A groundbreaking international collaboration between the University of Queensland and the University of Minnesota has unveiled a significant biological breakthrough that could redefine the medical community’s understanding of Major Depressive Disorder (MDD). By examining the "energy currency" of the human body, researchers have identified a distinct cellular energy pattern in young adults suffering from depression, offering a potential pathway for earlier diagnosis and more personalized treatment strategies. The study, published in the prestigious journal Translational Psychiatry, focuses on adenosine triphosphate (ATP), a molecule essential for storing and transferring energy within cells, and its role in the manifestation of depressive symptoms.
For decades, the diagnosis of depression has relied heavily on subjective clinical assessments and patient-reported symptoms, such as persistent sadness, loss of interest, and chronic fatigue. However, this new research shifts the focus toward objective biological markers. Scientists from the University of Queensland’s (UQ) Queensland Brain Institute (QBI) and the University of Minnesota conducted a comprehensive analysis of ATP levels in both the brain and blood cells of individuals aged 18 to 25. This specific age demographic is considered critical by mental health professionals, as it represents a peak period for the onset of major psychiatric disorders and a vital window for early intervention.
The Science of Cellular Energy in Mental Health
At the core of this discovery is the mitochondrion, often referred to as the "powerhouse" of the cell. Mitochondria are responsible for producing ATP through a process known as cellular respiration. In a healthy physiological state, mitochondria adjust ATP production based on the body’s demands—increasing output during periods of physical or psychological stress and maintaining a baseline during rest.
The research team, led by Associate Professor Susannah Tye of QBI and Dr. Katie Cullen of the University of Minnesota, utilized advanced imaging techniques to measure these levels in real-time. The study employed specialized magnetic resonance spectroscopy (MRS) to observe ATP production in the brain, while simultaneously analyzing blood samples to see if these energy patterns were mirrored systemically.
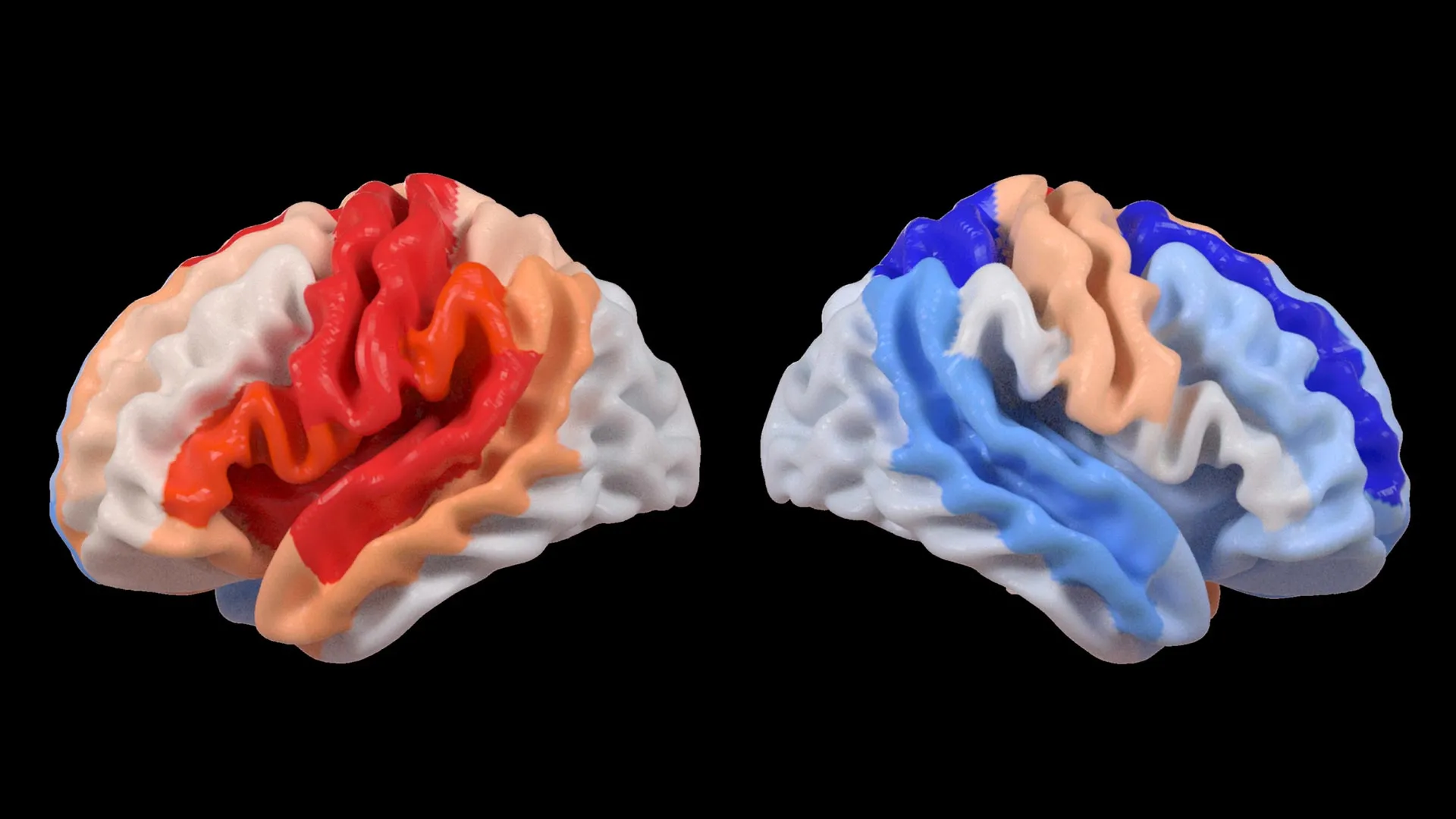
The findings revealed a counterintuitive and surprising "energy signature" among participants with MDD. Unlike healthy control subjects, those with depression exhibited abnormally high levels of ATP while at rest. However, when these same cells were required to respond to increased demand or stress, they demonstrated a significantly reduced capacity to boost energy production. This suggests a state of cellular exhaustion or "overworking" that occurs early in the progression of the illness.
