Photosynthetic bacteria, particularly cyanobacteria, are credited with fundamentally transforming Earth’s atmosphere and paving the way for complex life. Their pioneering role in oxygenic photosynthesis, culminating in the Great Oxygenation Event approximately 2.5 billion years ago, irrevocably altered the planet’s habitability, making aerobic respiration – and thus, the evolution of animals, fungi, and plants as we know them – possible. Despite their ancient origins, these microorganisms continue to be vital to Earth’s ecosystems today, contributing significantly to global biomass and playing crucial roles in the carbon and nitrogen cycles. Their remarkable adaptability allows them to thrive in diverse and often extreme environments, from scorching hot springs to frigid Arctic landscapes, and even on urban surfaces. Among these resilient organisms, Anabaena sp. PCC 7120, often referred to simply as Anabaena, has served as a model organism for over three decades, offering invaluable insights into the biology of multicellular cyanobacteria.
Now, groundbreaking research from the Institute of Science and Technology Austria (ISTA) has revealed a surprising evolutionary twist in the intricate workings of these ancient life forms. A biological system once thought to be solely dedicated to the precise segregation of DNA during cell division has, in a remarkable feat of evolutionary repurposing, been transformed into a cellular structure that dictates the very shape of cyanobacterial cells. This discovery, published in the prestigious journal Science, not only sheds new light on the dynamic nature of protein systems and their evolutionary trajectories but also offers a deeper understanding of how multicellularity arose and is maintained in these ecologically critical bacteria.
The Genesis of an Oxygenated World: Cyanobacteria’s Transformative Impact
The advent of oxygenic photosynthesis by cyanobacteria marked a pivotal moment in Earth’s history. Prior to this evolutionary leap, Earth’s atmosphere was largely devoid of free oxygen. The metabolic byproduct of cyanobacteria, oxygen, gradually accumulated over hundreds of millions of years, a period known as the Great Oxygenation Event (GOE). This dramatic shift from an anoxic to an oxygen-rich environment, which began around 2.4 to 2.3 billion years ago, had profound consequences. It rendered many anaerobic organisms extinct while simultaneously creating an environment where organisms utilizing oxygen for energy production – aerobic respiration – could flourish. This metabolic innovation is the bedrock upon which complex multicellular life, including all animals, is built.
Benjamin Springstein, a postdoctoral researcher in the Loose group at ISTA, emphasizes the profound significance of these ancient microbes: "Cyanobacteria are essentially pioneers of oxygenic photosynthesis," he states. "They are responsible for the Great Oxygenation Event about 2.5 billion years ago, when oxygen accumulated in the atmosphere and made aerobic life possible. Without them, it’s safe to say that none of us would be here today."
Even in the present day, the ecological importance of cyanobacteria cannot be overstated. They are a ubiquitous and essential component of aquatic and terrestrial ecosystems. Their photosynthetic activity forms the base of many food webs, and their contributions to global primary productivity are substantial. Furthermore, their involvement in biogeochemical cycles, particularly those of carbon and nitrogen, is critical for maintaining planetary health. Their ability to fix atmospheric nitrogen, a process that converts it into a usable form for other organisms, is indispensable for plant growth and agricultural productivity.
Unraveling the Evolutionary Shift: From DNA Segregation to Cell Sculpting
The recent findings from ISTA, in collaboration with researchers from the Institut Pasteur de Montevideo (Uruguay), Kiel University (Germany), and the University of Zürich (Switzerland), illuminate a sophisticated evolutionary adaptation within multicellular cyanobacteria, exemplified by Anabaena. The study’s lead author, Benjamin Springstein, along with Professor Martin Loose and their colleagues, identified a system known as ParMR, a molecular machinery traditionally associated with the faithful inheritance of genetic material during cell division. However, in Anabaena and potentially other multicellular cyanobacteria, this system has undergone a dramatic functional metamorphosis.
