In a landmark study that bridges the gap between physical mechanobiology and molecular pharmacology, researchers from the LKS Faculty of Medicine at the University of Hong Kong (HKUMed) have successfully identified a critical biological "exercise sensor" within the human skeletal system. This discovery, centered on a specific protein known as Piezo1, provides a long-awaited explanation for how the human body translates physical movement into bone density and strength. The research, published in the prestigious international journal Signal Transduction and Targeted Therapy, offers a transformative perspective on treating osteoporosis, particularly for aging populations and patients whose physical limitations prevent them from engaging in traditional exercise.
The study was spearheaded by the Department of Medicine at the School of Clinical Medicine, HKUMed, in collaboration with international experts from the French National Centre for Scientific Research (CNRS). By decoding the molecular signals that bones use to "feel" exercise, the team has laid the groundwork for a new class of pharmaceuticals known as "exercise mimetics"—medications designed to trick the body into producing the benefits of physical activity through chemical activation.
The Biological Mechanism: How Bones "Feel" Movement
At the heart of the discovery is the Piezo1 protein, located on the surface of mesenchymal stem cells (MSCs) within the bone marrow. These stem cells are essentially the body’s internal "building blocks," capable of differentiating into various types of tissue, including bone-forming cells (osteoblasts) or fat-storing cells (adipocytes).
Under normal conditions of physical activity, the mechanical stress placed on the skeleton—such as the impact of walking, running, or lifting—activates the Piezo1 protein. The research team found that Piezo1 acts as a gatekeeper for these stem cells. When physical force is detected, Piezo1 triggers a signaling pathway that instructs the stem cells to transform into bone tissue. Simultaneously, this activation suppresses the production of fat cells within the marrow.
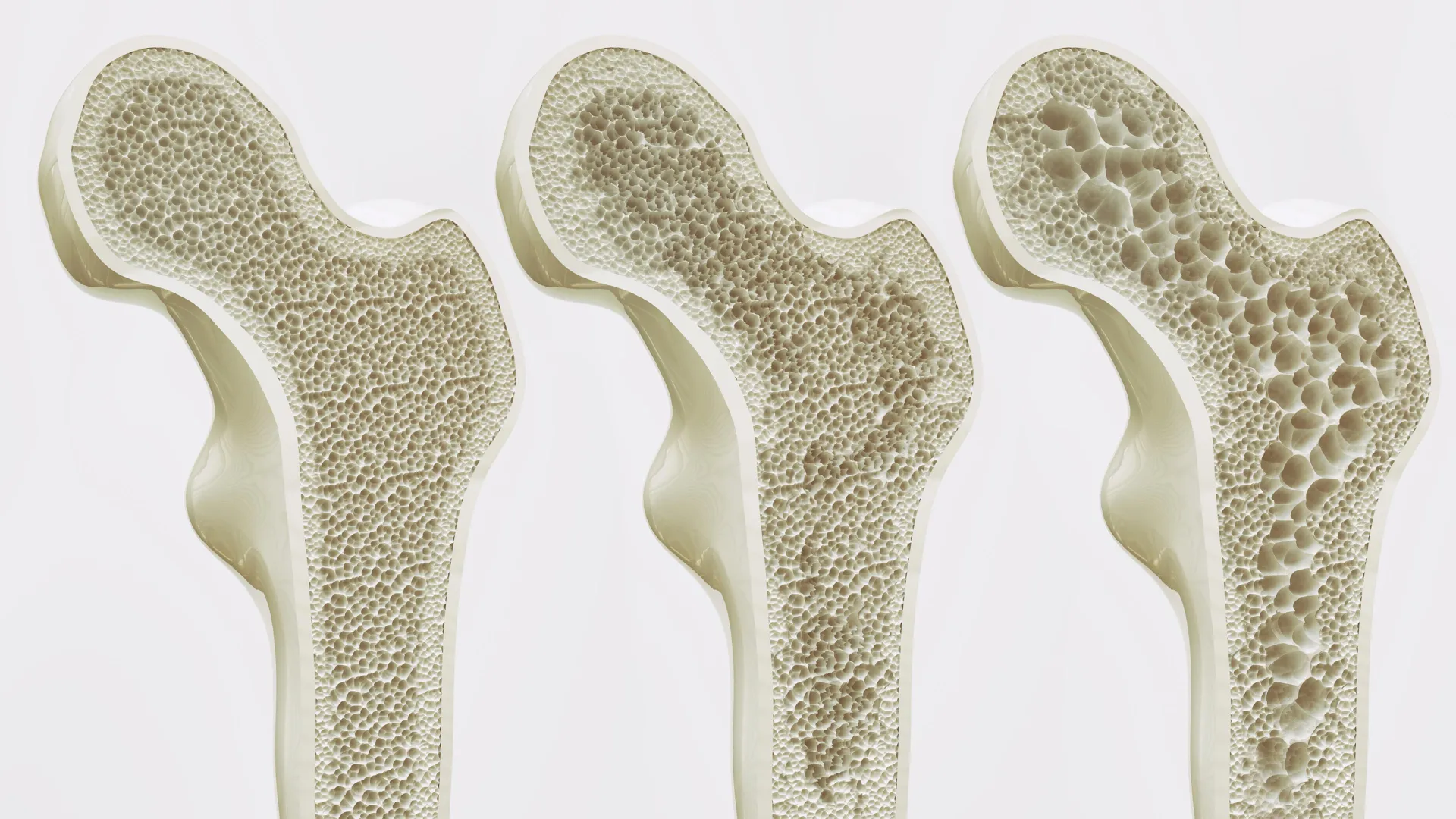
However, the researchers observed a starkly different outcome when Piezo1 was absent or inactive. In laboratory mouse models and human stem cell cultures, the deficiency of this protein led to a dramatic shift in cellular behavior. Without the mechanical "instruction" provided by Piezo1, the stem cells defaulted to producing fat cells rather than bone. This shift not only reduces bone density but also leads to the accumulation of marrow fat, which further degrades the structural integrity of the skeleton.
