Researchers at Johns Hopkins Medicine have unveiled a significant breakthrough in the understanding of Alzheimer’s disease, identifying a specific protein and its gaseous byproduct as critical components in the maintenance of cognitive health. Supported by a new wave of funding from the National Institutes of Health (NIH), the study explores the role of Cystathionine γ-lyase (CSE), an enzyme primarily known for producing hydrogen sulfide ($H_2S$). While hydrogen sulfide is commonly recognized for its distinct "rotten egg" odor and its toxicity at high concentrations, the research suggests that in minute, naturally occurring quantities within the brain, it serves as a vital neuroprotective agent.
The findings, published in the Proceedings of the National Academy of Sciences (PNAS), indicate that CSE is a fundamental regulator of memory formation and cellular integrity. Led by Bindu Paul, M.S., Ph.D., an associate professor of pharmacology, psychiatry, and neuroscience at the Johns Hopkins University School of Medicine, the team utilized genetically engineered mouse models to demonstrate that a deficiency in this protein triggers a cascade of neurodegenerative symptoms that mirror the progression of Alzheimer’s disease in humans.
The Biochemical Role of Hydrogen Sulfide in the Brain
Hydrogen sulfide belongs to a class of molecules known as gasotransmitters, which include nitric oxide and carbon monoxide. These gases are produced endogenously and act as signaling molecules that regulate various physiological processes. In the brain, $H_2S$ is involved in modulating synaptic plasticity, the process by which connections between neurons strengthen or weaken over time—a cornerstone of learning and memory.
The central challenge for researchers has long been the "hormetic" nature of hydrogen sulfide: it is beneficial at low levels but lethal at high ones. Because direct administration of the gas to the brain is medically unsafe, the Johns Hopkins team focused on the enzyme responsible for its production. By understanding how CSE maintains optimal $H_2S$ levels, scientists hope to develop pharmacological interventions that can stimulate the protein’s activity, thereby fortifying the brain’s natural defenses against aging and disease.
Dr. Paul’s research highlights that when CSE levels are insufficient, the brain loses a primary mechanism for neutralizing oxidative stress. This leads to an accumulation of reactive oxygen species, which damage DNA and compromise the structural integrity of neurons. The study found that mice lacking the CSE gene exhibited heightened levels of DNA damage and a significant weakening of the blood-brain barrier—a specialized system of microvessels that protects the brain from toxins while allowing essential nutrients to pass through.
Experimental Evidence: The Barnes Maze and Cognitive Decline
To quantify the impact of CSE on cognitive function, the research team conducted a series of behavioral experiments using the Barnes maze, a standard tool in neuroscience for assessing spatial learning and memory. The maze consists of a circular platform with multiple holes around the perimeter, only one of which leads to a dark escape chamber. Mice, which naturally prefer dark, enclosed spaces over bright, open areas, must use visual cues to remember the location of the escape route.
The results provided a clear timeline of cognitive deterioration linked to CSE deficiency. At two months of age—roughly equivalent to early adulthood in humans—mice lacking the CSE protein performed at the same level as healthy control mice, locating the escape chamber within three minutes. However, by the time the mice reached six months of age, a stark divergence emerged. While the healthy mice continued to navigate the maze efficiently, the CSE-deficient mice struggled significantly, failing to remember the location of the shelter despite repeated training.
"The decline in spatial memory indicates a progressive onset of neurodegenerative disease that we can attribute directly to the loss of CSE," stated Suwarna Chakraborty, a researcher in Dr. Paul’s lab and the study’s first author. This progression suggests that the absence of the protein does not cause immediate failure but rather a gradual erosion of the brain’s ability to process and retain information, much like the early stages of dementia in humans.
