One of the most persistent and formidable challenges in the relentless pursuit of understanding aging and its associated diseases lies in the ability to accurately track and target senescent cells. These cellular entities, often colloquially referred to as "zombie cells," represent a critical biological paradox: they cease their normal process of division, a hallmark of cellular retirement, yet they stubbornly refuse to undergo programmed cell death, or apoptosis. Over time, this accumulation of non-functional, yet metabolically active, cells can create a significant burden within the body, contributing to a spectrum of debilitating conditions. Scientific evidence has increasingly linked the presence and buildup of senescent cells to the pathogenesis of chronic diseases, including various forms of cancer, neurodegenerative disorders such as Alzheimer’s disease, and the fundamental processes that drive organismal aging itself.
For years, the scientific community has diligently explored diverse strategies aimed at either eradicating these harmful cells or reversing their detrimental effects. However, a fundamental and persistent obstacle has hindered progress: the sheer difficulty in reliably distinguishing senescent cells from the vast population of healthy, functioning cells within living tissues. This lack of precise identification has been a significant bottleneck, limiting the efficacy of therapeutic interventions and complicating fundamental research.
A Groundbreaking Discovery: Aptamers Emerge as Precision Tools
In a significant stride forward, a dedicated team of researchers at the Mayo Clinic has unveiled a promising new strategy that could revolutionize the identification of senescent cells. Detailed in a recent publication in the esteemed journal Aging Cell, their innovative technique leverages the unique properties of molecules known as "aptamers" to precisely tag these elusive senescent cells.
Aptamers are essentially short, synthetic strands of DNA, meticulously engineered to fold into intricate three-dimensional structures. This precise folding is crucial, as it bestows upon them the remarkable ability to bind with high specificity to particular proteins that are expressed on the surfaces of cells. Think of them as highly specialized molecular keys, designed to fit only specific cellular locks.
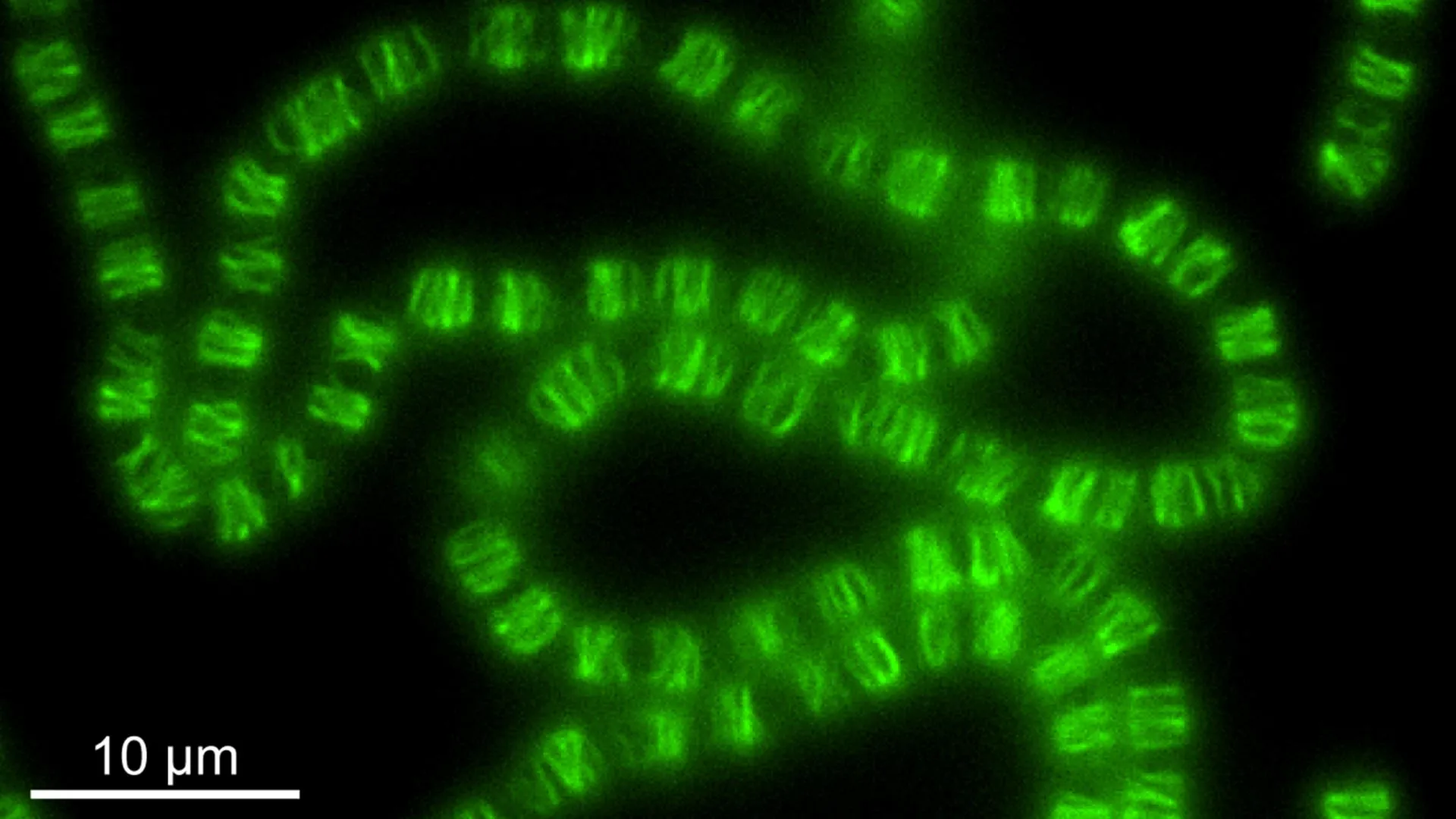
In their groundbreaking study, the Mayo Clinic scientists embarked on an extensive screening process involving an astronomical number of more than 100 trillion random DNA sequences. Their rigorous examination, conducted using mouse cells as a model system, successfully identified several rare aptamers that exhibited a potent affinity for proteins specifically associated with senescent cells. Once these aptamers successfully attach to their target proteins, they effectively act as molecular beacons, flagging the senescent cells and making them readily identifiable for further analysis and potential therapeutic targeting.
