Indianapolis-based pharmaceutical giant Eli Lilly and Company has achieved a remarkable milestone, surpassing all global pharmaceutical competitors to claim the top spot in revenue for fiscal year 2025. This historic financial triumph coincides with another significant regulatory victory: the U.S. Food and Drug Administration (FDA) approved Foundayo (orforglipron) on April 1st, a groundbreaking once-daily oral GLP-1 receptor agonist for obesity. Foundayo represents the first oral GLP-1 specifically approved for weight management in a small molecule format, and its expedited clearance marks the fastest new molecular entity approval since 2002. Despite these monumental achievements, Wall Street has exhibited a muted response, with Lilly’s stock experiencing a nearly 14% decline since the beginning of January. This dichotomy of unprecedented corporate success and investor skepticism underscores the complex dynamics at play within the highly competitive and rapidly evolving pharmaceutical landscape.
Lilly’s Unprecedented Ascent to Pharmaceutical Leadership
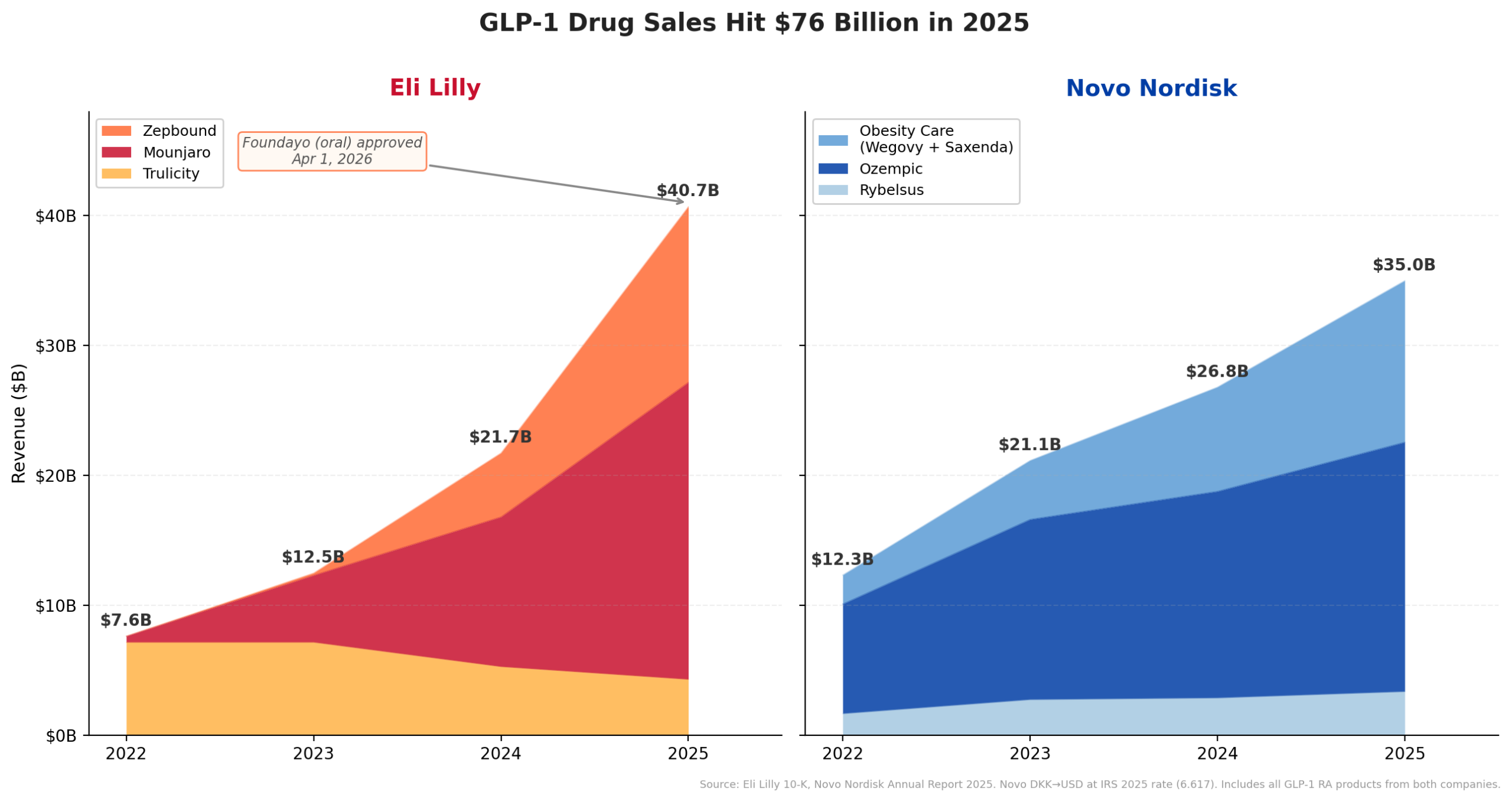
Eli Lilly’s financial performance in fiscal year 2025 was nothing short of extraordinary, with the company reporting a staggering $65.18 billion in revenue. This figure represents a colossal 44.7% year-over-year growth rate, positioning Lilly not only as the fastest-growing among the top 20 pharmaceutical companies but also vaulting it past long-standing industry titans. For the first time in its history, Lilly outstripped Merck & Co., which posted $65.01 billion, Pfizer ($62.58 billion), and AbbVie ($61.16 billion). While the revenue margin over Merck was a narrow $170 million, the divergence in growth trajectories was stark: Lilly’s nearly 45% expansion dwarfed Merck’s modest 1.3%. This dramatic shift in the industry hierarchy signals a new era of leadership, driven predominantly by the burgeoning demand for innovative metabolic disease therapies.
The driving force behind this monumental surge is unequivocally Lilly’s portfolio of traditional injectable GLP-1 receptor agonists. These drugs have reshaped the treatment paradigms for type 2 diabetes and, more recently, obesity. Mounjaro (tirzepatide), initially approved for type 2 diabetes, generated an astonishing $9.0 billion in the first half of 2025 alone, marking a 90% increase year-over-year. Its sister product, Zepbound (also tirzepatide), specifically indicated for chronic weight management, contributed an additional $5.7 billion in H1 2025. Notably, Zepbound was only launched in late 2023 and has rapidly gained traction, now available in a convenient multi-dose KwikPen. Combined, Lilly’s GLP-1 offerings are on track to exceed an annualized revenue of $30 billion, demonstrating their immense market penetration and therapeutic impact.