The Bone-Fat Balance and the Inflammatory Cycle
The study provides a detailed look at the "bone-fat switch," a phenomenon that has long puzzled geriatricians. As humans age, their bone marrow naturally begins to host more fat and less healthy bone tissue. The HKUMed study identified that the lack of Piezo1 activation triggers the release of specific inflammatory signals, namely Ccl2 and lipocalin-2.
These inflammatory markers create a hostile microenvironment within the bone marrow. They actively push mesenchymal stem cells toward fat production while inhibiting the growth of new bone. This creates a vicious cycle: as bones become more porous and fatty, they become less capable of supporting physical movement, which in turn leads to even less Piezo1 activation and more bone loss. By demonstrating that blocking these inflammatory signals can help restore bone health, the researchers have provided a secondary target for future therapeutic interventions.
Global and Local Context: The Rising Crisis of Osteoporosis
The implications of this research are underscored by the staggering global prevalence of osteoporosis. According to data from the World Health Organization (WHO), osteoporosis is a "silent epidemic" that affects millions. Statistics indicate that approximately one in three women and one in five men over the age of 50 will suffer a fracture due to weakened bones.
In Hong Kong, the situation is particularly acute due to the city’s rapidly aging demographic. Current data suggests that osteoporosis affects 45% of women and 13% of men aged 65 and older in the territory. These fractures are not merely physical injuries; they are life-altering events. A hip fracture in an elderly patient often leads to a permanent loss of independence, with a significant percentage of patients requiring long-term nursing care or facing increased mortality rates within the first year following the injury.
The economic burden is equally significant. Healthcare systems worldwide spend billions of dollars annually on the treatment and rehabilitation of osteoporotic fractures. As the "silver tsunami" of aging populations continues to grow, the demand for treatments that can prevent bone loss before fractures occur has never been higher.
Chronology of the Discovery and Research Methodology
The journey toward identifying Piezo1 began with the observation that bedridden patients and astronauts in microgravity environments experience rapid and severe bone loss. This suggested that the absence of mechanical load was the primary driver of skeletal degradation.
