A team of scientists at King’s College London has identified a new form of aluminum, one of the most abundant metals on Earth, that could offer a far less expensive and more sustainable alternative to widely used rare earth metals. Led by Dr. Clare Bakewell, a Senior Lecturer in the Department of Chemistry, the researchers successfully synthesized highly reactive aluminum molecules capable of breaking some of the strongest chemical bonds known to science. Their findings, recently published in the prestigious journal Nature Communications, reveal entirely new molecular structures that open the door to previously unknown types of chemical behavior, potentially disrupting the global supply chain for industrial catalysts.
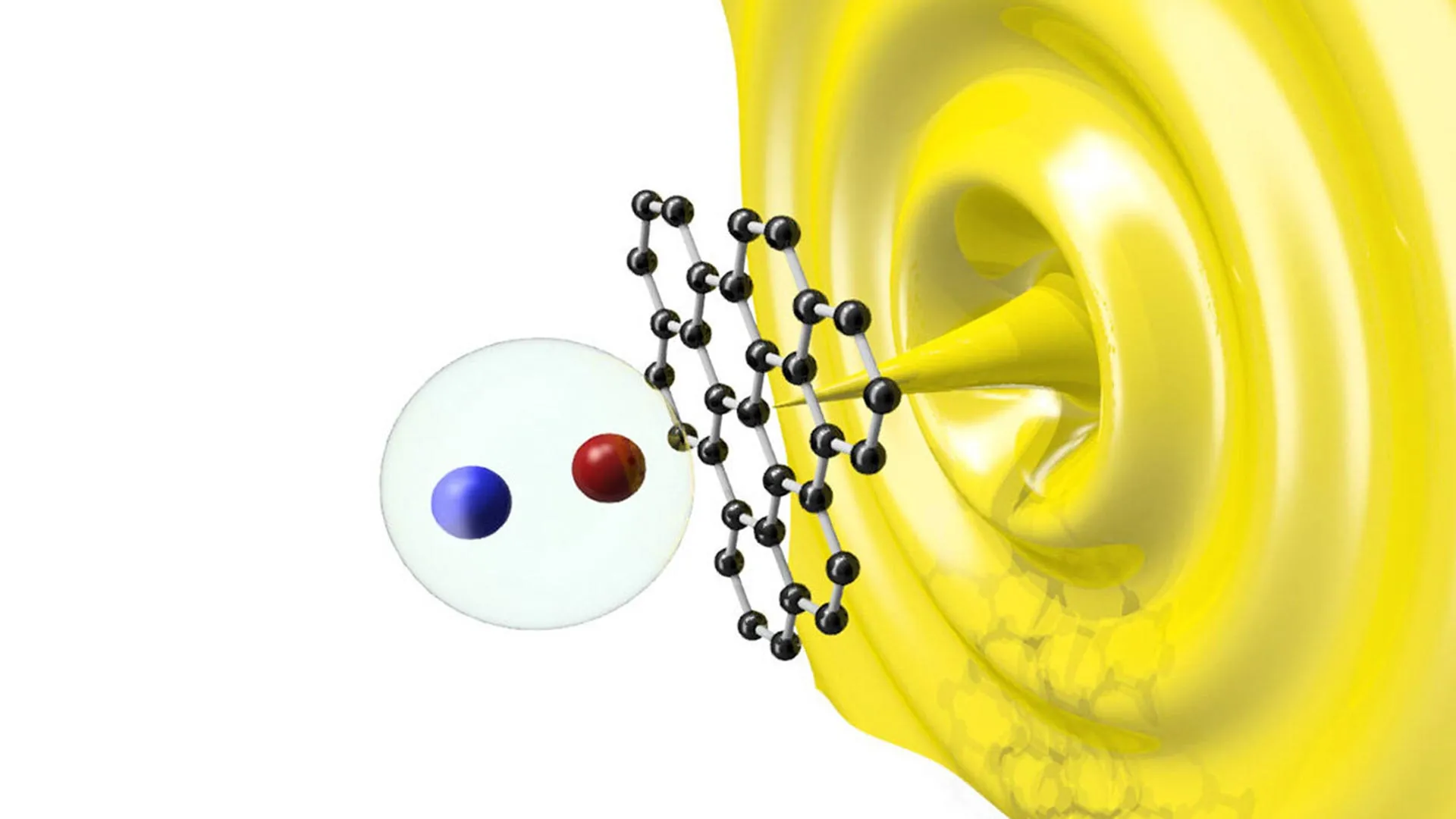
The discovery centers on the first known example of a cyclotrialumane, a compound consisting of three aluminum atoms arranged in a trimeric, triangular configuration. This unusual geometric arrangement provides the molecule with remarkable reactivity, allowing it to perform tasks typically reserved for expensive transition metals such as platinum, palladium, and rhodium. Crucially, the researchers noted that the structure remains intact even when dissolved in various solutions, providing the necessary stability for practical applications in diverse chemical reactions.
