Researchers from the Department of Medicine at the School of Clinical Medicine, LKS Faculty of Medicine, University of Hong Kong (HKUMed) have achieved a significant breakthrough in the field of musculoskeletal health by identifying a specific biological process that explains how physical activity maintains bone density. This discovery, centered on a protein that functions as an internal "exercise sensor," provides a potential molecular blueprint for developing new treatments for osteoporosis. These findings are particularly critical for individuals who are physically unable to perform weight-bearing exercises, such as the elderly, bedridden patients, and those suffering from chronic debilitating illnesses.
The study, published in the prestigious peer-reviewed journal Signal Transduction and Targeted Therapy, highlights the role of a protein known as Piezo1. Located on the surface of mesenchymal stem cells within the bone marrow, Piezo1 acts as a mechanical sensor that detects the physical forces generated during movement. When activated, it triggers a series of signaling pathways that promote bone formation while simultaneously inhibiting the accumulation of fat within the bone marrow. This dual action is vital for maintaining the structural integrity of the skeleton throughout the aging process.
The Biological Mechanism of Bone Maintenance and Decay
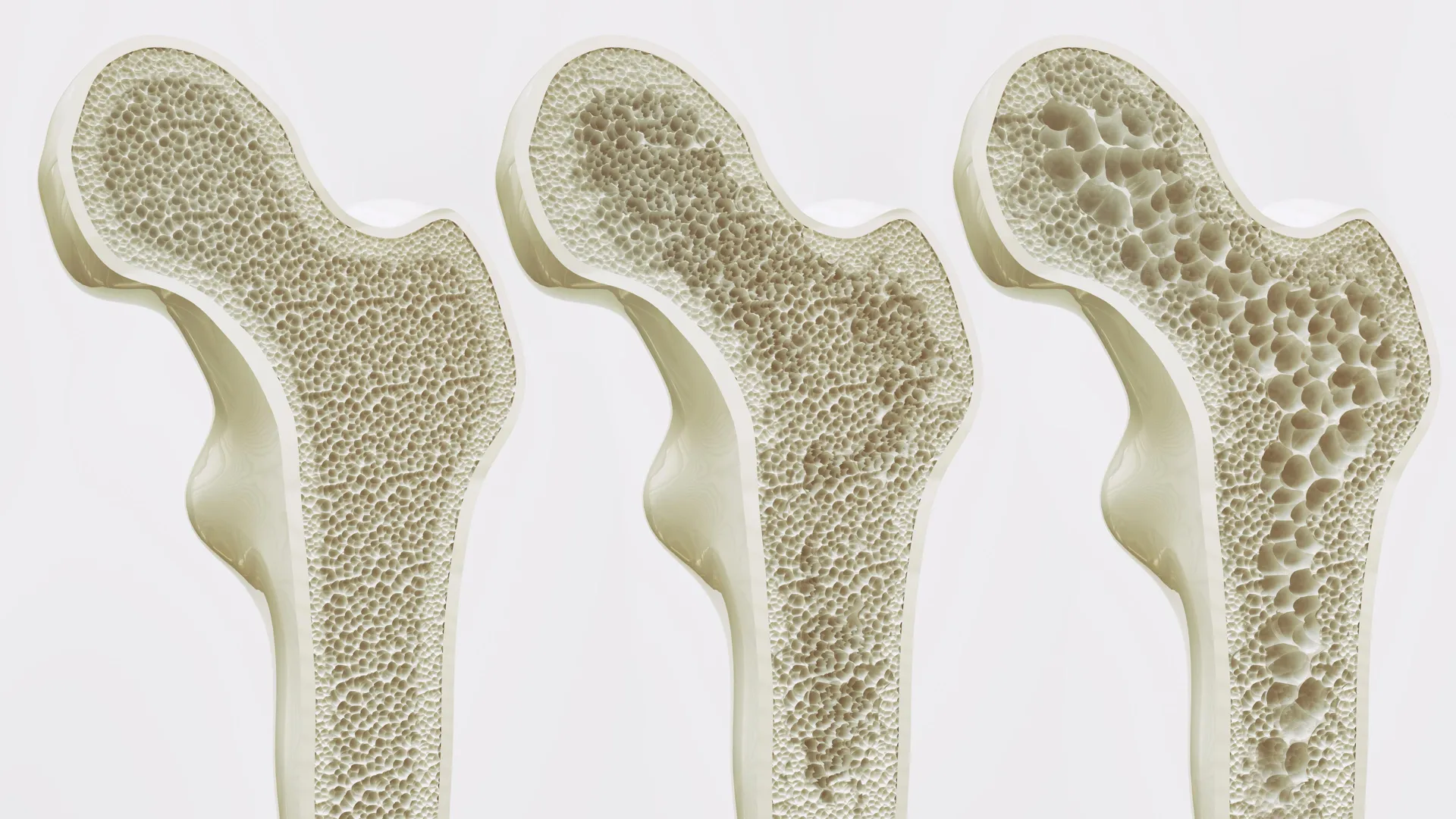
To understand the significance of this discovery, it is necessary to examine the internal environment of the bone marrow. Bone marrow contains mesenchymal stem cells (MSCs), which are versatile cells capable of differentiating into various types of tissue, including bone-forming cells (osteoblasts) and fat cells (adipocytes). In a healthy, active individual, physical mechanical stress—such as walking, running, or lifting—signals these stem cells to prioritize the creation of new bone.
However, as the human body ages, this biological balance begins to shift. A phenomenon known as "lineage misallocation" occurs, where mesenchymal stem cells increasingly favor the production of fat cells over bone tissue. This shift leads to a condition where the interior of the bone becomes increasingly porous and fatty, a hallmark of osteoporosis. As fat accumulates in the marrow, it effectively crowds out healthy bone-forming activity, leading to a rapid decline in bone mineral density and an increased risk of catastrophic fractures.