Instead of its original role in ensuring each daughter cell receives an accurate copy of the chromosome and any plasmids, the ParMR system has been repurposed. It now functions as a crucial component of the cell’s internal scaffolding, akin to a cytoskeleton, actively contributing to the maintenance and definition of cell shape. This transformation represents a profound example of how biological systems can be co-opted for entirely new purposes over evolutionary timescales.
The Intricacies of Bacterial DNA: Chromosomes and Plasmids
To understand the significance of this evolutionary shift, it’s essential to grasp the basics of DNA organization and replication in bacteria like Anabaena. Bacteria reproduce asexually, primarily through binary fission, a process where a single cell divides into two identical daughter cells. For this to occur successfully, the bacterial cell must accurately duplicate its genetic material and ensure that each new cell receives a complete set of instructions for survival and function.
Bacterial DNA is organized into two main forms: chromosomes and plasmids. The bacterial chromosome is the primary genetic blueprint, typically a circular molecule containing all the essential genes required for the organism’s life. This chromosome is tightly packed within the cell, often in a region called the nucleoid. Plasmids, on the other hand, are smaller, extrachromosomal DNA molecules that carry additional genes. While not essential for basic survival under normal conditions, these genes can confer advantageous traits, such as antibiotic resistance or the ability to metabolize specific compounds. A key characteristic of plasmids is their mobility; they can be readily transferred between bacteria, facilitating rapid adaptation and the spread of novel traits within a population.
A DNA Segregation System Reimagined: The Case of ParMR
Springstein’s research journey into Anabaena began in 2014, focusing on its fundamental biology and evolutionary history. During the unprecedented global pause in laboratory research brought about by the COVID-19 pandemic, Springstein dedicated time to an in-depth review of scientific literature. It was during this period of introspection and theoretical exploration that he made a "serendipitous observation."
