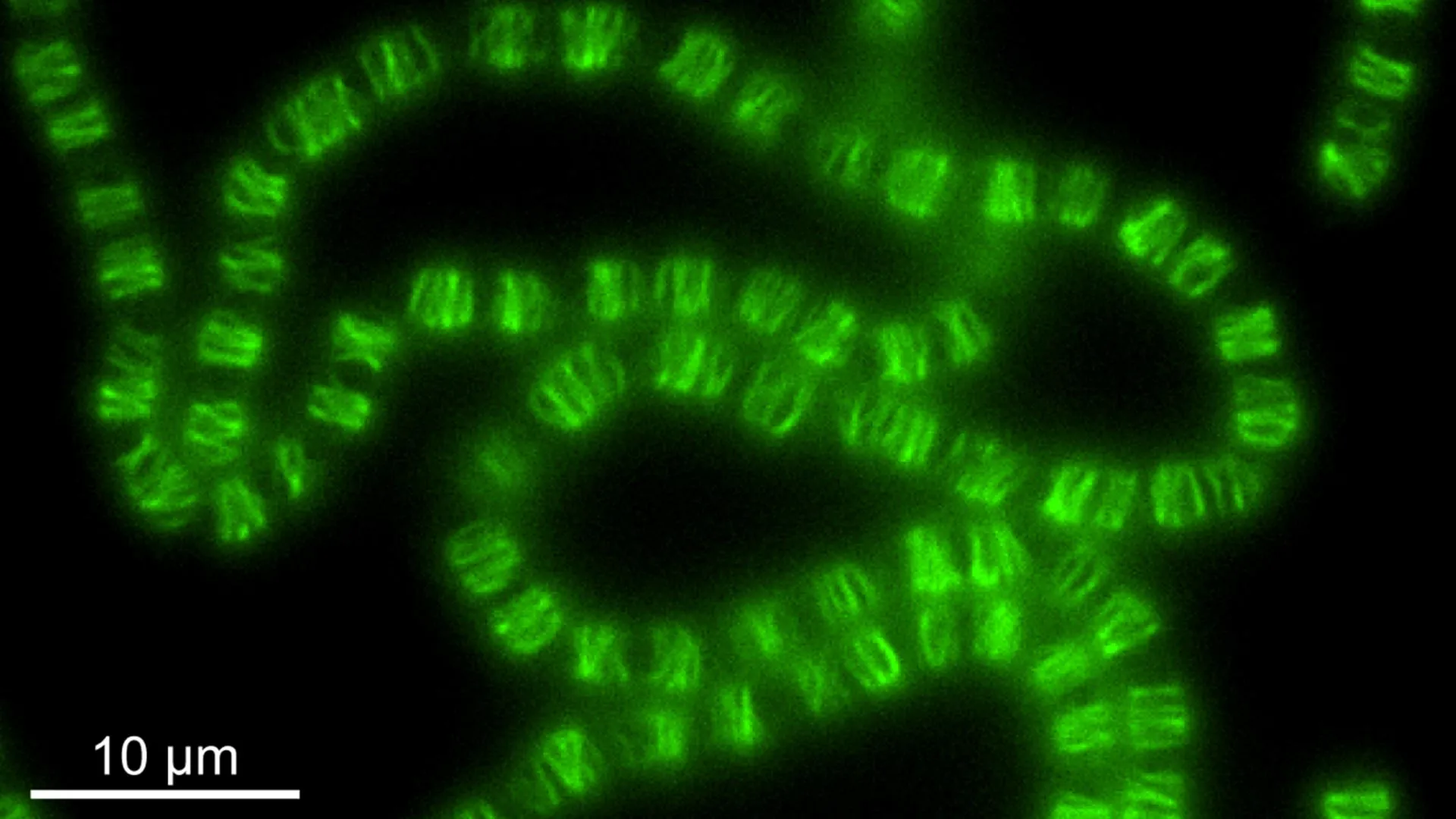
Furthermore, the research identified that these directed flows are contained within a specialized region at the cell’s anterior. This distinct area is demarcated from the rest of the cellular interior by a sophisticated barrier formed by actin and myosin. This "actin-myosin condensate barrier" functions as a physical boundary, effectively channeling and directing the protein-laden flows specifically towards the advancing edge of the cell.
Visualizing Cellular Currents: Innovation in Imaging
To meticulously observe and characterize these internal cellular currents, the OHSU team developed a novel modification of a standard fluorescence-based imaging technique. Rather than employing a laser to quench fluorescence, as is common in some experimental protocols, they devised a method to activate fluorescent molecules at a precise single point and then meticulously track their subsequent movement.
