Plants, through the remarkable process of photosynthesis, harness sunlight to fuel their existence for the majority of their lives. However, a critical, albeit brief, period exists when this vital capability is not yet online. In the nascent stages following seed germination, young plants are entirely reliant on stored fatty acids for their energy needs. This fundamental dependency underscores the importance of understanding the cellular machinery that facilitates this transition. Central to this process is the peroxisome, a sophisticated, membrane-bound organelle found not only in plants but also in human cells, playing a pivotal role in the breakdown of fatty acids. The unique characteristics of plant cells, particularly their size and the visibility of their peroxisomes, have positioned them as an invaluable model system for unraveling the intricate workings of these essential cellular compartments.
Dr. Bonnie Bartel, the Ralph and Dorothy Looney Professor of Biosciences, highlights the advantages of employing the model plant Arabidopsis thaliana in this research. "The plant we use, Arabidopsis, has large cells and peroxisomes so large that we can see inside them with a light microscope," Dr. Bartel explained. "The peroxisome gets even larger during the seed to seedling stage, when the plant is relying on fatty acids for energy, before shrinking back down to its normal size once the plant can photosynthesize." This dynamic fluctuation in peroxisome size directly correlates with the plant’s metabolic state, offering a clear visual indicator of cellular activity during a critical developmental window.
The Pivotal Role of Protein PEX11 in Peroxisome Dynamics
The research team, led by Dr. Bartel, has dedicated significant effort to investigating these enlarged peroxisomes, with a particular focus on a protein designated as PEX11. For an extended period, the scientific community has recognized PEX11’s involvement in the division of peroxisomes. However, groundbreaking new research, recently published in the esteemed journal Nature Communications, has unveiled a previously unrecognized function for PEX11: its crucial role in regulating the expansion and contraction of peroxisomes during the initial phases of plant development.
Nathan Tharp, the lead author of the study and a doctoral candidate at Rice University, emphasized the broader significance of understanding peroxisome function. "Peroxisomes are implicated in some human diseases and used in bioengineering," Tharp stated. "They can, however, be rather tricky to study." This inherent complexity underscores the scientific challenge and the potential impact of their findings.
Navigating the Complexity of PEX11 Gene Regulation with CRISPR Technology
Elucidating the precise function of a protein often involves a standard scientific approach: disabling the gene responsible for its synthesis and observing the resulting phenotypic changes. In the case of PEX11, this endeavor presented a significant hurdle. The PEX11 protein is not encoded by a single gene but rather by a family of five distinct genes. Initial attempts to disrupt individual genes yielded minimal observable effects on peroxisome behavior. Conversely, the complete elimination of all five PEX11 genes proved lethal to the plant, preventing any meaningful analysis of the protein’s specific functions. This situation created a complex puzzle, making it exceedingly difficult to isolate and pinpoint PEX11’s exact role.
To overcome this intricate genetic landscape, Tharp employed advanced CRISPR-Cas9 gene-editing technology. This innovative approach allowed for the precise and selective inactivation of various combinations of the five PEX11 genes, enabling the researchers to meticulously investigate the impact of partial gene disruptions. "I was able to use recent advances in CRISPR to go in and break specific combinations of the five genes," Tharp elaborated. "It was only then that we were able to see that PEX11 is clearly involved in controlling the growth of the peroxisome during the seed to seedling stage." This strategic application of cutting-edge technology was instrumental in dissecting the multifaceted genetic control of peroxisome size.
