The sense of smell has long been regarded as the most enigmatic of the human senses, a biological frontier that has eluded the precise mapping accorded to vision, hearing, and touch. While scientists have mapped the way the retina captures light and how the cochlea processes sound, the nose remained a chaotic landscape where millions of neurons seemed to be scattered without a discernible pattern. However, a landmark study published in the journal Cell has finally provided a definitive biological atlas for olfaction. Researchers at Harvard Medical School have constructed the first detailed map of the nose’s internal architecture, revealing that the detection of scent is governed by a highly organized, "striped" system that mirrors the complex neural circuits of the brain.
Led by Sandeep Robert Datta, a professor of neurobiology in the Blavatnik Institute at Harvard Medical School, the research team utilized cutting-edge genetic sequencing and spatial mapping technologies to analyze the nasal tissues of mice. Their findings challenge decades of scientific assumptions, proving that the olfactory system is not a random collection of sensors but a precision-engineered map. This discovery marks a pivotal shift in sensory biology, offering a potential blueprint for treating smell disorders and advancing our understanding of how the brain interprets the chemical world.
The Mystery of the Olfactory Frontier
For decades, the biological community has struggled to explain how the nose organizes its vast array of sensors. In vision, the physical arrangement of receptors on the retina directly corresponds to the spatial layout of the world we see. In hearing, the arrangement of hair cells in the ear corresponds to different frequencies of sound. Smell, however, appeared to be the exception to this rule of spatial organization.
"Olfaction is super-mysterious," Datta observed, noting that compared to other senses, the basic biology of smell has remained significantly less understood. The complexity of the system is the primary reason for this knowledge gap. While human color vision relies on just three types of light-sensitive receptors, mice possess more than 1,000 different types of smell receptors, and humans possess approximately 350 to 400. Each of these receptors is tuned to detect specific odor molecules or chemical signatures.
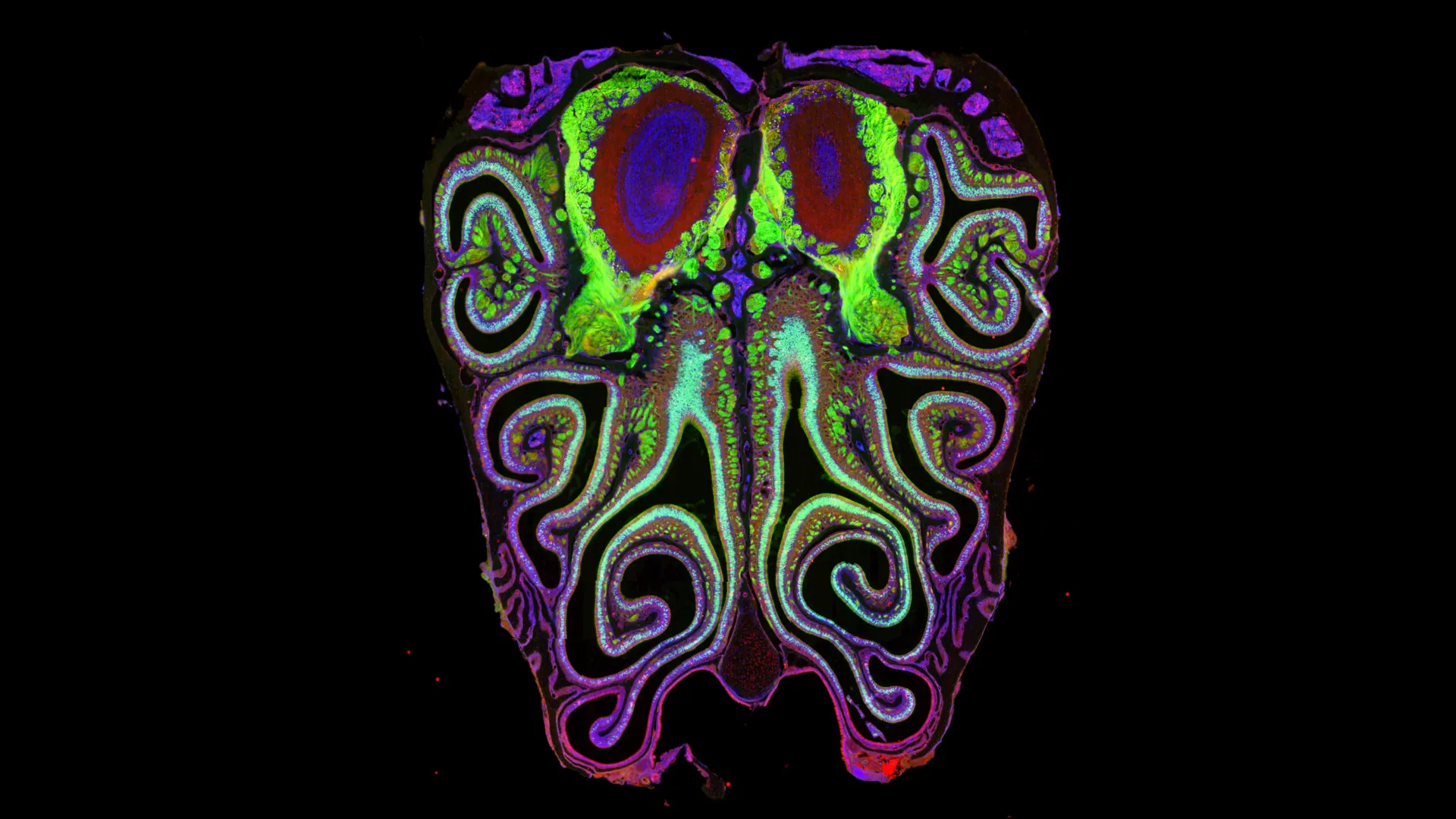
The sheer scale of the system is staggering. A mouse has roughly 20 million olfactory neurons, each expressing only one type of receptor. For years, the prevailing theory was that these neurons were distributed more or less randomly across the olfactory epithelium—the specialized tissue inside the nose. Previous studies suggested the existence of a few broad "zones," but within those zones, the arrangement was thought to be a lottery. The new Harvard study effectively dismantles this theory of randomness, replacing it with a model of strict biological order.