One of their key experimental innovations was humorously christened FLOP, an acronym for "Fluorescence Leaving the Original Point." The name, born from a moment of playful experimentation, belied the experiment’s profound success. "It wasn’t a flop at all," Dr. Cathy Galbraith stated with evident satisfaction. "It was the opposite. It is anything but a flop, because it worked." The successful application of the FLOP experiment provided the visual evidence necessary to confirm the existence and dynamics of these internal cellular flows.
Implications for Cancer Cell Aggression and Metastasis
The implications of this discovery extend significantly into the realm of oncology, offering potential explanations for the aggressive migratory behavior observed in certain cancer cells. The ability of cancer cells to rapidly and efficiently transport proteins to their leading edge is a hallmark of invasiveness and metastasis, the process by which cancer spreads to distant parts of the body.
"We know these highly invasive cells have this really cool mechanism to push proteins really fast, really rapidly where they need them at the front of the cell," Dr. Jim Galbraith explained. He drew a compelling analogy between different cell types and vehicles: "All cells have basically the same components inside, much like a Porsche and a Volkswagen have many of the same parts, but when those parts are assembled into the final machine, they behave and function very differently." This suggests that while normal cells utilize these internal flows for essential functions, cancer cells may have evolved to exploit or amplify this system for their own proliferative and invasive agenda.
By deciphering how cancer cells uniquely leverage this internal transport system compared to their healthy counterparts, scientists may be empowered to devise novel therapeutic strategies. These could target and disrupt the aberrant activity of these cellular currents, potentially slowing or halting the spread of malignant tumors. "If you can understand the differences, you can target future therapies based on how cancer cells and normal cells work differently," Dr. Galbraith emphasized, highlighting the translational potential of their fundamental research.
A Symphony of Expertise: Advanced Imaging and Interdisciplinary Collaboration
The groundbreaking nature of this discovery was facilitated by a remarkable convergence of expertise from diverse scientific disciplines, including engineering, physics, advanced microscopy, and cell biology. Crucially, the research benefited from the invaluable contributions of collaborators at the Janelia Research Campus in Virginia. These included specialists in fluorescence correlation spectroscopy and cutting-edge 3D super-resolution imaging, technologies essential for visualizing cellular structures and processes at unprecedented detail.
"The instrumentation we needed doesn’t exist in most places," Dr. Cathy Galbraith noted, underscoring the specialized nature of the equipment required. "Janelia had a one-of-a-kind setup that let us test and confirm what we were seeing." The study relied heavily on advanced imaging platforms developed at Janelia, notably iPALM (interferometric Photoactivated Localization Microscopy), a technique renowned for its ability to resolve cellular structures at the nanometer scale.
"iPALM allowed us to physically see the compartments," Dr. Jim Galbraith stated. "There’s no other light-based technique that could do that." This advanced imaging capability was critical in visualizing the actin-myosin condensate barrier and the fluid dynamics within it.
The "Pseudo-Organelle": A Newly Identified Functional Compartment
The researchers have aptly described this newly identified internal transport system as a "pseudo-organelle." Unlike traditional organelles that are enclosed by a membrane, a pseudo-organelle is a functional compartment that exerts significant influence over cellular organization and behavior without a lipid bilayer boundary. This classification highlights its critical role in cellular function despite its unconventional structure.
"Just as small shifts in the jet stream can change the weather, small changes in these cellular winds could change how diseases begin or progress," Dr. Cathy Galbraith posited, drawing another powerful analogy to underscore the far-reaching implications of their findings. The team is confident that this discovery will have a ripple effect across multiple scientific fields, including cancer research, the development of targeted drug delivery systems, advancements in tissue engineering and regeneration, and the burgeoning field of synthetic biology.
"All you had to do was look," Dr. Cathy Galbraith concluded, reflecting on the seemingly simple yet profound nature of their observation. "The flows were there all along. Now we know how cells use them." This sentiment speaks to the power of meticulous observation and the willingness to challenge established scientific dogma.
The published study lists Dr. Brian English of Janelia Research Campus and Dr. Ulrike Boehm, formerly of Janelia and now with Carl Zeiss AG in Germany, as coauthors alongside the Galbraiths. This collaborative effort underscores the interdisciplinary nature of modern scientific advancement.
The research was generously supported by funding from several prominent institutions, including the National Institute of General Medical Sciences of the National Institutes of Health (Award number R01GM117188), the U.S. National Science Foundation (Award numbers 2345411 and 171636), the W. M. Keck Foundation, the Howard Hughes Medical Institute Janelia Visiting Scientist Program, and the Howard Hughes Medical Institute. Additional support for the iPALM work came from an award from the Advanced Imaging Center at Janelia, and the SIM (Structured Illumination Microscopy) imaging was partially funded by a Core Research Facilities grant from the OHSU School of Medicine, illustrating a broad base of institutional support for this fundamental scientific endeavor.















Leave a Reply