The fundamental relationship between restorative sleep and physical health has long been recognized by the medical community, yet the specific neurological mechanisms governing this connection have remained largely shrouded in mystery. In a landmark study published in the journal Cell, researchers at the University of California, Berkeley, have successfully mapped the intricate brain circuits responsible for the release of growth hormone during sleep. This discovery provides the first comprehensive look at a complex feedback system that balances hormonal surges with the brain’s transition between sleep and wakefulness, offering a potential paradigm shift in how clinicians approach sleep disorders, metabolic diseases, and neurodegenerative conditions.
For decades, endocrinologists and sleep scientists have observed that growth hormone (GH) levels peak during the early stages of deep, non-rapid eye movement (non-REM) sleep. This hormone is the primary driver of physical repair, facilitating muscle protein synthesis, bone density maintenance, and the regulation of glucose and fatty acid metabolism. However, the "black box" of the brain’s hypothalamus—the region responsible for these signals—prevented researchers from understanding exactly why poor sleep quality leads to a precipitous drop in GH production. The UC Berkeley team, led by postdoctoral fellow Xinlu Ding and Professor Yang Dan, has finally identified the specific neurons and feedback loops that coordinate this vital biological process.
The Architecture of the Hypothalamic Circuit
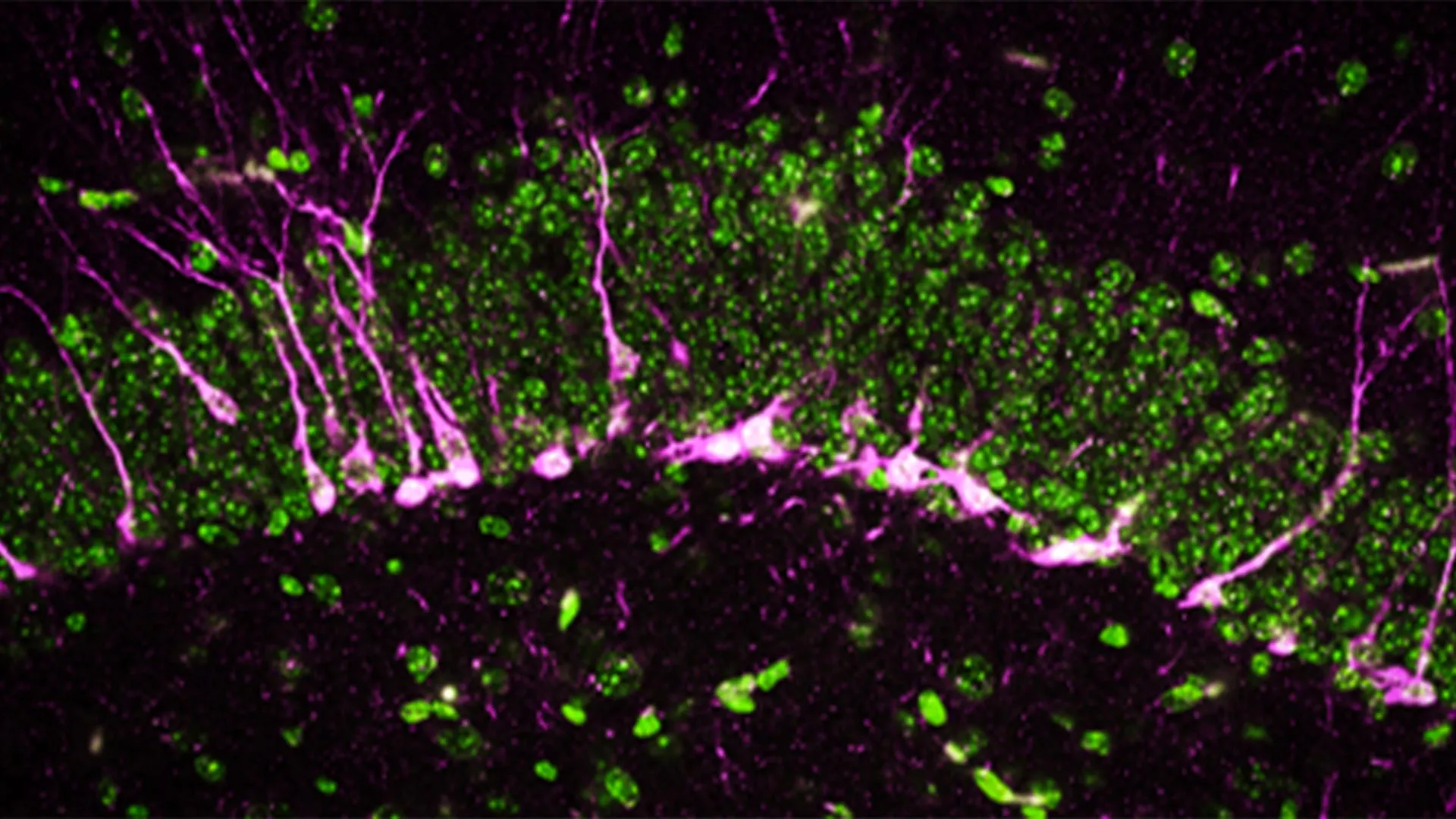
At the heart of the discovery is the hypothalamus, a primitive and essential structure buried deep within the mammalian brain. The research team identified two distinct populations of neurons that act as the "gas" and "brake" pedals for growth hormone. The first group releases growth hormone-releasing hormone (GHRH), which signals the pituitary gland to flood the bloodstream with GH. The second group releases somatostatin, a potent inhibitor that shuts down GH production.
While the existence of these hormones was known, their real-time behavior during the different stages of sleep had never been visualized in living subjects. Using advanced optogenetic techniques and electrode recordings in mice, the researchers observed that these neurons do not function in isolation. Instead, they engage in a highly coordinated dance that changes rhythm as the subject moves from light sleep to deep non-REM sleep and eventually into REM (dream) sleep.