"This approach has unequivocally established the principle that aptamers represent a powerful technology that can be effectively employed to distinguish senescent cells from their healthy counterparts," stated biochemist and molecular biologist Jim Maher, III, Ph.D., a principal investigator of the study. He further elaborated, "While this particular study represents a crucial first step, the compelling results strongly suggest that this innovative approach holds significant potential for eventual application to human cells, opening new avenues for research and clinical interventions."
The Genesis of Innovation: A Serendipitous Conversation
The genesis of this significant scientific breakthrough can be traced back to an unexpected yet pivotal moment: a casual conversation between graduate students at the Mayo Clinic. This seemingly serendipitous exchange, occurring outside the formal structure of research presentations, laid the groundwork for a collaborative effort that would yield remarkable results.
Keenan Pearson, Ph.D., who had recently completed his doctoral studies at the Mayo Clinic Graduate School of Biomedical Sciences, had been immersed in investigating the potential applications of aptamers in combating brain cancer and neurodegenerative diseases, working under the guidance of Dr. Maher. Concurrently, elsewhere on the Mayo Clinic campus, Sarah Jachim, Ph.D., was engaged in her graduate research, focusing on the intricate biology of aging and the characteristics of senescent cells in the laboratory of Nathan LeBrasseur, Ph.D.
Their paths converged at a scientific event, where the natural inclination of researchers to discuss their work led to an exchange of ideas. As Dr. Pearson shared details of his aptamer research, Dr. Jachim provided insights into her studies on senescent cells. This interdisciplinary dialogue sparked a novel line of inquiry in Dr. Pearson’s mind: could the sophisticated aptamer technology he was utilizing be adapted to specifically recognize and bind to senescent cells?
"I immediately recognized the potential of the idea, but I lacked the specific expertise in preparing and culturing senescent cells for experimental testing, which was Sarah’s area of specialization," recalled Dr. Pearson, who ultimately took on the role of lead author for the published study. This recognition of complementary expertise was the critical catalyst for the subsequent collaboration.