Cellular Analysis and Structural Brain Damage
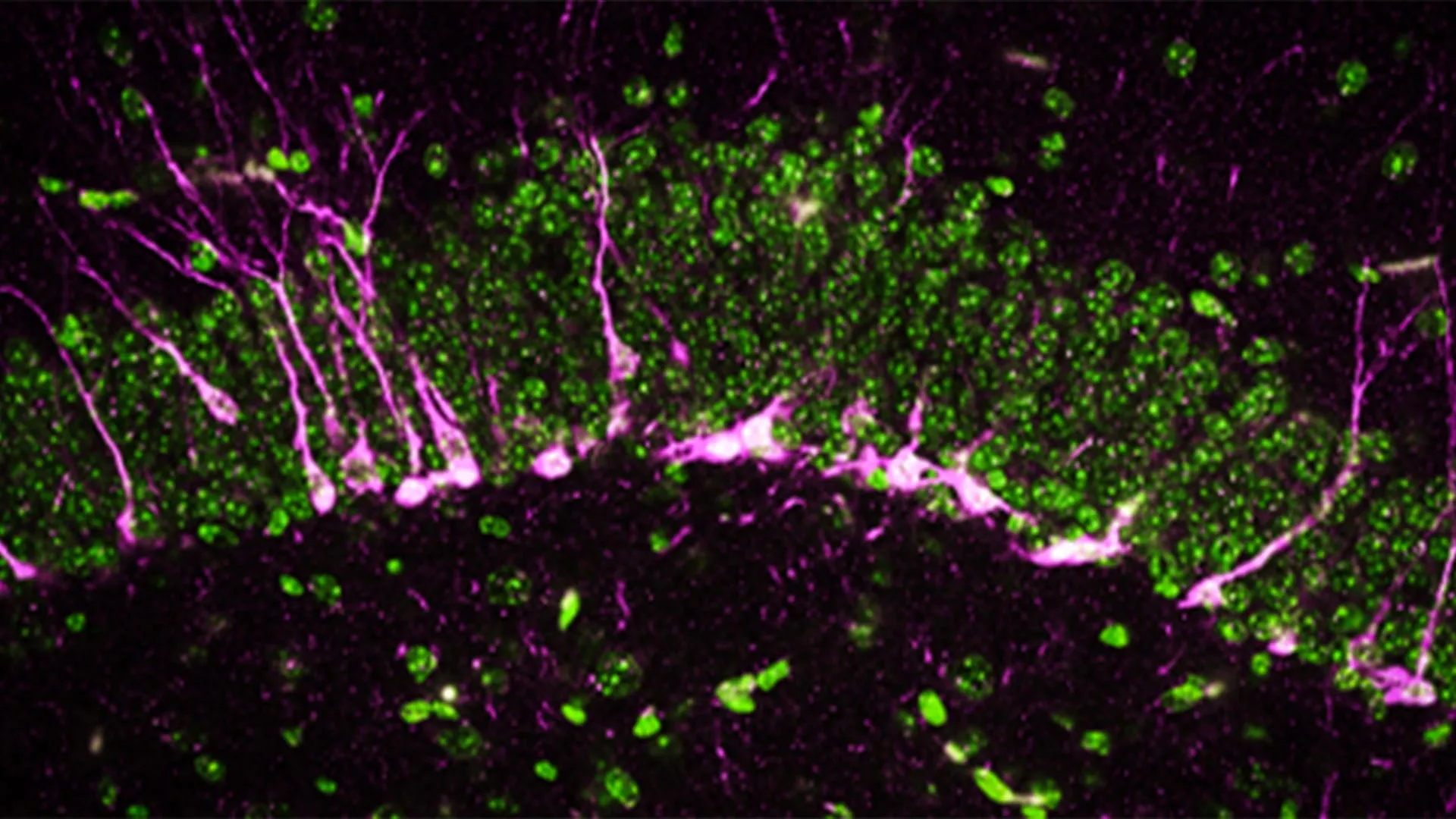
Beyond behavioral observations, the researchers utilized high-powered electron microscopy and biochemical assays to examine the physical changes occurring within the brain tissue. They focused specifically on the hippocampus, the region of the brain responsible for converting short-term memories into long-term ones.
A key finding was the disruption of neurogenesis—the process by which new neurons are generated. In healthy brains, new neurons are constantly being formed and integrated into existing circuits. In the CSE-deficient mice, the researchers discovered that proteins essential for this process were either missing or severely reduced. Furthermore, the newly formed neurons were unable to migrate effectively to the hippocampus, essentially stalling the brain’s ability to repair and update its cognitive "hardware."
