A groundbreaking international study, spearheaded by researchers from the University of Ottawa Faculty of Medicine in Canada, has introduced a paradigm-shifting approach to gene therapy delivery, suggesting that harnessing the body’s intrinsic biological communication systems may be the key to precisely targeting therapeutic agents. Published in the journal Cell Biomaterials, the research provides compelling evidence that ultra-tiny, bubble-like structures known as small extracellular vesicles (sEVs) can be selectively utilized to deliver powerful gene-silencing therapies to specific organs, including historically challenging targets like the brain and kidneys. This discovery could unlock new avenues for treating a myriad of diseases, moving medicine closer to correcting underlying genetic causes rather than merely managing symptoms.
The Evolving Landscape of Modern Medicine and the Delivery Challenge
The 21st century has witnessed a quiet but profound revolution in medical science, characterized by a pivot from traditional pharmaceutical interventions towards advanced biological therapies. At the forefront of this transformation are RNA and gene-editing therapies, which promise to fundamentally alter disease progression by directly addressing genetic abnormalities or dysfunctional gene expression. Gene therapy, broadly defined, involves introducing, removing, or changing genetic material in cells to treat disease. RNA therapies, a closely related field, utilize various forms of RNA to influence protein production or silence specific genes. Among these, small interfering RNA (siRNA) has emerged as a particularly potent tool, capable of suppressing the expression of disease-causing genes with remarkable efficacy and duration. A single dose of certain siRNA therapeutics, for instance, has been shown to block gene expression for up to six months, offering unprecedented potential for long-term therapeutic benefit.
However, the immense promise of these next-generation therapies has been consistently hampered by a critical hurdle: the safe, efficient, and precise delivery of these delicate molecules to the intended cells and tissues within the complex human body. Systemic administration often leads to off-target effects, degradation of the therapeutic agent, or inadequate concentrations reaching the desired site. The blood-brain barrier, for example, notoriously impedes the delivery of most therapeutic agents to the central nervous system, leaving neurodegenerative diseases particularly challenging to treat. Similarly, the intricate filtration system of the kidneys presents unique difficulties for targeted drug delivery.
Small Extracellular Vesicles: Nature’s Nanocarriers
Enter small extracellular vesicles (sEVs), often referred to as exosomes, which are nanometer-scale lipid bilayer vesicles naturally secreted by virtually all cell types. Discovered decades ago, sEVs were initially dismissed as cellular debris, but subsequent research unveiled their crucial role as metabolic messengers, facilitating intercellular communication by ferrying a diverse cargo of proteins, lipids, and nucleic acids – including various forms of RNA – between cells. Refined over millions of years of evolution, these biological nanoparticles are perfectly designed for transport within the body, offering inherent biocompatibility and low immunogenicity, making them ideal candidates for drug delivery vehicles.
For over a decade, the scientific community and pharmaceutical industry have harbored considerable excitement regarding sEVs’ potential to revolutionize drug delivery. Major investments have poured into companies attempting to harness these vesicles. Yet, widespread clinical success remained elusive, primarily due to a fundamental misunderstanding of sEV biology. The prevailing approach assumed a "one-size-fits-all" model, where a single type of sEV could be engineered to deliver therapeutics universally across different tissues and organs. This oversimplified view often led to inconsistent results and limited therapeutic efficacy, as sEVs derived from one cell type might be quickly cleared from circulation or preferentially taken up by unintended cells.
A Biologically Informed Paradigm Shift: Specificity is Key
The Ottawa-led team, under the guidance of senior author Derrick Gibbings, a Professor in the Department of Cellular and Molecular Medicine and uOttawa’s Associate Vice-President, Research Support and Infrastructure, challenged this universalist assumption. Their study’s central revelation is that sEVs are not all alike; rather, their cell of origin fundamentally dictates their tropism – their natural tendency to travel to and be taken up by specific tissues in the body. This critical insight underscores a basic principle of biology: cellular communication is inherently highly specific and finely tuned.
"Our approach was to learn from nature – and work with nature – to find sEVs that could deliver to the tissues and cells where there were the most compelling targets for siRNA therapeutics," Gibbings explained. He drew a compelling analogy to illustrate this specificity: "sEVs carry specific messages to specific cells. If your house is on fire, you don’t phone your cardiac surgeon or your mechanic. If your computer crashes, you don’t phone your plumber." This commonsense understanding of communication, applied to the cellular realm, formed the bedrock of their successful strategy. Instead of forcing a general solution, the multidisciplinary international team (including scientists from Brazil and the U.S.) meticulously explored the natural behavior of sEVs, identifying and selecting those naturally predisposed to reach specific target tissues.