Embracing a "Crazy" Idea: A Collaborative Leap of Faith
The nascent idea, born from the students’ collaborative brainstorming, was then presented to their mentors, including Dr. Maher, and also to Darren Baker, Ph.D., a researcher whose work is prominently focused on developing therapies specifically targeting senescent cells.
According to Dr. Maher, the concept initially sounded rather unconventional, even "crazy." However, its inherent intrigue and the potential it held for addressing a critical research gap compelled further investigation. Ultimately, the mentors embraced the proposed collaboration, recognizing its unique interdisciplinary synergy.
"We were genuinely enthusiastic about the fact that this was a student-driven idea, representing a true confluence of two distinct yet complementary research areas," Dr. Maher remarked, highlighting the organic nature of the project’s inception.
The research progressed with remarkable speed. The early experimental stages yielded encouraging findings much sooner than anticipated, a testament to the promising nature of the initial hypothesis. This early success motivated the team to expand its scope, drawing in additional students from various laboratories to contribute their specialized skills.
The collaborative effort saw the invaluable contributions of then-graduate students Brandon Wilbanks, Ph.D., Luis Prieto, Ph.D., and M.D.-Ph.D. student Caroline Doherty. Their expertise encompassed advanced techniques such as sophisticated microscopy and the comprehensive analysis of a diverse array of tissue samples. This broader involvement accelerated the pace of discovery and validation.
"The momentum built as we witnessed the project’s potential for success, which naturally encouraged us to invest more effort and resources," Dr. Jachim commented, reflecting on the growing confidence and enthusiasm within the research group.
Unveiling New Insights into Cellular Biology
Beyond providing a novel and effective method for identifying senescent cells, this pioneering study has also yielded unexpected but significant new information about the inherent biology of these "zombie cells."
"Up to this point, there has been a notable absence of universally recognized markers that definitively characterize senescent cells," explained Dr. Maher. "Our study was intentionally designed with an open-ended approach regarding the specific target surface molecules on senescent cells. The elegance of this methodology lies in allowing the aptamers themselves to autonomously select the molecules to which they will bind."
Remarkably, several of the identified aptamers demonstrated a strong affinity for a specific variation of fibronectin, a protein commonly found on the surface of mouse cells. While the precise functional relationship between this fibronectin variant and the senescent state remains an active area of investigation, this discovery offers valuable clues. It provides researchers with a potential new avenue to better define the unique molecular signatures that distinguish senescent cells from their healthy counterparts, thereby enhancing our understanding of cellular aging.
The Horizon of Possibility: Future Implications for Health and Longevity
While the researchers express considerable optimism regarding their findings, they also emphasize the need for further, more extensive studies before aptamers can be reliably deployed for the identification of senescent cells in human clinical settings. The transition from laboratory models to human application is a rigorous and time-consuming process, involving thorough validation and safety assessments.
However, the potential applications of this aptamer-based technology extend far beyond mere diagnostic or detection capabilities. Scientists envision a future where aptamers could serve as sophisticated delivery vehicles, capable of carrying therapeutic agents directly to senescent cells. This highly targeted approach could significantly enhance the efficacy of treatments while minimizing potential side effects on healthy tissues, a major goal in the development of new aging and disease therapies.
Dr. Pearson highlighted another significant advantage of aptamers: their comparative cost-effectiveness and adaptability when contrasted with traditional antibodies. Antibodies, while widely used in cell identification and research, can be expensive to produce and often have limitations in their stability and ability to be modified for specific delivery purposes. Aptamers, being synthetic DNA molecules, offer greater flexibility in design and production, potentially leading to more accessible and versatile diagnostic and therapeutic tools.
"This project has successfully demonstrated a truly novel concept," concluded Dr. Maher. "The future trajectory of this research holds immense promise, with subsequent studies poised to extend this innovative aptamer-based approach to address the complex challenges posed by senescent cells in a wide range of human diseases." The implications for developing novel interventions for age-related ailments, from cardiovascular disease and osteoarthritis to neurodegenerative conditions and cancer, are substantial, offering a glimpse into a future where targeted cellular interventions could significantly improve human healthspan and quality of life.















Leave a Reply