The Foundayo Approval: A New Frontier in Oral Therapy
The recent FDA approval of Foundayo (orforglipron) on April 1st represents a strategic expansion of Lilly’s already formidable GLP-1 franchise. This approval, granted under the FDA’s Commissioner’s National Priority Voucher program, was exceptionally swift, occurring just 50 days after filing. This expedited review, nearly 10 months ahead of its scheduled date, underscores the FDA’s recognition of orforglipron’s potential public health significance. The National Priority Voucher program is designed to incentivize the development of novel therapies that address critical unmet medical needs or offer significant advancements, often leading to accelerated review pathways.
Foundayo stands out in the crowded GLP-1 market due to its unique characteristics. Until recently, the landscape of blockbuster GLP-1 drugs, including Novo Nordisk’s Ozempic and Wegovy, and Lilly’s Mounjaro and Zepbound, consisted predominantly of injectable formulations. While Novo Nordisk did receive approval for an oral formulation of semaglutide (Rybelsus for diabetes, and an oral Wegovy for obesity in December 2025), these are peptide-based drugs that require specific administration conditions—taken on an empty stomach with restricted water intake to ensure optimal absorption. Foundayo, conversely, is a small molecule GLP-1 receptor agonist. This chemical distinction provides a significant practical advantage: it can be taken once daily at any time, without food or water restrictions, thereby offering unprecedented convenience and flexibility for patients.
Strategic Vision and Market Expansion
Lilly CEO Dave Ricks has articulated a clear strategic vision for Foundayo. While acknowledging that Foundayo may not achieve the same level of weight loss efficacy as the company’s injectable Zepbound (patients in ATTAIN-1 trials on the highest dose lost an average of 12.4% of body weight over 72 weeks, compared to over 20% with injectable tirzepatide), Ricks emphasized its role as a complementary, rather than competitive, offering. As he told CNBC, Foundayo is "not more effective" but "more accessible, it’s easier to fit into your daily routine." This positioning reflects a broader market strategy to cater to diverse patient needs and preferences. Ricks’s overarching goal is to ensure that "people are on the medicine that meets their health goals. If it has Lilly on the box, that’s the goal we have."
