The Mechanical Paradox of Post-Stroke Neuroplasticity
Stroke remains one of the leading causes of long-term disability worldwide, affecting millions of individuals who often face permanent motor impairments. Traditional understanding of stroke recovery has focused primarily on the "lesion site"—the specific area where blood flow was interrupted and tissue was damaged. However, the Stevens INI study shifts the focus toward the "contralesional" hemisphere, or the side of the brain that did not suffer the primary injury.
The central finding of the research is that in patients with severe physical impairments, the brain’s undamaged regions appear structurally younger than their chronological age would suggest. This "youthfulness" was not found in patients with mild impairments, suggesting that the more severe the loss of function, the more aggressively the brain attempts to rewire itself. This structural rejuvenation is believed to be a manifestation of the brain’s attempt to compensate for lost motor control by recruiting healthy networks to take over the tasks once performed by the damaged areas.
Dr. Hosung Kim, PhD, associate professor of research neurology at the Keck School of Medicine of USC and the study’s co-senior author, noted that larger strokes typically accelerate the aging process in the damaged hemisphere. However, the paradox lies in the opposite hemisphere. "This pattern suggests the brain may be reorganizing itself, essentially rejuvenating undamaged networks to compensate for lost function," Kim stated. This "rejuvenation" is not a sign of the brain returning to a literal state of youth, but rather a structural shift—possibly involving increased synaptic density or altered connectivity—that AI models associate with younger brains.
Leveraging Artificial Intelligence to Map Brain Health
To uncover these subtle structural changes, the research team employed a sophisticated type of artificial intelligence known as a graph convolutional network (GCN). Unlike standard deep learning models that might analyze a flat image of a brain scan, GCNs are designed to understand the complex, non-Euclidean relationships between different regions of the brain, treating the organ as a living network of interconnected nodes.
The AI was trained on a massive dataset of tens of thousands of MRI scans from healthy individuals to establish a baseline for "normal" brain aging. By learning the structural nuances of how the brain typically matures and declines over decades, the model became capable of estimating the "biological age" of specific brain regions. When the researchers applied this model to the scans of stroke survivors, they could calculate the "brain-predicted age difference" (brain-PAD).