Giant Peroxisomes: A Window into Peroxisome Growth Regulation
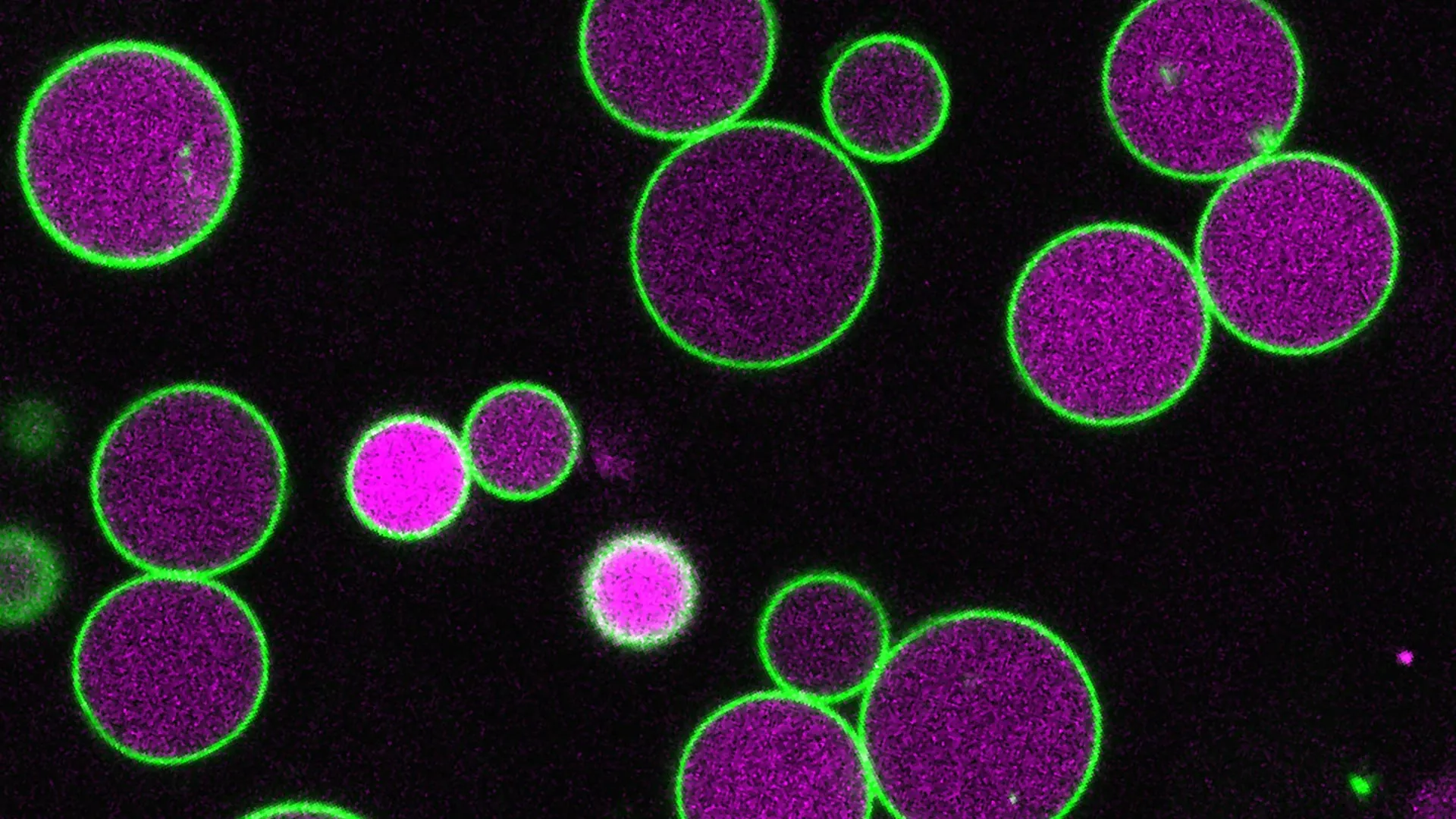
The experimental manipulation of PEX11 gene combinations led to the creation of two distinct lines of mutant plants. In both mutant strains, peroxisomes exhibited the expected enlargement during the critical seed-to-seedling transition, consistent with the plant’s reliance on stored fatty acids. However, a striking deviation from normal development was observed: instead of returning to their typical, smaller size, a subset of these peroxisomes continued to expand, exceeding normal size limits by a significant margin. In some instances, these abnormally enlarged peroxisomes stretched across the entire width of the plant cell, presenting a visually dramatic phenotype.
Further examination of these mutant cells revealed another crucial deficiency: a marked absence of vesicles, which are small, membrane-bound structures typically formed within the peroxisome during the processing of fatty acids. Under normal physiological conditions, these vesicles are thought to play a role in managing peroxisome growth. They form as the peroxisome expands, and their formation appears to be associated with the removal of portions of the peroxisome’s outer membrane.
Tharp’s observations suggest a direct link between vesicle formation and peroxisome size control. "The vesicles taking pieces of membrane as they form may help control the peroxisome’s growth," he theorized. "In our PEX11 mutants, these vesicles either don’t form or are abnormally small and rare, and so we see these massive peroxisomes, way larger than normal." This finding indicates that PEX11, through its influence on vesicle dynamics, plays a critical regulatory role in preventing uncontrolled peroxisome expansion.
Implications Beyond Plants: A Conserved Evolutionary Mechanism
While the primary focus of this research was on plant physiology, Tharp’s scientific curiosity extended to exploring whether the identified growth control mechanism might be conserved across different organisms. To test this hypothesis, he introduced the yeast homolog of PEX11, known as Pex11, into the mutant plant cells. The results of this cross-species experiment were compelling. "We put yeast Pex11 into our mutant plant cells to see if it could return the peroxisomes back to normal," Tharp reported. "And it did."
This remarkable outcome strongly suggests that the protein Pex11 performs a functionally equivalent role in yeast as PEX11 does in plants, despite the vast evolutionary divergence between these two kingdoms of life. This evolutionary conservation implies that the protein may also exert a comparable regulatory influence in other cell types, including human cells.
