In a landmark study published in The Lancet Digital Health, researchers from the USC Mark and Mary Stevens Neuroimaging and Informatics Institute (Stevens INI) at the Keck School of Medicine of USC have uncovered a paradoxical phenomenon in the way the human brain responds to traumatic injury. The research reveals that individuals suffering from severe physical impairments following a stroke often exhibit a "younger" structural brain age in regions that were not directly damaged by the event. This discovery, made possible through advanced artificial intelligence and a massive global dataset, offers a transformative look at neuroplasticity—the brain’s ability to reorganize and adapt its structure in response to external and internal changes.
The findings suggest that the brain does not simply degrade following a stroke; rather, it engages in a sophisticated, albeit sometimes incomplete, process of reorganization. While the hemisphere directly affected by the stroke shows signs of accelerated biological aging, the opposite, undamaged hemisphere appears to undergo a structural "rejuvenation." This youthful appearance in the contralesional side of the brain is believed to be a compensatory mechanism, as the brain attempts to reroute neural signals and reclaim lost motor functions through undamaged networks.
The Technological Vanguard: AI and Brain Aging Metrics
To quantify these subtle structural shifts, the research team utilized a sophisticated form of artificial intelligence known as a graph convolutional network (GCN). Unlike traditional neural networks, GCNs are uniquely suited for analyzing complex, interconnected data structures like the human brain. By training this AI on tens of thousands of MRI scans from healthy individuals across a wide age spectrum, the scientists established a baseline for "normal" brain aging.
The model was then applied to the MRI data of 514 stroke survivors. The AI estimated the biological age of 18 specific brain regions in each hemisphere. By comparing the predicted biological age with the patient’s actual chronological age, researchers calculated the "brain-predicted age difference" (brain-PAD). A positive brain-PAD indicates a brain that appears older than the person’s actual age—often a sign of disease or decline—while a negative brain-PAD suggests a brain that appears structurally younger and more resilient.