The HKUMed research team, led by Professor Xu Aimin, Director of the State Key Laboratory of Pharmaceutical Biotechnology, sought to identify the exact "switch" that controls this decision-making process in stem cells. Through a series of sophisticated experiments involving mouse models and human stem cell cultures, they identified Piezo1 as the primary mechanosensor responsible for translating physical movement into chemical signals that favor bone growth.
The Role of Piezo1 as a Mechanical Sensor
The research team observed that when Piezo1 is activated through physical activity, it acts as a gatekeeper. In mice, the presence of Piezo1 limited fat buildup and stimulated the production of new bone tissue. Conversely, when the protein was genetically removed or remained inactive due to a lack of physical stimulation, the stem cells rapidly converted into fat cells. This absence of Piezo1 activity also triggered the release of specific inflammatory signals, namely Ccl2 and lipocalin-2.
These inflammatory markers were found to create a "pro-fat" environment within the bone marrow. The presence of Ccl2 and lipocalin-2 further pushed mesenchymal stem cells toward fat production, creating a feedback loop of bone deterioration. Crucially, the researchers demonstrated that by blocking these specific inflammatory signals, they could partially restore healthier bone conditions, suggesting a secondary route for therapeutic intervention alongside the activation of Piezo1 itself.
"We have essentially decoded how the body converts movement into stronger bones," stated Professor Xu Aimin. "We have identified the molecular exercise sensor, Piezo1, and the signaling pathways it controls. This gives us a clear target for intervention. By activating the Piezo1 pathway, we can mimic the benefits of exercise, effectively tricking the body into thinking it is exercising, even in the absence of movement."
Supporting Data: The Global and Local Burden of Osteoporosis
The implications of this research are underscored by the staggering global statistics regarding bone health. According to the World Health Organization (WHO), osteoporosis is a "silent epidemic" because bone loss occurs without symptoms until a fracture happens. Globally, it is estimated that one in three women and one in five men over the age of 50 will suffer an osteoporotic fracture. These injuries are not merely physical setbacks; they are often life-altering events that lead to long-term disability, chronic pain, and a significant decrease in life expectancy.