The Scientific Breakthrough: Understanding the Cyclotrialumane Structure
The core of the discovery lies in the unique bonding environment of the aluminum atoms. In standard industrial chemistry, transition metals are prized for their ability to donate and accept electrons easily, a property that allows them to facilitate complex transformations. Aluminum, traditionally classified as a "main group" metal, has historically been viewed as less versatile in these specific roles. However, the King’s College London team has challenged this paradigm by engineering a low-valent aluminum trimer.
By arranging three aluminum atoms in a triangle, the researchers created a molecule with a high degree of "strain" and electron density that mimics the behavior of transition metals. This cyclotrialumane can split dihydrogen (H2) molecules and enable the step-by-step insertion and chain growth of ethene, a fundamental two-carbon hydrocarbon used in the production of plastics and various synthetic materials. The ability to manipulate ethene in this manner is a cornerstone of modern industrial chemistry, and achieving it with aluminum represents a significant leap forward in organometallic research.
The stability of the compound in solution is particularly noteworthy. Many highly reactive molecules are "labile," meaning they break down or rearrange themselves too quickly to be useful in a controlled laboratory or industrial setting. The King’s College London team found that their aluminum trimer maintains its structural integrity, allowing it to act as a reliable scaffold for building more complex molecular architectures.
Economic and Geopolitical Context: The Search for Abundant Alternatives
The drive toward aluminum-based chemistry is fueled by the escalating costs and logistical challenges associated with precious metals. For decades, transition metals have served as the "workhorses" of chemical synthesis. Platinum, for instance, is essential for catalytic converters in vehicles and various pharmaceutical manufacturing processes. However, these metals are not only rare but are often concentrated in geographically and politically sensitive regions.
According to Dr. Bakewell, the reliance on these elements poses a long-term risk to global industry. "Transition metals are the workhorses of chemical synthesis and catalysis," Bakewell stated. "But many of the most useful are becoming increasingly difficult to access and extract—often being located in regions of political instability, increasing the demand and price."
The economic disparity between aluminum and its precious metal counterparts is staggering. Aluminum is the most abundant metal in the Earth’s crust, making up approximately 8.1% of its mass. In contrast, metals like platinum and palladium are found in concentrations of only parts per billion. This abundance translates directly to cost efficiency; the researchers estimate that aluminum is approximately 20,000 times less expensive than precious metals like platinum. By shifting the burden of catalysis from rare elements to aluminum, the chemical industry could see a dramatic reduction in overhead costs and a stabilization of supply chains currently vulnerable to geopolitical shocks.
Expanding the Frontiers of Chemical Knowledge
Beyond the immediate goal of mimicking transition metals, the King’s College London team is uncovering reactions that were previously thought impossible for aluminum. This "new frontier" of chemistry suggests that aluminum might not just be a cheaper substitute, but a superior tool for certain niche applications.