This methodology allowed the team to move beyond simple visual inspections of lesions. Traditional imaging can identify where tissue has died, but it often fails to capture the systemic changes occurring in healthy tissue as it reacts to the injury. The use of brain-PAD provides a holistic biomarker of brain health that reflects the cumulative impact of both the injury and the brain’s subsequent attempts at repair.
The Paradox of the Younger Brain
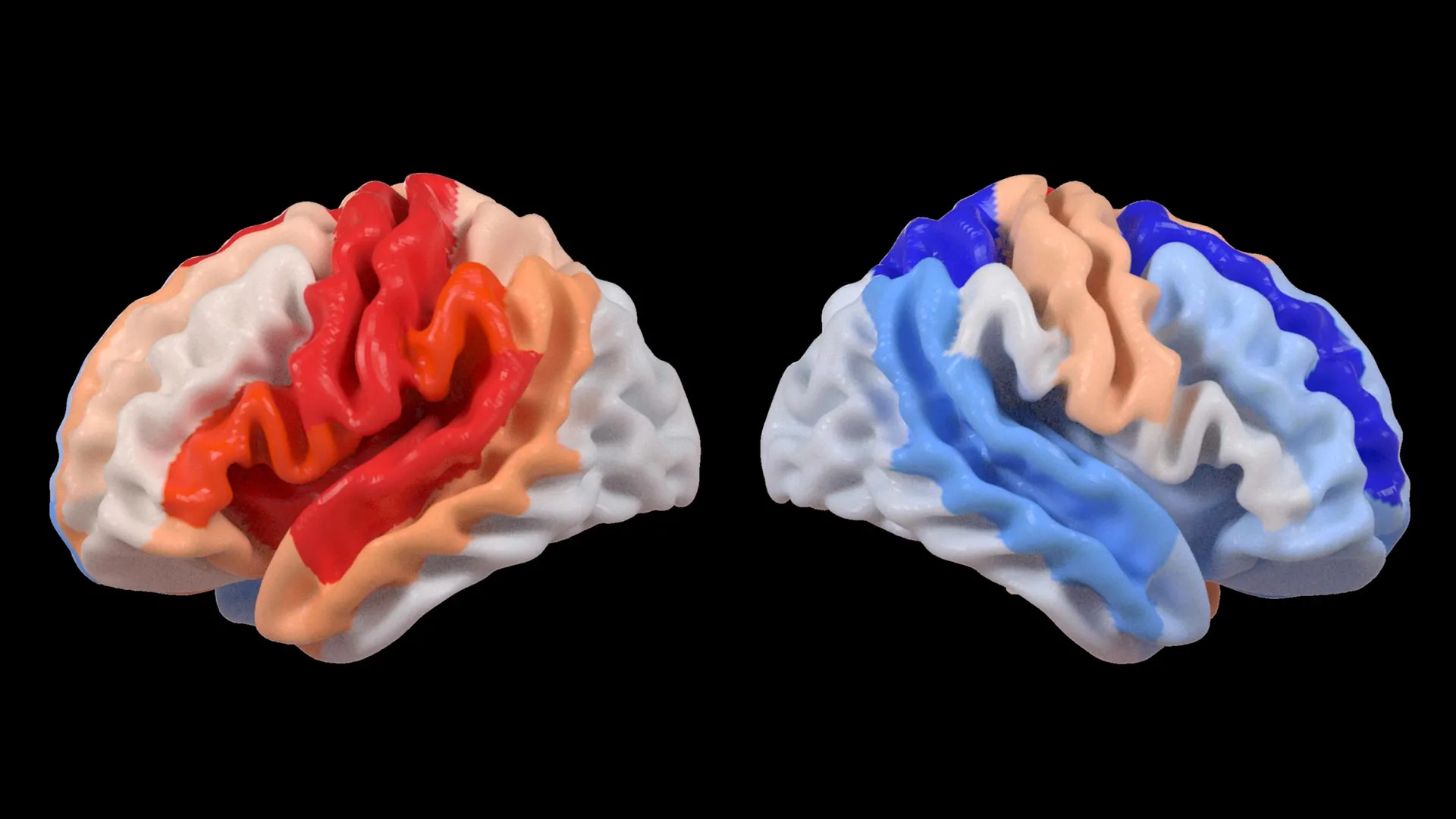
The most striking result of the study was the divergence between the two hemispheres of the stroke survivors. In the ipsilesional hemisphere (the side where the stroke occurred), the AI detected significant "accelerated aging." This was expected, as the trauma of an ischemic or hemorrhagic stroke causes rapid cell death and inflammation, which mimics the structural decline seen in advanced age.
However, in the contralesional hemisphere (the side opposite the injury), the researchers observed the opposite effect. Patients with the most severe motor impairments—those who struggled most with movement, coordination, and balance—exhibited brain structures that appeared significantly younger than their chronological age. This effect was most pronounced in the frontoparietal network, a critical circuit responsible for executive function, attention, and the planning of complex movements.
"We found that larger strokes accelerate aging in the damaged hemisphere but paradoxically make the opposite side of the brain appear younger," said Hosung Kim, PhD, associate professor of research neurology at the Keck School of Medicine of USC and co-senior author of the study. "This pattern suggests the brain may be reorganizing itself, essentially rejuvenating undamaged networks to compensate for lost function."
Dr. Kim noted that this "youthfulness" is likely a reflection of the brain’s effort to adjust. When the primary motor pathways are destroyed, the brain may recruit the contralesional side to take over duties it does not normally handle. This increased activity and structural remodeling may result in a denser, more robust appearance in MRI scans, which the AI interprets as a younger biological state.
Global Collaboration and the ENIGMA Consortium
The scale of this study was made possible by the Enhancing NeuroImaging Genetics through Meta-Analysis (ENIGMA) Stroke Recovery Working Group. ENIGMA is a global consortium that pools neuroimaging data from over 50 countries, allowing researchers to conduct studies with statistical power that would be impossible at a single institution.