In Hong Kong, the challenge is particularly acute due to the city’s rapidly aging demographic. Current data indicates that osteoporosis affects approximately 45% of women and 13% of men aged 65 and older in the territory. The healthcare costs associated with treating hip fractures and providing long-term rehabilitative care place an immense strain on the public health system. Furthermore, for many elderly patients, a single fracture can lead to a permanent loss of independence, requiring transition into residential care facilities.
The research team notes that current pharmacological treatments for osteoporosis, such as bisphosphonates or hormone-related therapies, often come with side effects or limitations in long-term efficacy. Moreover, most existing medical advice emphasizes weight-bearing exercise as a primary preventative measure—advice that is impossible to follow for patients who are bedbound due to stroke, advanced frailty, or severe injury.
Perspectives from the Research Leadership
The study was a collaborative international effort, involving experts from Hong Kong and France. Dr. Wang Baile, Research Assistant Professor at HKUMed and co-leader of the study, emphasized the humanitarian aspect of the discovery. "This discovery is especially meaningful for older individuals and patients who cannot exercise due to frailty, injury, or chronic illness," Dr. Wang said. "Our findings open the door to developing ‘exercise mimetics’—drugs that chemically activate the Piezo1 pathway to help maintain bone mass and support independence."
Professor Eric Honoré, a team leader at the Institute of Molecular and Cellular Pharmacology of the French National Centre for Scientific Research (CNRS), highlighted the broader scientific impact of the study. He noted that the identification of Piezo1 as a bone-specific sensor offers a strategy that goes beyond traditional physical therapy. "In the future, we could potentially provide the biological benefits of exercise through targeted treatments, thereby slowing bone loss in vulnerable groups and substantially reducing their risk of fractures," Honoré explained.
The collaboration between HKUMed and French institutions like the Université Côte d’Azur and Inserm underscores the global importance of the research. By pooling resources and expertise in molecular pharmacology and biotechnology, the team was able to map the complex transition from mechanical force to cellular differentiation.
Analysis of Implications and Future Clinical Translation
The identification of Piezo1 represents a pivot point in the study of "mechanotransduction"—the process by which cells convert mechanical stimuli into electrochemical activity. While mechanotransduction has long been recognized in the sense of touch and hearing, its role in bone metabolism was less clearly defined until now.
The concept of "exercise mimetics" is a burgeoning field in medical science. If a pharmaceutical agent can successfully activate the Piezo1 pathway, it could theoretically provide the skeletal benefits of a daily walk or jog to a patient who is paralyzed or recovering from surgery. This would not only prevent the immediate degradation of bone tissue but also prevent the systemic inflammation associated with marrow fat accumulation.
However, the path from a laboratory discovery to a clinical treatment involves several more years of rigorous testing. The HKUMed team is currently focused on translating these findings into clinical applications. The next steps involve identifying small molecules that can safely and effectively target Piezo1 in humans without causing adverse effects in other tissues where the protein might be present.
Funding and Institutional Support
The scale of this research was made possible through extensive support from multiple international and local funding bodies. In Hong Kong, the project received backing from the Areas of Excellence Scheme and the General Research Fund of the Research Grants Council, as well as the Health and Medical Research Fund under the Health Bureau.
International support was provided by the National Key R&D Program of China, the National Natural Science Foundation of China, the Human Frontier Science Program, and several French organizations including the French National Research Agency and Fondation de France. The Macau Science and Technology Development Fund also contributed to the study, reflecting a regional commitment to addressing the health challenges of an aging population.
As the global population continues to age, the need for innovative solutions to age-related diseases becomes increasingly urgent. The discovery of the Piezo1 exercise sensor by the HKUMed team offers a promising new frontier in the fight against osteoporosis, providing hope that the strength and independence of the elderly can be preserved through the power of molecular medicine. This research not only advances our understanding of human biology but also sets the stage for a future where the health benefits of movement can be accessed by all, regardless of their physical limitations.














Leave a Reply