Precision in Action: Targeting Kidneys and the Brain
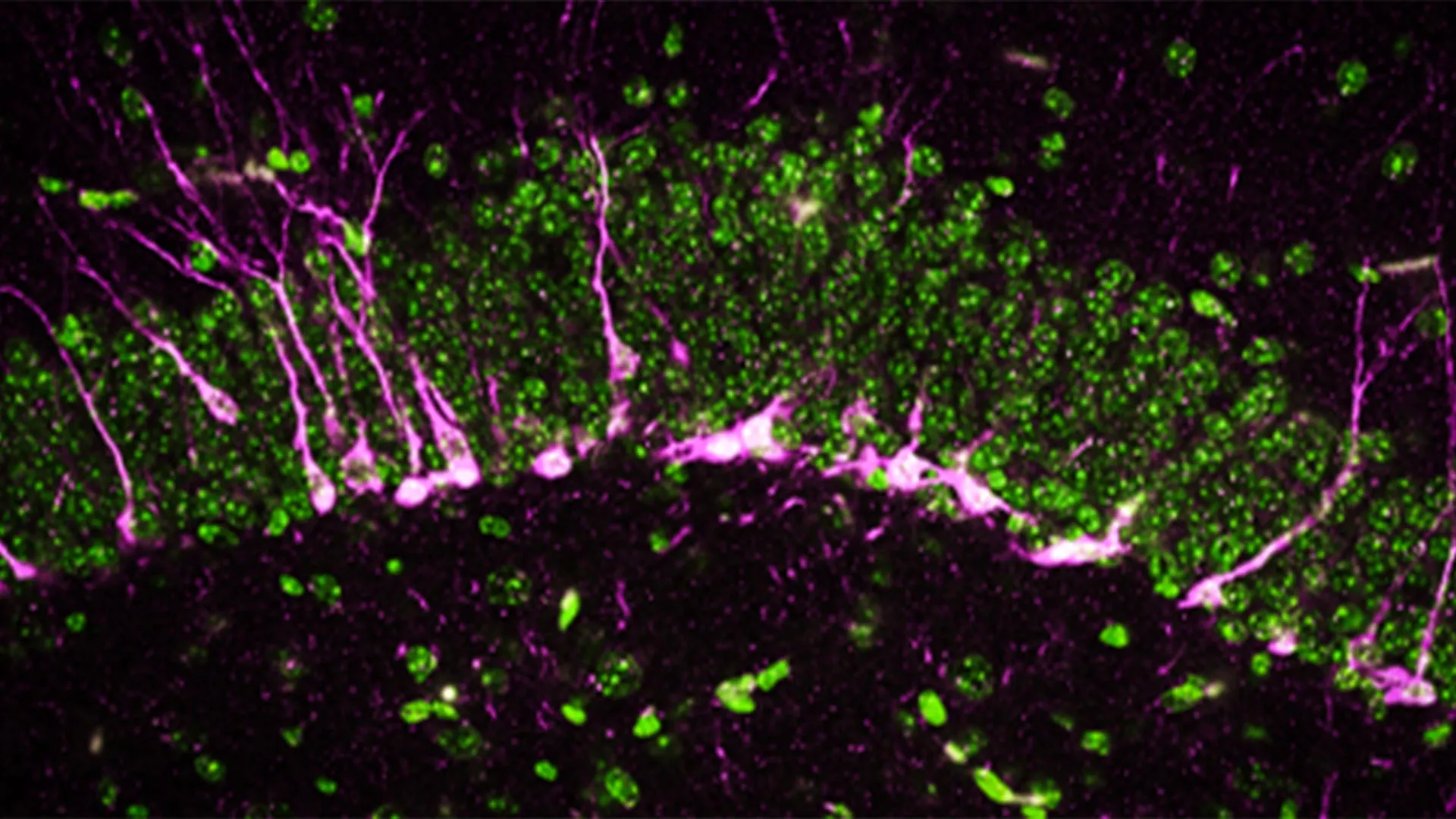
This biologically informed strategy yielded remarkable success in preclinical models. The researchers identified specific sEV populations that, when injected into the bloodstream, efficiently delivered siRNA directly to the kidneys. This targeted delivery significantly reduced disease symptoms in mouse models of chronic kidney disease, a condition affecting an estimated 15% of adults in the United States alone, often progressing to end-stage renal disease requiring dialysis or transplantation. Current treatments for chronic kidney disease primarily focus on managing symptoms and slowing progression, making a gene-silencing therapeutic a potentially transformative intervention.