A Chronology of Olfactory Discovery
To understand the magnitude of this breakthrough, it is necessary to look at the timeline of olfactory research. The modern era of smell science began in 1991, when Richard Axel and Linda Buck identified the large family of genes that encode olfactory receptors. This discovery, which earned them the Nobel Prize in 2004, revealed that the olfactory system is one of the largest gene families in the mammalian genome.
Following the 1991 discovery, the scientific community spent the next thirty years attempting to visualize how these receptors were laid out. Early mapping efforts in the late 1990s and early 2000s were limited by the technology of the time. Researchers could only track a few receptors at once, leading to the "zone" hypothesis—the idea that certain receptors lived in the top of the nose and others in the bottom, but without fine-grained organization.
The turning point occurred over the last five years as genomic tools became sufficiently powerful to handle the data required for a full-scale map. Datta’s team spent years refining a combination of single-cell sequencing and spatial transcriptomics to bridge the gap between identifying a cell’s function and knowing its exact physical location. The publication of their findings on April 28 represents the culmination of this multi-decade search for order in the nasal cavity.
Methodology: Mapping 5.5 Million Neurons
The scale of the study is unprecedented in the field of neuroscience. The researchers analyzed approximately 5.5 million neurons across more than 300 mice. To manage this massive dataset, they employed two primary technologies: single-cell RNA sequencing and spatial transcriptomics.
Single-cell sequencing allowed the team to look at individual neurons and determine which of the 1,000+ receptor types each one was expressing. However, sequencing alone strips away the physical context of the cell. To solve this, they used spatial transcriptomics, a method that allows researchers to pinpoint exactly where those specific gene expressions are occurring within a slice of tissue.
"This is now arguably the most sequenced neural tissue ever, but we needed that scale of data in order to understand the system," Datta explained. By layering the genetic identity of millions of cells over their physical coordinates, the researchers were able to visualize a pattern that had been hidden in plain sight.
The Discovery of the Horizontal Stripes
The data revealed a striking geometric pattern. Instead of a random distribution, the neurons carrying specific receptor types are organized into a series of overlapping horizontal bands, or stripes. These stripes run from the dorsal (top) to the ventral (bottom) regions of the nose.
