Saudi Arabia is rapidly solidifying its position as the preeminent growth market for clinical trials within the Middle East, a trajectory propelled by substantial government funding, a rapidly advancing healthcare infrastructure, and the ambitious economic diversification outlined in Vision 2030. The confluence of these factors is creating a fertile ground for complex, multicenter studies, positioning the Kingdom as a vital player in global pharmaceutical research and development.
A Surge in Healthcare Spending and Pharmaceutical Market Growth
The Kingdom’s healthcare and pharmaceutical sectors are experiencing a robust expansion phase, underscoring its strategic importance in the life sciences arena. Projections from GlobalData indicate a sustained upward trend, with total healthcare spending anticipated to rise by 4.7% in 2026, reaching an estimated $61.05 billion. This growth is further reflected in per capita expenditure, which is expected to climb from approximately $1,692.21 in 2025 to $1,741.75 in 2026. In parallel, the Saudi pharmaceutical market is forecast to see a significant increase of 6.07% in 2026, projecting sales to reach $17.23 billion. These figures highlight a clear commitment to enhancing healthcare access and capabilities, which directly benefits the clinical trials ecosystem.
Vision 2030: A Catalyst for Healthcare Transformation
Central to this burgeoning growth is Saudi Arabia’s Health Sector Transformation Programme, an integral component of Vision 2030. This ambitious initiative, supported by mandatory health insurance, is actively fostering a more integrated and efficient public-private healthcare ecosystem. The program’s core objectives include the development of a more comprehensive, effective, and innovative health system that emphasizes financial sustainability, disease prevention, and the widespread adoption of e-health services and digital solutions.
A tangible manifestation of this vision is the establishment of the SEHA Virtual Hospital. This pioneering initiative connects over 150 hospitals nationwide, offering a broad spectrum of approximately 30 specialized services. By facilitating virtual consultations, enabling remote diagnostics, and streamlining e-prescribing across a national network, SEHA significantly enhances access to specialized medical care, particularly for populations in remote areas. This digital infrastructure not only simplifies patient follow-up but also improves the collection of critical health data, a crucial element for clinical trial recruitment and monitoring.
Building the Foundations for a Thriving Clinical Trials Landscape
Vision 2030’s overarching strategy is instrumental in laying the groundwork for a robust clinical trials environment. The program’s emphasis on expanding healthcare access, modernizing hospital and clinic facilities, and incentivizing private sector investment is creating a network of research-ready sites equipped with contemporary infrastructure. This development translates into smoother patient referral pathways and more stable, well-resourced settings conducive to conducting complex and long-term studies.
The Kingdom boasts an extensive network of general and specialist hospitals, complemented by thousands of clinics, polyclinics, and mobile healthcare units. This diverse infrastructure provides sponsors and Contract Research Organizations (CROs) with a wide array of potential trial sites, spanning different geographical regions and care settings. Such diversity is invaluable for recruiting large and representative study populations, and for executing multicenter protocols within a unified national framework.
The increasing participation of the private sector, coupled with the implementation of mandatory health insurance for private sector employees, is further enhancing the system’s preparedness for clinical research. Compulsory insurance ensures a consistent demand for healthcare services throughout the year, while growing private investment bolsters capacity and familiarity with international research standards. This evolving public-private healthcare matrix expands the variety and capabilities of potential trial sites, offering sponsors greater flexibility in matching their protocol requirements with specific facility types.
Furthermore, the rising incidence of chronic conditions such as diabetes and cardiovascular disease, alongside strong projected growth in healthcare and pharmaceutical expenditure, positions Saudi Arabia as a strategically important location for sponsors targeting high-burden therapeutic areas. This epidemiological profile makes the infrastructure developed under Vision 2030 particularly relevant for researchers focusing on chronic and lifestyle-related diseases, who require access to large, well-characterized patient populations.