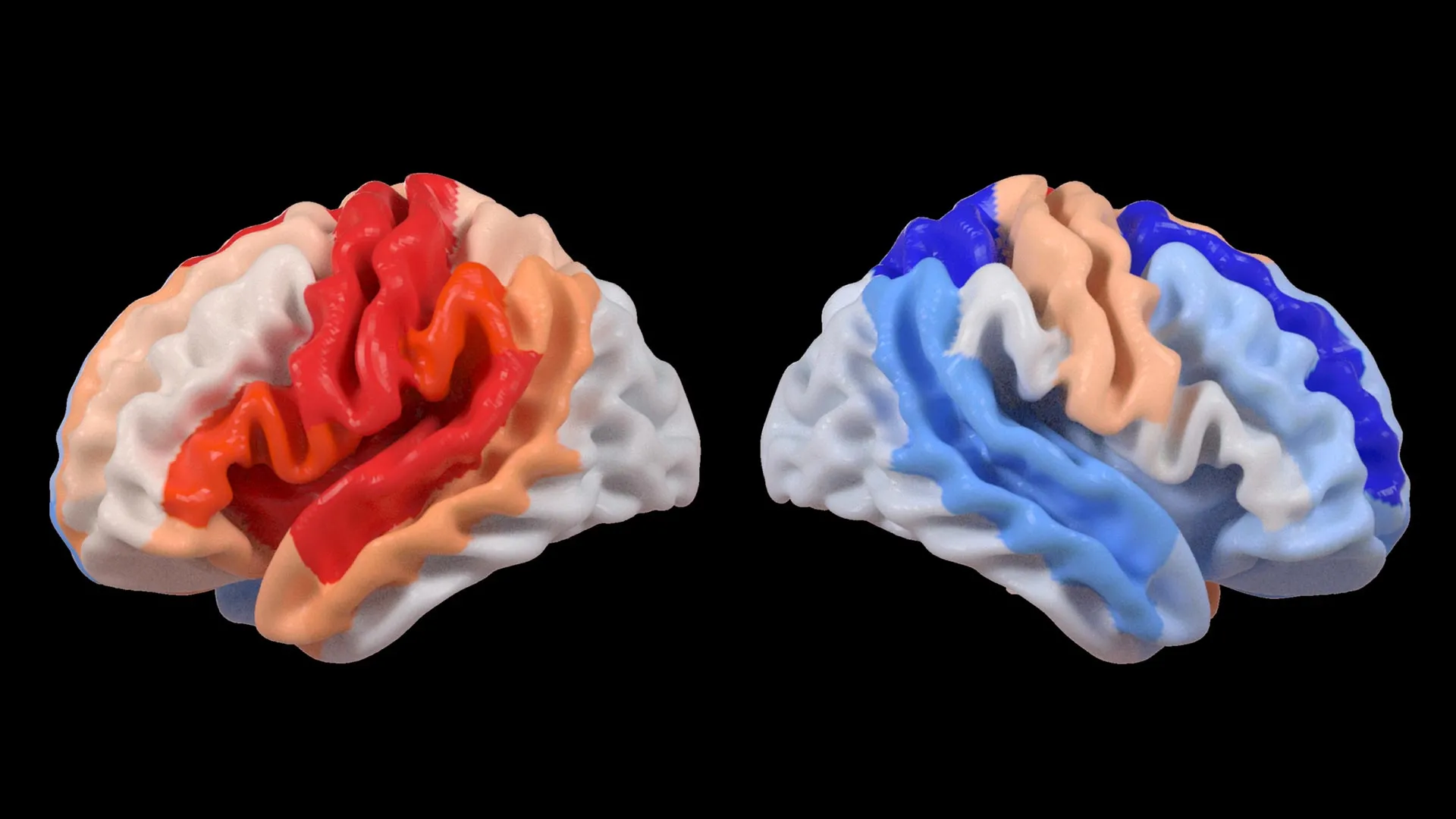
Brain-PAD is a critical metric in modern neuroscience. If a person’s brain-PAD is positive, it suggests their brain looks "older" than their actual years, often a sign of neurodegeneration or poor health. Conversely, a negative brain-PAD indicates a "younger" brain. In the context of this study, the researchers analyzed 18 specific brain regions across both hemispheres. They found that in chronic stroke survivors with severe motor deficits, the frontoparietal network in the healthy hemisphere showed a significantly negative brain-PAD. This network is vital for movement planning, coordination, and attention, making it the primary candidate for the brain’s compensatory efforts.
The Global Scale of the ENIGMA Collaboration
The scale of this study is unprecedented in the field of stroke neuroimaging. It was conducted under the umbrella of the Enhancing NeuroImaging Genetics through Meta-Analysis (ENIGMA) Stroke Recovery Working Group. ENIGMA is a global consortium that pools data from more than 50 countries, allowing researchers to achieve a level of statistical power that would be impossible for any single institution to reach on its own.
For this specific project, the team analyzed brain scans from more than 500 stroke survivors collected across 34 different research centers in eight countries. By standardizing MRI data and clinical information from diverse populations, the researchers were able to filter out "noise" and identify patterns that are universal to the human response to stroke.
Arthur W. Toga, PhD, director of the Stevens INI and Provost Professor at USC, emphasized the importance of this international cooperation. "By pooling data from hundreds of stroke survivors worldwide and applying cutting-edge AI, we can detect subtle patterns of brain reorganization that would be invisible in smaller studies," Toga explained. This global approach ensures that the findings are not limited to a specific demographic or healthcare system, but represent a fundamental biological insight into human neurobiology.
Chronology and Evolution of the Research
The path to these findings involved several distinct phases of data collection, model training, and clinical correlation:
- Data Harmonization: The ENIGMA group spent years collecting and standardizing MRI scans from stroke survivors in various stages of recovery. This involved ensuring that scans from different machines and different countries could be compared accurately.