- Initial Phase: The team, led by Professor Xu Aimin, focused on the behavior of mesenchymal stem cells. They sought to identify the specific "mechanosensor" that allows these cells to detect gravity and physical pressure.
- Identification of Piezo1: Using advanced genetic screening and protein analysis, the researchers narrowed down the candidates to the Piezo family of proteins, which are known to be sensitive to mechanical force in other organs, such as the lungs and blood vessels.
- Experimental Validation: The team utilized mouse models where Piezo1 was specifically deleted from bone marrow stem cells. These mice exhibited accelerated bone loss and high levels of marrow fat, mirroring the conditions seen in elderly human patients.
- Human Stem Cell Testing: To ensure the findings were applicable to humans, the researchers conducted parallel experiments on human mesenchymal stem cells, confirming that the Piezo1 signaling pathway operates similarly across species.
- Synthesis of Findings: The final phase involved identifying the downstream signals (Ccl2 and lipocalin-2) and testing whether pharmacological intervention could replicate the effects of Piezo1 activation.
Expert Insights: Moving Toward "Exercise in a Pill"
The lead researchers emphasize that this discovery is not about replacing exercise for healthy individuals, but rather providing a lifeline for those who physically cannot move.
Professor Xu Aimin, Director of the State Key Laboratory of Pharmaceutical Biotechnology and Chair Professor at HKUMed, noted that current treatments for bone loss are often reactive rather than proactive. "We need to understand how our bones get stronger when we move before we can find a way to replicate those benefits at the molecular level," Professor Xu stated. "This study is a critical step toward that goal, identifying the exact sensor that turns movement into medicine."
Dr. Wang Baile, Research Assistant Professor at HKUMed and co-leader of the study, highlighted the potential for "exercise mimetics." These drugs would chemically stimulate the Piezo1 pathway, effectively "tricking" the bone marrow into believing the body is engaging in vigorous physical activity. This would be a breakthrough for patients with paralysis, severe frailty, or those recovering from major surgery.
Professor Eric Honoré, a collaborator from the French National Centre for Scientific Research, suggested that the impact could extend even further. "This offers a promising strategy beyond traditional physical therapy," he said. "By targeting the Piezo1 pathway, we could potentially provide the biological benefits of exercise to the most vulnerable groups, substantially reducing their risk of fractures and improving their overall quality of life."
Broader Implications and Analysis
The identification of Piezo1 has implications that reach beyond osteoporosis. The "exercise mimetic" concept could theoretically be applied to other systems affected by inactivity. For example, muscle atrophy and cardiovascular decline are also common in bedridden patients. If a similar mechanical sensor exists in muscle tissue, researchers might eventually develop a suite of mimetics that maintain entire body systems during periods of forced immobility.
Furthermore, this research has significant value for the future of space exploration. Astronauts on long-duration missions to Mars or the Moon face significant bone density loss due to the lack of gravity. Understanding the Piezo1 pathway could lead to the development of supplements that protect the skeletal health of explorers in zero-gravity environments.
From a clinical perspective, the next challenge lies in the development of a compound that can safely and effectively activate Piezo1 in humans without causing off-target effects in other tissues where Piezo1 is present. The HKUMed team is currently focused on translating these laboratory findings into clinical trials, a process that will involve rigorous testing of safety and efficacy.
Conclusion and Future Directions
The collaborative effort between HKUMed and their international partners represents a significant milestone in the field of endocrinology and metabolism. By pinpointing Piezo1 as the bone’s internal exercise sensor, the team has moved the scientific community closer to a future where bone loss is no longer an inevitable consequence of aging or illness.
The study received support from a wide array of prestigious institutions, including the Research Grants Council of Hong Kong, the National Natural Science Foundation of China, and the French National Research Agency. This broad base of support reflects the international importance of the work. As the research moves into its next phase, the goal remains clear: to develop therapies that preserve independence, reduce the burden on healthcare systems, and allow the aging population to live longer, stronger lives.














Leave a Reply