Navigating Challenges and Seizing Opportunities
Despite the significant strides being made, Saudi Arabia faces certain challenges in its pursuit of becoming a regional clinical trial hub. While the infrastructure is robust, specialized expertise remains concentrated in major urban centers, which can limit the scope of highly complex studies and the diversity of patient recruitment. Additionally, patient enrollment can be hindered by factors such as low public awareness, cultural hesistancy, and a limited understanding of the benefits of participating in clinical trials.

From a logistical standpoint, the distribution of investigational products and biological samples across a vast and geographically dispersed country presents considerable cost and risk. Maintaining strict temperature control and ensuring compliance, especially at remote sites and in warm climates, requires meticulous planning and execution. The evolving public-private healthcare landscape introduces further complexities, with potential variations in procedures, data standards, and inter-provider coordination that necessitate careful navigation.
Successfully integrating into the global clinical trials ecosystem also demands continuous improvement in data quality, ethical standards, transparency, and innovation. This integration hinges on close collaboration among government entities, academic institutions, healthcare providers, and international partners.
Oximio’s Strategic Role in Enhancing Clinical Trial Capabilities
Specialized logistics providers like Oximio are playing a pivotal role in ensuring the operational success, safety, and compliance of multicenter clinical trials in Saudi Arabia. Oximio brings over two decades of global experience in clinical trial logistics, adhering to stringent Good Distribution Practice (GDP) standards and employing proven project management methodologies to deliver tailored, end-to-end supply chain solutions. Their comprehensive services encompass sourcing, warehousing, temperature-controlled transportation, site delivery, and regulatory documentation management, allowing sponsors to consolidate these critical touchpoints with a single, expert provider, thereby reducing operational risks and enhancing efficiency.
Oximio has established its inaugural facility in Saudi Arabia, specifically dedicated to clinical trial services. A cornerstone of this investment is the development of advanced storage and transportation infrastructure, including state-of-the-art, GDP-compliant warehouses designed to manage medicines and ancillary supplies under precisely monitored ambient and cold-storage conditions.
Continuous temperature monitoring throughout storage and transit is paramount to ensuring that investigational products and samples remain within prescribed limits. This meticulous control is vital for the integrity and validity of study data, especially in regions like Saudi Arabia where extreme climatic conditions pose a significant risk. Deviations from temperature requirements can compromise both the scientific quality of a study and regulatory compliance.
By offering a single point of coordination for import processes, customs clearance, destruction of expired stock, and return logistics, Oximio effectively mitigates regulatory risks and alleviates administrative burdens for sponsors who may be unfamiliar with local requirements. This integrated approach minimizes handovers and the potential for miscommunication, which are critical for ensuring trials proceed smoothly with minimal loss or delay.
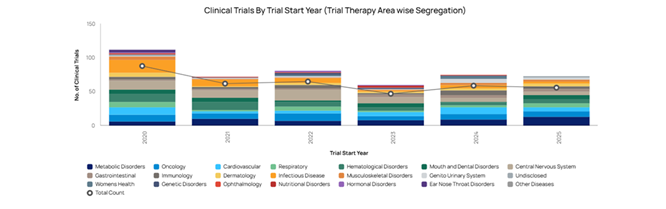
Saudi Arabia: A Beacon of Clinical Research Innovation in the Middle East
The emergence of advanced clinical logistics capabilities, exemplified by providers like Oximio, is instrumental in facilitating the compliant and temperature-controlled movement of drugs and samples. This capability is essential for the successful execution of complex multicenter trials. Collectively, these strengths are transforming Saudi Arabia from merely a growing market into a burgeoning regional hub for clinical research, poised to drive innovation and improve health outcomes across the Middle East.
The ongoing commitment to healthcare infrastructure development, coupled with strategic investments in specialized services, signals a promising future for clinical trials in Saudi Arabia. As the Kingdom continues to embrace innovation and foster international collaboration, it is well-positioned to attract a greater volume of global research, ultimately benefiting patients and advancing medical science.
To gain deeper insights into clinical trial locations and opportunities within the Middle East, further resources and reports are available for download.















Leave a Reply