He discovered that the ParMR system, a genetic locus typically found on plasmids and known for its role in plasmid segregation, was present within the chromosomal DNA of Anabaena and certain related cyanobacteria. This unusual genomic location immediately sparked his suspicion. The conventional understanding of ParMR was tied to its function on mobile genetic elements. Its presence on the chromosome suggested a potential adaptation for a different purpose, possibly involving the segregation of the main bacterial chromosome itself.
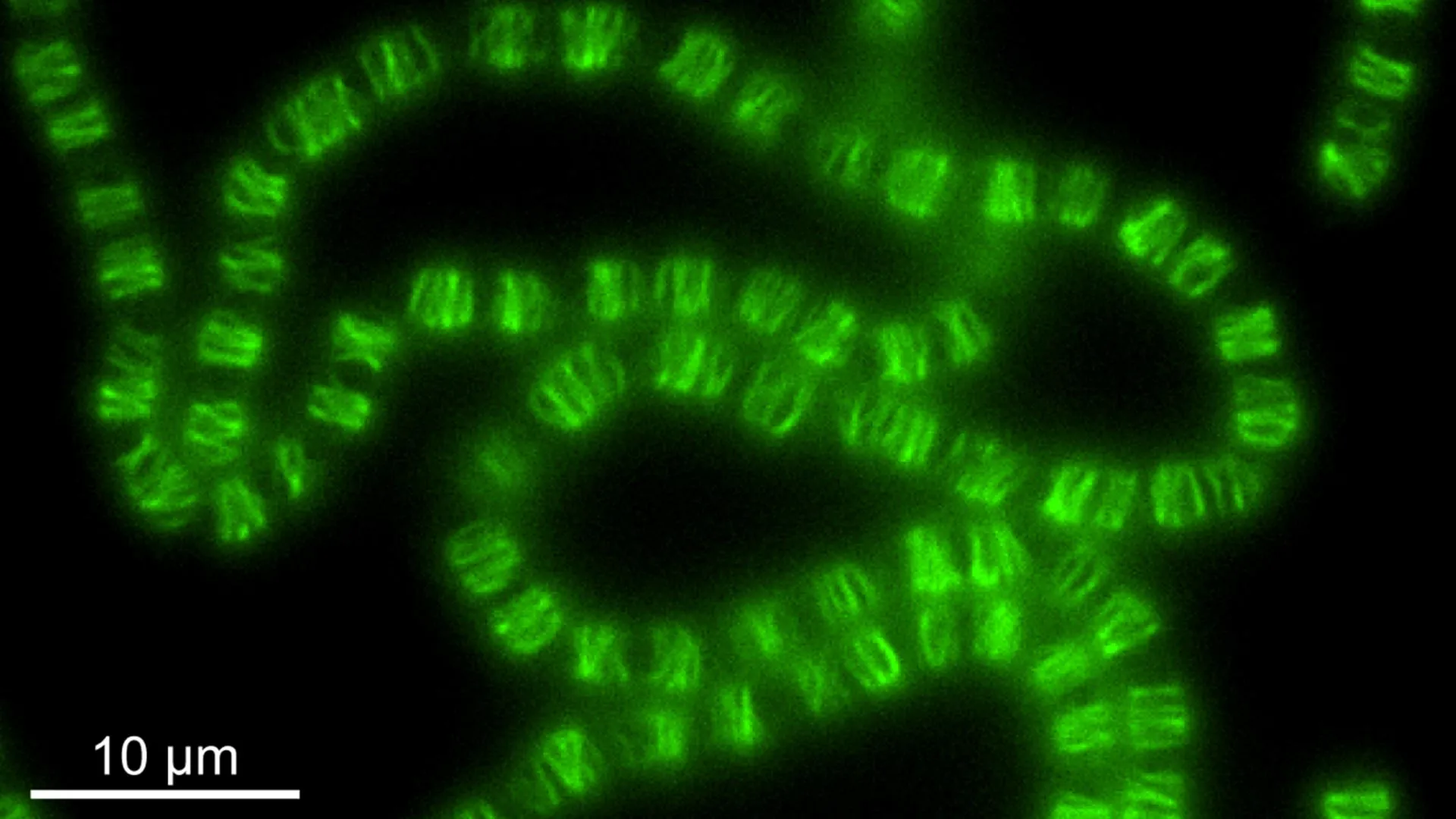
Upon joining ISTA as an IST-Bridge Fellow, Springstein was equipped to rigorously test this hypothesis. His experimental investigations, however, yielded results that diverged significantly from his initial expectations. He found that ParR, one component of the system, had lost its affinity for DNA. Instead, it demonstrated a strong propensity to bind to lipid membranes, particularly the inner cell membrane. Concurrently, the other component, ParM, was not observed to form the dynamic filament structures typically seen in the cytoplasm for DNA movement. Rather, ParM assembled into filament networks localized just beneath the inner membrane. These protein polymer networks formed a layer reminiscent of a cell cortex, a structural element found in eukaryotic cells that provides shape and mechanical support.
The implications were profound: this system was not operating as a conventional DNA segregation mechanism within the cell’s interior, forming spindle-like structures to pull chromosomes apart. Instead, its activity was concentrated at the cell membrane, and its apparent function was the organization and structural integrity of the cell itself.
Filamentous Architects: A Cellular Skeleton Emerges
To gain a deeper understanding of this novel cellular architecture, the research team embarked on in vitro reconstitution experiments. By purifying the components of the CorMR system (as it would later be renamed) and assembling them outside of a living cell, they could observe their intrinsic behaviors. These experiments revealed that the ParM filaments exhibited dynamic instability, a characteristic crucial for cellular processes like cell division and movement. The filaments would spontaneously grow and then rapidly depolymerize, a behavior mirroring that of microtubules in more complex eukaryotic cells.