For this specific project, the team analyzed scans collected across 34 different research centers in eight countries. By standardizing MRI data and clinical motor function scores from diverse populations, the researchers created the largest and most comprehensive stroke neuroimaging dataset of its kind. This international collaboration ensured that the findings were not localized to a specific demographic but represented a fundamental characteristic of the human brain’s response to injury.
"By pooling data from hundreds of stroke survivors worldwide and applying cutting-edge AI, we can detect subtle patterns of brain reorganization that would be invisible in smaller studies," said Arthur W. Toga, PhD, director of the Stevens INI and Provost Professor at USC. "These findings of regionally differential brain aging in chronic stroke could eventually guide personalized rehabilitation strategies."
Context: The Global Burden of Stroke and Recovery
To understand the significance of this research, one must consider the global impact of stroke. According to the World Stroke Organization, one in four adults over the age of 25 will have a stroke in their lifetime. Annually, over 12 million people worldwide suffer a stroke, and it remains a leading cause of long-term disability.
Current rehabilitation protocols often rely on physical therapy and occupational therapy to "retrain" the brain. However, recovery is notoriously inconsistent. Some patients regain near-total function, while others remain severely impaired despite years of effort. Neurologists have long suspected that the difference lies in neuroplasticity, but they lacked a precise way to measure it in a clinical setting.
The discovery of contralesional rejuvenation provides a new lens through which to view these inconsistencies. If the "younger" brain age is a marker of the brain’s attempt to compensate, doctors might one day use MRI-based brain aging scores to predict which patients are most likely to benefit from specific types of therapy. For instance, a patient showing strong signs of contralesional reorganization might be a candidate for advanced therapies like Transcranial Magnetic Stimulation (TMS), which can stimulate specific brain networks to enhance the natural compensation process.
Timeline of Discovery and Future Directions
The journey toward these findings began several years ago with the formation of the ENIGMA Stroke Recovery Working Group, which aimed to harmonize disparate datasets from around the world.
- Phase 1: Data Harmonization. Researchers spent years collecting and standardizing MRI scans and clinical motor scores from international partners, ensuring that data from a machine in Norway could be accurately compared with data from a machine in Australia.
- Phase 2: Model Training. The USC team developed and trained the graph convolutional network using large-scale datasets of healthy brains to establish a reliable "aging clock."
- Phase 3: Cross-Sectional Analysis. The team applied the model to the chronic stroke dataset, leading to the current publication in The Lancet Digital Health.
- Phase 4: Longitudinal Tracking (Ongoing). The researchers are now moving toward longitudinal studies, following the same patients over months and years to see how brain-PAD changes as they progress through different stages of recovery.
The next step for the USC Stevens INI team is to determine whether this "youthful" structural change is always beneficial. While it indicates neuroplasticity, it occurs most prominently in those with the most severe impairments. This suggests that while the brain is trying to help, the compensation may not be efficient enough to restore full movement. Future research will investigate how to make this reorganization more effective.
Implications for Personalized Medicine
The long-term goal of this research is the development of personalized stroke recovery tracks. Currently, most stroke survivors receive a standardized course of rehabilitation. However, if a physician can see that a patient’s frontoparietal network is already showing signs of "rejuvenation," they might tailor the therapy to lean into that specific network’s strengths.
Furthermore, this research has implications beyond stroke. The concept of regional brain aging could be applied to other neurological conditions, such as traumatic brain injury (TBI), multiple sclerosis, or even early-stage Alzheimer’s disease. By understanding how the brain attempts to protect itself or compensate for loss, scientists can develop interventions that support these natural biological processes.
The study was supported by the National Institutes of Health (NIH) grant R01 NS115845, with additional support from various international institutions, including the University of British Columbia, Monash University, Emory University, and the University of Oslo. As the field of neuroimaging continues to integrate with artificial intelligence, the "black box" of the human brain is slowly being opened, revealing a resilient organ that, even in the face of severe trauma, attempts to find a way back to youth.














Leave a Reply