The introduction of an oral GLP-1 option for obesity is expected to significantly expand the addressable patient population. Many individuals are hesitant or unable to manage injectable medications, making an easily administered oral alternative a game-changer for broader market penetration. This strategic move aligns with the explosive growth of the GLP-1 market, which has ballooned from under $12 billion in 2022 to an estimated $65 billion annualized run rate by mid-2025. Lilly’s commitment to this expansion is further evidenced by its aggressive global strategy, having already submitted orforglipron for approval in over 40 countries worldwide. The initial licensing of orforglipron from Japanese drugmaker Chugai in 2018 for $50 million upfront now appears to be a highly prescient investment, yielding substantial returns and strategic advantage.
Wall Street’s Measured Response: A Puzzle of Perception
Despite Eli Lilly’s remarkable financial performance and the strategic coup of Foundayo’s approval, the market’s reaction has been surprisingly subdued, with the stock declining nearly 14% since January 1st. This seemingly contradictory response invites analysis into the underlying factors influencing investor sentiment.
Several hypotheses emerge to explain Wall Street’s cautious stance. Firstly, the "buy the rumor, sell the news" phenomenon might be at play. The immense potential of Lilly’s GLP-1 pipeline, including oral candidates, has been widely anticipated and likely priced into the stock over the past year, leading to a period of consolidation or profit-taking following official announcements. Lilly’s stock has seen substantial gains in previous periods, reflecting high expectations.
Secondly, valuation concerns may be weighing on investors. With its unprecedented growth, Lilly’s market capitalization has soared, potentially leading some analysts to believe that the current valuation already reflects much of the future growth potential, leaving less room for significant upside in the short term. The challenge for Lilly now is to continuously exceed these elevated expectations.
Thirdly, the competitive landscape, while currently dominated by Lilly and Novo Nordisk, remains intensely dynamic. While Foundayo offers a unique convenience advantage, its slightly lower efficacy compared to injectables and the presence of Novo Nordisk’s own oral GLP-1s (Rybelsus and oral Wegovy) could introduce complexities. Investors might be factoring in the long-term competition and the potential for new entrants or further advancements in the GLP-1 space. The market may also be considering the immense manufacturing and supply chain challenges associated with scaling production for such high-demand drugs, which could impact profitability or market share in the short to medium term.
Finally, broader market trends and macroeconomic factors cannot be entirely discounted. General market volatility or shifts in investor preference away from growth stocks in certain sectors could also contribute to the observed stock performance, even for a company demonstrating such robust fundamentals.
The Broader Impact and Future Outlook
Lilly’s ascent to the top of the pharmaceutical revenue charts, powered by its GLP-1 franchise and strategically bolstered by Foundayo, signifies a pivotal moment for the industry. This achievement not only redefines leadership but also underscores the profound impact of innovative therapies in addressing prevalent global health challenges like obesity and type 2 diabetes. The company’s aggressive investment in R&D and its strategic acquisitions and licensing agreements have clearly paid dividends.
Looking ahead, Lilly faces the dual challenge of sustaining its unprecedented growth and managing the complex expectations of the market. Its diversified GLP-1 portfolio, now including a highly convenient oral small molecule, positions it strongly for continued market expansion and patient reach. The company’s focus on complementary therapies, rather than direct competition within its own pipeline, suggests a sophisticated strategy to maximize market share across different patient segments.
The GLP-1 market is projected to continue its explosive growth, with analysts forecasting it to reach hundreds of billions of dollars in the coming decade. Lilly, alongside its primary competitor Novo Nordisk, is poised to be at the forefront of this revolution. However, maintaining leadership will require continuous innovation, effective global market penetration, and the ability to navigate potential regulatory and competitive headwinds. The cautious reaction from Wall Street, while perplexing at first glance, serves as a reminder that even in moments of historic triumph, the pharmaceutical industry demands relentless performance and strategic foresight to justify premium valuations. Lilly’s journey to the summit is a testament to its innovation, but the path to maintaining that position will be equally challenging and closely watched by investors and industry observers alike.














Leave a Reply