Further collaboration with ISTA Professor Florian Schur and his PhD student Manjunath Javoor brought advanced imaging techniques to bear on the problem. Using cryo-electron microscopy (cryo-EM), a powerful method for visualizing molecular structures at near-atomic resolution, the team was able to dissect the intricate assembly of these filaments. Their cryo-EM analysis revealed a significant difference from similar bacterial systems: the filaments formed by this repurposed system in Anabaena were bipolar. This means they could grow and shrink from both ends, a property that likely contributes to their dynamic structural role. In contrast, many other bacterial filament systems are polar, growing and shrinking primarily from one end.
The Moment of Truth: Loss of Function Reveals Cell Shape Control
The definitive evidence for the system’s new role emerged when its function was experimentally disrupted in living Anabaena cells. When the CorMR system was removed or inactivated, the consequences for cell morphology were dramatic.
"Cells lacking the system lost their normal rectangular-like cell shape and instead became round and swollen," explained Springstein.
These observed changes—a loss of defined shape and a tendency towards spherical morphology—are hallmarks of disruptions to genes responsible for maintaining cell shape in other bacterial species. This direct correlation strongly indicated that the primary function of this ancient DNA segregation machinery had indeed been repurposed for cell structure maintenance, rather than its original task of DNA distribution. Recognizing its new role and cellular location, the researchers proposed renaming the system "CorMR," an acronym derived from "cortex-like membrane protein system."
A Gradual Transformation: Tracing the Evolutionary Pathway of CorMR
The evolution of multicellularity in cyanobacteria, including the adaptation of the CorMR system, was a gradual process. Daniela Megrian, a collaborator from the Institut Pasteur in Montevideo, conducted bioinformatic analyses that provided crucial insights into the step-by-step evolutionary journey of the CorMR system.
The transformation was not a single, sudden event but rather a series of incremental changes. Initially, the system transitioned from its ancestral location on plasmids to the bacterial chromosome. This chromosomal integration likely provided a more stable and reliable platform for its evolving functions. Subsequently, the protein components of the system, ParM and ParR, underwent structural and size modifications, adapting them for their new roles. A critical step was the development of the ability of ParR to bind to cell membranes, anchoring the system to the cell’s periphery. Finally, the expression and regulation of the CorMR system became integrated with other cellular control mechanisms, likely through its interaction with additional protein systems, further refining its function in shaping the cell.
This sequence of evolutionary events highlights a fundamental principle in biology: evolution often works by modifying and repurposing existing molecular machinery. The repurposing of an ancient DNA segregation mechanism into a cell-shaping system in cyanobacteria provides a striking illustration of this principle. It demonstrates how biological innovations can arise from subtle alterations to well-established pathways, leading to entirely new functionalities and contributing to the diversification and complexity of life on Earth.
Broader Implications: Evolution, Complexity, and Future Research
The discovery of the CorMR system’s dual role has significant implications for our understanding of evolutionary biology and the development of cellular complexity. It provides a tangible example of how a system critical for basic bacterial reproduction can be co-opted to contribute to multicellular organization and shape. This finding could offer a new lens through which to examine the evolution of more complex cellular structures, including the cytoskeleton in eukaryotes, which shares some functional parallels with the CorMR system.
The ability of Anabaena to form filaments and develop into a multicellular organism is dependent on precise control over cell division and the maintenance of individual cell integrity. The CorMR system appears to be a key player in this process, ensuring that the filamentous structure remains robust and functional. This could be particularly important in environmental conditions where mechanical stress or the need for coordinated movement among cells is a factor.
Future research will likely focus on further elucidating the precise molecular mechanisms by which CorMR interacts with the cell membrane and other cellular components to dictate cell shape. Investigating the evolutionary history of this system in other cyanobacterial lineages could reveal whether this repurposing is a widespread phenomenon or a specific adaptation in Anabaena and its close relatives. Understanding the interplay between genetic inheritance systems and cellular morphogenesis in these ancient microbes may also provide insights into the fundamental principles that govern the evolution of multicellularity across the tree of life. The ongoing study of cyanobacteria continues to reveal their profound importance, not just for the Earth’s past and present, but also for understanding the very foundations of life’s complexity.















Leave a Reply