One of the most exciting aspects of the research is the formation of 5- and 7-membered aluminum and carbon rings through reactions with ethene. These complex ring structures are difficult to synthesize using traditional methods. "What’s special about this work is that we’re pushing the boundaries of chemical knowledge," Dr. Bakewell explained. "Most excitingly, we can use this aluminum trimer to build completely new compounds with levels of reactivity that have never been observed before. These capabilities go beyond the transition metals we were originally trying to mimic, to the forefront of chemical research."
The creation of these rings indicates that the aluminum trimer can facilitate "chain growth," a process where small molecules are linked together to form larger, more complex structures. This is a vital process in the creation of polymers, advanced materials, and specialized chemical intermediates used in medicine and agriculture.
Chronology of Discovery and Peer Validation
The path to this discovery involved years of fundamental research into the properties of Group 13 elements. The King’s College London Department of Chemistry has long been a hub for organometallic innovation, focusing on how earth-abundant elements can be "tuned" through ligand design to exhibit non-traditional reactivity.
- Phase One: Ligand Design: The team developed specific organic "stabilizers" (ligands) that could surround the aluminum atoms, preventing them from reacting prematurely with the environment while leaving enough space for target molecules like ethene to interact with the metal core.
- Phase Two: Synthesis of the Trimer: Through a series of controlled reduction reactions, the researchers succeeded in isolating the triangular cyclotrialumane.
- Phase Three: Reactivity Testing: The team subjected the compound to various substrates, confirming its ability to break H-H bonds and facilitate the insertion of ethene.
- Phase Four: Publication and Peer Review: The findings were documented and submitted to Nature Communications, where they underwent rigorous peer review before being published. The scientific community has reacted with significant interest, as the paper provides a blueprint for further exploration into low-valent main group chemistry.
Implications for Green Chemistry and Sustainability
The environmental impact of traditional metal extraction is a growing concern for global regulators. Mining for platinum group metals (PGMs) is an energy-intensive process that often involves the use of toxic chemicals and results in significant carbon emissions. Furthermore, the "catalytic footprint" of a product—the environmental cost of the metals used to make it—is a key metric in the transition toward a circular economy.
Aluminum mining (primarily through bauxite) is also an industrial process with an environmental footprint, but the sheer abundance of the metal and the existing, highly efficient global recycling infrastructure for aluminum make it a much more sustainable candidate for long-term industrial use. If aluminum-based catalysts can be successfully scaled, they would contribute to "Green Chemistry" goals by:
- Reducing the energy required for metal extraction.
- Minimizing the ecological damage associated with rare earth mining.
- Lowering the toxicity of industrial waste streams, as aluminum is generally less toxic than heavy transition metals.
Future Outlook: From the Laboratory to the Factory Floor
While the discovery is a landmark achievement, Dr. Bakewell and her team are careful to manage expectations regarding immediate industrial implementation. The research is currently in the "exploratory phase," where the primary goal is to understand the fundamental mechanics of the new aluminum structure.
"We’re very much in the exploratory phase and we’re just at the start of beginning to unlock the capability of these earth-abundant materials," Bakewell noted. "But from what we’ve seen already, this chemistry could support a transition to cleaner, greener and cheaper chemical production, whilst making new discoveries along the way."
The next steps for the research team involve testing the aluminum trimer against a wider array of chemical bonds and exploring its potential in "catalytic cycles"—where the molecule can perform a reaction, release the product, and then return to its original state to start the process again. Achieving a true catalytic cycle is the "holy grail" for this technology, as it would allow small amounts of the aluminum compound to produce large quantities of chemical products, further increasing its economic viability.
As the global community seeks to decouple industrial growth from environmental degradation and volatile resource markets, the work coming out of King’s College London provides a promising roadmap. By reimagining the potential of one of the world’s most common metals, Dr. Bakewell and her colleagues are not just finding a cheaper way to conduct chemistry; they are redefining the limits of what common elements can achieve.














Leave a Reply