Equally significant was their success in delivering treatments to the brain. By administering sEVs directly into the central nervous system, the team demonstrated effective delivery of therapeutics, leading to improved outcomes in a model of neurodegenerative disease. This achievement is particularly noteworthy given the formidable challenge posed by the blood-brain barrier, which excludes approximately 98% of small-molecule drugs and virtually all large-molecule biotherapeutics. The ability of specific sEVs to bypass or efficiently cross these barriers opens up unprecedented possibilities for treating devastating neurological conditions like Alzheimer’s, Parkinson’s, Huntington’s disease, and various forms of dementia, for which effective treatments remain largely elusive.
Crucially, the team methodically demonstrated similar success in larger animal models. The results scaled predictably based on body size and were not substantially altered by species-specific biological differences, a critical indicator that the approach could translate effectively from preclinical studies to human treatments. This translational potential marks a significant step forward, addressing a common hurdle in drug development where promising findings in rodent models often fail to replicate in larger species or humans.
Scaling Up for Clinical Impact and Future Directions
The study’s findings build upon decades of exciting progress in siRNA therapeutics. Since the discovery of RNA interference (RNAi) in the late 1990s, the field has rapidly matured, leading to the approval of several siRNA-based drugs for conditions like transthyretin amyloidosis and primary hyperoxaluria. However, the full potential of siRNA has been constrained by delivery limitations. The Ottawa team’s work offers a viable pathway to overcome these constraints, making a broader range of diseases amenable to gene-silencing therapies.
While the scientific evidence is compelling, several hurdles remain before this technology can reach widespread clinical application. Producing sEVs at the large scale required for pharmaceutical manufacturing is a significant challenge, necessitating advances in bioreactor technology and purification methods. Furthermore, while siRNA itself can have a long duration of action, ensuring the sustained presence and activity of sEV-delivered therapeutics in the body needs further optimization. The global scientific community is actively engaged in addressing these production and pharmacokinetic challenges.
Despite these hurdles, Gibbings remains optimistic about the future. He and his team are actively seeking partners – either investors, industry collaborators, or academic clinicians – to advance this technology into clinical trials. Their immediate focus is on severe kidney diseases that currently have limited treatment options, specifically highlighting a chronic kidney disease caused by a genetic variation in the APOL1 gene. This condition affects a large patient population, particularly individuals of African ancestry, and frequently leads to kidney failure requiring dialysis or transplant, with high mortality rates. Targeting APOL1 expression with precisely delivered siRNA could offer a life-saving intervention.
"We have collected a lot of data that shows that sEVs can be effective, safe and scalable delivery vehicles," Gibbings stated, emphasizing the robustness of their findings. The shift from a generic, engineered approach to a biologically guided one represents a crucial maturation point in the field of sEV therapeutics. It signifies a deeper understanding of cellular mechanisms and a commitment to working with nature rather than attempting to override it.
Broader Implications and Industry Outlook
This research holds profound implications for the entire pharmaceutical and biotechnology landscape. By validating a highly specific, nature-inspired delivery system, the study could accelerate the development of a new generation of targeted therapies, particularly for diseases previously considered "undruggable" due to delivery constraints. The estimated global gene therapy market, valued at over $10 billion in 2023 and projected to grow substantially, stands to benefit immensely from such advancements, potentially opening up new market segments and therapeutic indications.
Industry analysts are likely to view this discovery as a significant de-risking factor for sEV-based drug development. The move away from a "universal" sEV model towards a tissue-specific approach provides a clearer, more predictable development pathway, potentially reducing the high failure rates associated with novel drug delivery systems. This specificity not only enhances efficacy but also minimizes off-target toxicity, a critical consideration for regulatory approval and patient safety.
Furthermore, the insights gained from this study could inform the design of other nanoparticle-based drug delivery systems, encouraging a more nuanced, biologically driven approach to their engineering. The principle that "cell of origin determines destination" for sEVs might translate to designing synthetic nanoparticles with surface modifications that mimic the targeting capabilities of natural vesicles.
In conclusion, the University of Ottawa-led study marks a pivotal moment in the quest for precise gene therapy delivery. By recognizing and leveraging the inherent specificity of small extracellular vesicles, researchers have unveiled a powerful, nature-inspired solution to a long-standing challenge in medicine. This paradigm shift, moving from broad assumptions to biologically informed precision, not only promises to accelerate the development of transformative therapies for chronic kidney disease and neurodegenerative disorders but also sets a new standard for how we approach drug delivery in the era of genetic medicine. The quiet revolution in gene therapy is indeed poised for its next leap, guided by the elegant wisdom of biology itself.






Leave a Reply