Methodology and Study Design
The study involved a cohort of 18 participants, all within the 18-to-25 age bracket, who had received a formal diagnosis of Major Depressive Disorder. This group was compared against a control group of healthy individuals with no history of mental health conditions. The University of Minnesota team spearheaded the clinical data collection, gathering high-resolution brain scans and peripheral blood samples.
The technological backbone of the study involved an imaging method developed by Professors Xiao Hong Zhu and Wei Chen. This technique allowed researchers to non-invasively quantify the rate of ATP synthesis in the living human brain. By pairing this with cellular analysis of blood samples, the researchers were able to confirm that the energy disruptions were not localized solely in the brain but were a systemic issue affecting the entire body.
Dr. Roger Varela, a researcher at QBI, noted that the observation of high resting ATP was particularly unexpected. In many chronic illnesses, researchers typically look for a deficit in energy. The presence of elevated energy molecules during periods of inactivity suggests that the biological systems of young people with depression are under constant strain, potentially leading to long-term mitochondrial wear and tear.
A New Understanding of Fatigue and Motivation
One of the most profound implications of the study relates to the symptom of fatigue. In the context of MDD, fatigue is often one of the most debilitating symptoms and is notoriously difficult to treat with standard antidepressants, such as Selective Serotonin Reuptake Inhibitors (SSRIs).
"Fatigue is a common and hard-to-treat symptom of MDD, and it can take years for people to find the right treatment for the illness," Associate Professor Susannah Tye explained. She noted that the lack of progress in developing new treatments has largely been due to a lack of fundamental research into the biological drivers of these symptoms.
The study’s findings suggest that the lethargy and lack of motivation (anhedonia) experienced by patients are not merely psychological states but are rooted in a physical inability of the cells to meet energy demands. When the brain and body cannot "ramp up" energy production to meet the challenges of daily life, the result is a systemic slowdown that manifests as low mood, cognitive fog, and physical exhaustion. This biological "bottleneck" explains why patients may feel "tired but wired"—their systems are working overtime at rest but have no reserve left for actual activity.
Chronology of Research and Collaborative Efforts
The partnership between the University of Queensland and the University of Minnesota represents a multi-year effort to bridge the gap between neuroimaging and molecular biology. The chronology of the research began with the development of the ATP-measuring imaging protocols in Minnesota, which were then applied to clinical populations of young adults.
Once the initial brain scan data suggested anomalies in energy metabolism, the researchers expanded the scope to include blood-based biomarkers. This move was strategic; while brain scans are expensive and require specialized equipment, blood tests are far more accessible in a primary care setting. By proving that the ATP patterns in the blood reflect those in the brain, the team has opened the door for the development of a simple diagnostic blood test for depression.
The study moved into its final analysis phase at the Queensland Brain Institute, where Dr. Varela and his colleagues synthesized the data to confirm the "overworking" hypothesis. The publication of their findings in Translational Psychiatry marks the culmination of this phase and the beginning of a broader discussion regarding metabolic psychiatry.
Implications for Treatment and the Reduction of Stigma
The shift toward a metabolic understanding of depression carries significant weight in the effort to reduce the social stigma surrounding mental health. For decades, depression has been stigmatized as a "weakness of character" or a purely emotional struggle. By demonstrating that depression involves measurable, physical changes in how cells process energy, this research reinforces the fact that MDD is a systemic biological illness.
"This shows multiple changes occur in the body, including in the brain and the blood, and that depression impacts energy at a cellular level," Dr. Varela stated. He emphasized that the research proves not all depression is the same. Each patient possesses a unique biological profile, and the way their mitochondria function—or malfunction—can dictate which treatments will be most effective.
This discovery paves the way for "precision psychiatry." Currently, many patients undergo a "trial and error" process with medications, often taking months or years to find a drug that works. If clinicians can identify a patient’s specific energy signature through blood work or imaging early on, they could potentially prescribe metabolic interventions or targeted therapies that address mitochondrial health directly, rather than relying solely on neurotransmitter modulation.
Future Directions and Economic Impact
The broader implications of this research extend to the economic and social sectors. Major Depressive Disorder is a leading cause of disability worldwide, costing the global economy billions of dollars annually in lost productivity and healthcare expenses. By focusing on the 18-to-25 demographic, this research targets the very group that is most likely to suffer long-term economic consequences if their illness remains untreated or poorly managed.
Early intervention is the key to preventing the "scarring" effect of chronic depression, where repeated episodes lead to permanent changes in brain structure and function. If the energy-related markers identified in this study can be used to catch depression in its nascent stages, the chances of full recovery are significantly higher.
Looking forward, the research team hopes to expand the study to larger and more diverse populations to determine if these ATP patterns hold true across different age groups and subtypes of depression. There is also interest in exploring how existing treatments, such as exercise, diet, and certain medications, affect these cellular energy levels.
The integration of metabolic science into psychiatry represents a new frontier. As researchers continue to unravel the complex relationship between the mind and the body’s most basic energy processes, the hope is that depression will one day be treated with the same biological precision as diabetes or heart disease. For the millions of young people struggling with the weight of MDD, this "energy currency" breakthrough offers more than just data—it offers a roadmap to a more effective and compassionate form of care.













Leave a Reply