Dr. Bartel underscored the broader significance of this finding. "Finding that this protein fills the same role in yeast and plant cells suggests that it may be a highly conserved protein," she stated. "Our findings in plants, in this relatively easy-to-study model, may thus be applicable to human cells and cells used for bioengineering." The implications of this discovery are far-reaching, potentially paving the way for novel therapeutic strategies for peroxisome-related human diseases and advancements in bioengineering applications.
Background Context and Timeline of Research
The study of peroxisomes has a rich history, dating back to their initial identification in the mid-20th century. Early research focused on their role in lipid metabolism and detoxification. Over time, their involvement in a wider array of cellular processes, including plant development, has become increasingly apparent. The discovery of the PEX gene family, which encodes proteins essential for peroxisome biogenesis and function, marked a significant turning point in the field.
The specific research leading to the Nature Communications publication likely spanned several years. The initial identification of PEX11’s role in peroxisome division would have been a precursor to investigating its more nuanced functions. The development and refinement of CRISPR-based techniques in the last decade have been crucial for overcoming the genetic complexities of the PEX11 gene family. The timeline would have involved:
- Early Stages: Characterization of peroxisomes and their role in fatty acid metabolism in plants. Identification of the PEX gene family and initial understanding of PEX11 in peroxisome division.
- Mid-Stages: Recognition of the dynamic changes in peroxisome size during early plant development. Challenges in dissecting the function of multi-gene protein families like PEX11.
- Advanced Stages: Application of advanced CRISPR technologies to enable precise gene editing. Experimental generation of mutant lines with specific PEX11 gene combinations. Observation and detailed analysis of peroxisome morphology and vesicle dynamics in mutant plants.
- Cross-Species Validation: Introduction of yeast Pex11 into mutant plant cells to assess functional conservation.
- Publication: Dissemination of findings through peer-reviewed publication in Nature Communications.
Broader Impact and Implications
The research on PEX11 and peroxisome growth control in Arabidopsis holds significant implications across multiple scientific disciplines.
Human Health and Disease:
Peroxisomal disorders, such as Zellweger syndrome and X-linked adrenoleukodystrophy, are serious genetic conditions that can lead to severe developmental abnormalities and neurological impairments. Understanding the fundamental mechanisms of peroxisome biogenesis and function, as illuminated by this study, is crucial for developing diagnostic tools and therapeutic interventions for these debilitating diseases. The conservation of PEX11’s function across species suggests that insights gained from plant models may directly translate to a better understanding of peroxisome-related pathology in humans. Future research could explore whether manipulating PEX11 or related pathways could offer novel therapeutic avenues for patients with peroxisomal disorders.
Biotechnology and Bioengineering:
Peroxisomes are increasingly recognized for their potential in various bioengineering applications. Their metabolic capabilities, including the breakdown of lipids and the synthesis of certain molecules, make them attractive targets for engineered cellular factories. For instance, peroxisomes have been engineered to produce biofuels or to break down pollutants. A deeper understanding of how to control peroxisome size and dynamics, as provided by this research, could lead to more efficient and robust engineered peroxisomes for industrial and environmental applications. The ability to precisely modulate peroxisome size could allow for the optimization of intracellular space and metabolic flux within engineered cells.
Plant Biology and Agriculture:
While this study focused on a fundamental developmental process, its findings could have indirect implications for agriculture. Enhancing seed oil mobilization efficiency could be crucial for improving crop yields or developing plants with altered nutritional profiles. Understanding the intricate regulation of peroxisome function during germination could offer insights into strategies for optimizing seed viability and seedling establishment under challenging environmental conditions. Further research might explore whether genetic modifications affecting PEX11 or related pathways could lead to improved crop performance.
Fundamental Cell Biology:
This research contributes to the broader understanding of organelle biogenesis and regulation, a cornerstone of cell biology. The dynamic nature of peroxisomes, their ability to grow and shrink in response to metabolic cues, and the intricate interplay between protein function and vesicle formation provide a model for studying how cells maintain cellular homeostasis and adapt to changing conditions. The discovery of a conserved regulatory mechanism for peroxisome size control across diverse eukaryotes highlights the power of comparative biology and the potential for drawing universal principles from model organisms.
In conclusion, the research conducted by Dr. Bartel’s team at Rice University represents a significant advancement in our understanding of peroxisome biology. By leveraging the advantages of plant model systems and employing cutting-edge genetic technologies, they have elucidated a critical mechanism governing peroxisome growth during early plant development. The implications of this work extend far beyond the realm of plant science, offering profound insights into human health, biotechnology, and the fundamental principles of cellular organization. The ongoing exploration of these cellular powerhouses promises to unlock further secrets with potentially transformative applications.











Leave a Reply