The study revealed that during REM sleep, both GHRH and somatostatin activity increases, creating a surge in GH levels. Conversely, during non-REM sleep, somatostatin activity drops significantly while GHRH rises modestly, creating a steady but different pattern of hormone secretion. This nuanced understanding suggests that every stage of sleep plays a specific, non-redundant role in maintaining the body’s hormonal equilibrium.
A New Chronology of Sleep Research
The history of sleep-hormone research dates back to the late 1960s, when scientists first used primitive blood sampling techniques to notice that GH levels spiked shortly after the onset of sleep. For fifty years, the standard method for studying this phenomenon involved drawing blood at 10-to-20-minute intervals throughout the night—a process that often disturbed the very sleep patterns being studied.
The UC Berkeley study represents a technological leap forward. By recording neural activity directly at the source in the brain, the researchers moved past the limitations of peripheral blood testing. This shift in methodology allowed the team to discover a previously unknown feedback loop involving the locus coeruleus, a region in the brainstem. The locus coeruleus is the brain’s primary source of norepinephrine, the chemical responsible for alertness, focus, and the "fight or flight" response.
The researchers found that as growth hormone builds up in the system, it actually feeds back into the locus coeruleus. Initially, this feedback helps maintain a state of sleep, but as GH levels reach a certain threshold, they begin to stimulate the locus coeruleus, nudging the brain toward wakefulness. This suggests that growth hormone acts not just as a repair agent for the body, but as a "sleep timer" for the brain.
Supporting Data: The High Cost of Sleep Deprivation
The implications of this neural circuit extend far beyond basic biology, touching upon a growing public health crisis. According to data from the Centers for Disease Control and Prevention (CDC), approximately one-third of American adults report getting less than the recommended seven to nine hours of sleep per night. The UC Berkeley study provides a physiological explanation for the statistical link between chronic sleep deprivation and metabolic syndrome.
Growth hormone is a critical regulator of insulin sensitivity and lipolysis (the breakdown of fats). When the hypothalamic circuit is disrupted by fragmented sleep, GH levels plummet, leading to:
- Increased Adiposity: Reduced GH prevents the body from efficiently burning fat, contributing to obesity.