The electron microscopy revealed even more alarming structural damage. The blood-brain barrier in the CSE-deficient mice showed large breaks and structural irregularities. A compromised blood-brain barrier is a hallmark of Alzheimer’s disease, as it allows inflammatory markers and pathogens to enter the brain, further accelerating the death of neurons. "The mice lacking CSE were compromised at multiple levels," noted co-first author Sunil Jamuna Tripathi. "The cellular and structural deficits we observed correlate precisely with the symptoms seen in clinical Alzheimer’s cases."
A Chronology of Research and the Legacy of Solomon Snyder
The current study is the culmination of nearly two decades of research into the CSE protein. The journey began in 2008, when researchers first developed a strain of mice lacking the CSE gene to study its role in the cardiovascular system. At that time, the protein was primarily linked to blood pressure regulation and blood vessel function.
In 2014, a team led by Solomon Snyder, M.D., D.Sc., D.Phil., a legendary figure in neuroscience and professor emeritus at Johns Hopkins, expanded the scope of CSE research. They reported that the protein played a protective role in Huntington’s disease, another devastating neurodegenerative condition. By 2021, the group had narrowed their focus to Alzheimer’s, finding that CSE function was impaired in mouse models of the disease and that supplemental micro-injections of hydrogen sulfide could temporarily restore some cognitive function.
The latest 2024 study represents a critical refinement of this work. While previous experiments used mice that were already genetically predisposed to neurodegeneration through other mutations, this research isolates the CSE protein itself. By showing that the loss of CSE alone is sufficient to induce Alzheimer’s-like symptoms, the team has established the protein as a "major player" in brain health.
Dr. Snyder, who retired from the Johns Hopkins faculty in 2023 but remains a co-corresponding author on the study, emphasized the significance of these findings. "This most recent work indicates that CSE alone is a major player in cognitive function and could provide a new avenue for treatment pathways in Alzheimer’s disease," he stated.
Broader Implications for Alzheimer’s Treatment
The search for effective Alzheimer’s treatments has been fraught with challenges. For decades, the "amyloid hypothesis"—the idea that the buildup of beta-amyloid plaques is the primary cause of the disease—dominated the research landscape. However, many drugs designed to clear these plaques have failed to significantly improve cognitive outcomes in clinical trials.
The Johns Hopkins research offers a different perspective, focusing on neuroprotection and vascular integrity rather than just plaque removal. By targeting the CSE/hydrogen sulfide pathway, researchers may be able to develop therapies that protect the brain from the "inside out," strengthening the blood-brain barrier and boosting the brain’s resilience to oxidative stress.
According to the U.S. Centers for Disease Control and Prevention (CDC), more than 6 million Americans are currently living with Alzheimer’s, a number expected to nearly triple by 2060. The economic burden is equally staggering, with costs related to the disease estimated at $345 billion annually in the United States alone. Given the limitations of current FDA-approved treatments, which often only manage symptoms rather than slowing disease progression, the identification of CSE as a therapeutic target is of paramount importance.
Collaborative Effort and Future Directions
The study was a massive collaborative effort involving researchers from several prestigious institutions, including Case Western Reserve University, the Leibniz Institute for Analytical Sciences in Germany, the Medical University of South Carolina, and West Virginia University School of Medicine.
The diverse array of funding sources—ranging from the Department of Defense and the American Heart Association to private foundations like the Valour Foundation and the Wick Foundation—underscores the cross-disciplinary interest in this research. The involvement of the Department of Veterans Affairs also highlights the potential relevance of these findings for veterans, who may be at higher risk for neurodegenerative conditions due to traumatic brain injuries.
Moving forward, the research team aims to identify small molecules or existing drugs that can safely cross the blood-brain barrier and enhance CSE activity. If successful, this could lead to a new class of "gas-boosting" therapeutics that offer hope to millions of families affected by Alzheimer’s. The journey from a "rotten egg gas" to a potential cure for dementia illustrates the often unpredictable and transformative nature of basic scientific research.














Leave a Reply