- AI Training: Researchers utilized tens of thousands of MRI scans from healthy controls to train the graph convolutional network. This phase established the "biological clock" the AI would use to judge the stroke survivors’ brains.
- Regional Analysis: The team moved beyond looking at the brain as a whole, instead segmenting it into 18 functional regions to see which specific areas were most affected by post-stroke reorganization.
- Clinical Correlation: The final and most crucial step involved comparing the AI’s "brain age" predictions with actual motor function scores. The survivors included in the study were in the chronic stage of recovery—more than six months post-stroke—meaning their brain structures had reached a relatively stable state after the initial trauma.
The results consistently showed that those who had failed to regain significant movement through traditional rehabilitation were the ones whose brains showed the most dramatic "youthful" shifts in the healthy hemisphere. This suggests that the brain’s attempt to compensate is a long-term, structural commitment rather than a temporary reaction.
Implications for the Future of Stroke Rehabilitation
The discovery of regional differential brain aging has profound implications for how the medical community views stroke recovery. Currently, rehabilitation is often a "one-size-fits-all" approach, involving physical and occupational therapy aimed at retraining the damaged side of the body. However, the Stevens INI findings suggest that the brain’s own internal strategy might involve shifting the heavy lifting to the healthy side.
If clinicians can identify which patients are undergoing this type of "youthful" reorganization, they might be able to tailor therapies to support it. For example, non-invasive brain stimulation could be used to enhance the activity of the frontoparietal network in the healthy hemisphere, potentially boosting its ability to compensate for the damaged motor system.
Furthermore, this research provides a new biomarker for neuroplasticity. Traditional imaging techniques often struggle to show why some patients improve while others with similar lesions do not. By using brain-PAD as a measure of structural reorganization, doctors could gain a clearer picture of a patient’s recovery potential and adjust expectations and treatment plans accordingly.
Analyzing the Impact: A Paradigm Shift in Neuroplasticity
This study challenges the traditional deficit-based model of brain injury. In the deficit model, an injury is seen purely as a loss of function and an acceleration of decay. The "rejuvenation" observed in the contralesional hemisphere suggests a more dynamic, "resource-allocation" model. When the primary motor cortex is destroyed, the brain appears to divert its biological resources toward maintaining and even "upgrading" other networks to maintain some level of functional output.
However, the researchers are careful to note that this "youthfulness" is not a perfect fix. The study showed that these younger-looking regions were most prevalent in those with the worst motor outcomes. This implies that while the brain is trying its best to adapt, the compensation is not always enough to restore full movement. It is a biological attempt at a "workaround" rather than a total repair.
The fact that this was most visible in the frontoparietal network is particularly telling. This network acts as the brain’s "executive coordinator." When the specialized "motor execution" hardware is damaged, the brain relies more heavily on its "planning and coordination" software to navigate the world.
Looking Ahead: Toward Personalized Medicine
The Stevens INI team is already planning the next phase of this research. The current study focused on the chronic stage of stroke (six months or more post-injury). The next step is a longitudinal study that follows patients from the immediate hours after a stroke through several years of recovery. By tracking how brain-PAD changes in real-time, researchers hope to identify the exact window when the brain is most "plastic" and receptive to intervention.
This work was supported by the National Institutes of Health (NIH) and involved collaborators from prestigious institutions including the University of British Columbia, Monash University, Emory University, and the University of Oslo. The breadth of this collaboration underscores the global priority of solving the puzzle of stroke recovery.
As AI continues to evolve, its ability to detect these "hidden" patterns of aging and rejuvenation will likely become a standard tool in neurology. For the millions of stroke survivors worldwide, this research offers more than just scientific insight—it offers the hope of a personalized roadmap to recovery, driven by a deeper understanding of the brain’s remarkable resilience. The "younger" brain structures found in this study are a testament to the organ’s tireless effort to preserve the self, even in the wake of devastating injury. Through the lens of artificial intelligence, we are finally beginning to see the full picture of that struggle and the incredible biological creativity it inspires.














Leave a Reply