- Insulin Resistance: Low GH levels interfere with glucose metabolism, significantly raising the risk of Type 2 diabetes.
- Muscle Atrophy and Bone Loss: Without the nocturnal surge of GH, the body’s ability to repair tissues is compromised, a factor especially critical for the elderly and developing teenagers.
Data from the study indicates that even minor disruptions in the non-REM stage can lead to a disproportionate drop in GH output. For teenagers, who require GH for longitudinal bone growth, the "height potential" mentioned in the study’s findings is directly tied to the efficiency of this hypothalamic circuit.
Official Responses and Clinical Perspectives
The research has drawn significant attention from the broader scientific community. Study first author Xinlu Ding emphasized the shift from observation to intervention. "People know that growth hormone release is tightly related to sleep, but only through drawing blood and checking growth hormone levels during sleep," Ding stated. "We’re actually directly recording neural activity… to see what’s going on. We are providing a basic circuit to work on in the future to develop different treatments."
Daniel Silverman, a postdoctoral fellow and study co-author, highlighted the potential for targeted therapies. "This circuit could be a novel handle to try to dial back the excitability of the locus coeruleus," Silverman noted. He suggested that future gene therapies could target specific cell types within this circuit to restore balance in patients with chronic insomnia or metabolic disorders.
Professor Yang Dan, who holds the Pivotal Life Sciences Chancellor’s Chair in Neuroscience at UC Berkeley, oversaw the research. The team’s work was supported by the Howard Hughes Medical Institute (HHMI), reflecting the high level of institutional confidence in the study’s potential to reshape endocrinology.
Broader Impact: From Metabolism to Neurodegeneration
One of the most provocative findings of the study is the link between the GH circuit and the locus coeruleus, an area of the brain that is among the first to show signs of degeneration in Alzheimer’s and Parkinson’s diseases. By identifying how growth hormone regulates the excitability of this region, the UC Berkeley team has opened a new door for neurology.
In patients with neurodegenerative diseases, sleep fragmentation is a common and debilitating symptom. If growth hormone feedback is essential for the health of the locus coeruleus, then chronic GH deficiency caused by poor sleep may accelerate cognitive decline. Conversely, therapies designed to stabilize the GHRH-somatostatin circuit could potentially protect the brainstem from the "over-excitation" that leads to neuronal death in dementia patients.
Furthermore, the study sheds light on the "brain fog" often associated with poor sleep. Because growth hormone influences the locus coeruleus—the center of attention and cognitive function—a lack of GH doesn’t just make the body feel sluggish; it directly impairs the brain’s ability to achieve a state of high-functioning arousal upon waking. As Ding noted, growth hormone may provide "cognitive benefits, promoting your overall arousal level when you wake up."
Future Directions in Sleep Medicine
The discovery of this "tightly balanced system" marks the beginning of a new era in sleep medicine. Current sleep aids largely focus on sedating the brain, often bypassing the natural hormonal cycles that make sleep restorative. The identification of the GHRH/somatostatin/locus coeruleus loop suggests that the next generation of treatments may be hormonal or neuro-modulatory rather than purely sedative.
Researchers are now looking toward human clinical trials to determine if the mouse models translate perfectly to human physiology. Given that the hypothalamus and locus coeruleus are highly conserved structures across all mammals, the likelihood of a similar circuit existing in humans is high.
As the medical community moves toward personalized medicine, understanding the specific neural handles of the growth hormone circuit could allow for "precision sleep" interventions. For athletes looking to optimize recovery, patients recovering from major surgery, or children with growth deficiencies, the ability to "dial in" the brain’s GH production through non-invasive stimulation or targeted pharmacology could revolutionize recovery and development.
This research, funded by HHMI and the Pivotal Life Sciences Chancellor’s Chair fund, stands as a testament to the importance of basic neuroscience in solving complex systemic health issues. By mapping the circuit that links the depths of sleep to the growth of the body, the UC Berkeley team has provided a roadmap for a healthier, more rested future.









Leave a Reply