Every mouse studied exhibited nearly the identical "striped" map. This consistency is a hallmark of a fundamental biological blueprint. Furthermore, the researchers discovered that this nasal map is not an isolated structure; it aligns perfectly with the map in the olfactory bulb, the part of the brain that first receives scent information. This means that the physical location of a receptor in the nose determines where its signal lands in the brain, creating a direct, organized circuit for sensory data.
"Our results bring order to a system that was previously thought to lack order, which changes conceptually how we think this works," Datta said. This "topographic" organization suggests that the brain uses the physical location of a scent’s detection to help categorize and identify what that scent is.
The Role of Retinoic Acid in Development
A critical component of the study was determining how this complex map forms during development. The researchers identified retinoic acid—a derivative of Vitamin A known to regulate gene activity—as the "architect" of the smell map.
The team discovered a gradient of retinoic acid within the developing nose. High concentrations of the molecule at one end and lower concentrations at the other act as a set of instructions for maturing neurons. Depending on where a neuron is located within this gradient, the retinoic acid influences which olfactory receptor gene it "switches on."
To prove this, the researchers manipulated the levels of retinoic acid in experimental models. When the gradient was altered, the entire receptor map shifted, with stripes moving upward or downward in response to the chemical change. This confirmed that the precise organization of the a thousand different receptors is a programmed developmental feat, ensuring that every animal of a species "sees" the chemical world through the same organized lens.
Validation and Peer Collaboration
The Harvard study’s findings were further bolstered by a separate, independent study led by Catherine Dulac, the Xander University Professor at Harvard. Dulac’s team, working in the Department of Molecular and Cellular Biology, reached consistent conclusions using different methodologies. Both studies were published in the same issue of Cell, providing a powerful consensus within the scientific community that the "random" model of olfaction is officially obsolete.
The alignment of these two studies suggests that the horizontal stripe organization is a robust and fundamental feature of mammalian biology. While the research was conducted in mice, the evolutionary conservation of sensory systems suggests that a similar, albeit slightly different, map likely exists in humans.
Implications for Human Health and Anosmia
The enrichment of our understanding of the olfactory map has profound implications for clinical medicine. Anosmia, or the loss of smell, gained global attention during the COVID-19 pandemic, but it has long been a debilitating condition for millions. Loss of smell is linked to significant safety risks (such as the inability to detect gas leaks or spoiled food), poor nutrition, and higher rates of depression and social isolation.
Current treatments for smell loss are extremely limited, often involving "smell training" with essential oils, which has varying degrees of success. "We cannot fix smell without understanding how it works on a basic level," Datta noted. By identifying the specific "stripes" and the developmental signals (like retinoic acid) that create them, scientists can now explore more targeted therapies.
Potential future applications include:
- Stem Cell Therapies: If scientists know exactly where specific receptors should be located, they could potentially use stem cells to regrow damaged olfactory tissue in the correct "striped" pattern.
- Brain-Computer Interfaces (BCIs): Understanding the map allows for the possibility of electronic implants that could bypass a damaged nose and stimulate the olfactory bulb directly, using the map as a guide to "code" different scents.
- Diagnostic Tools: The map could serve as a baseline to understand how neurodegenerative diseases like Alzheimer’s or Parkinson’s—which often present with smell loss as an early symptom—affect the olfactory system.
A New Era for Sensory Science
The mapping of the olfactory system represents the closing of a major chapter in sensory biology. We now understand that the nose is not merely a chemical detector but a sophisticated spatial processor. This research provides the "GPS" for the olfactory system, allowing future scientists to navigate the complex pathways that turn a physical molecule into a vivid memory or a sharp warning.
As the team at Harvard continues to investigate whether these horizontal stripes are arranged in a specific functional order—such as grouping "fruity" or "burnt" smells together—the broader scientific community is already looking at how this knowledge can be applied to restore one of the most vital, yet overlooked, human experiences. "Smell has a really profound and pervasive effect on human health," Datta concluded. "Without understanding this map, we’re doomed to fail in developing new treatments." With the map now in hand, the path to those treatments has never been clearer.














Leave a Reply