A pioneering research initiative led by the University of Adelaide is charting a new course for the global energy transition by transforming one of the world’s most persistent environmental threats—plastic waste—into a clean energy resource. By utilizing solar-driven chemical processes, researchers have demonstrated a viable pathway to convert discarded polymers into high-value hydrogen fuel and industrial chemicals. This breakthrough, recently detailed in the journal Chem Catalysis, arrives at a critical juncture as international bodies struggle to manage an escalating plastic crisis while simultaneously racing to meet decarbonization targets.
The study, spearheaded by PhD candidate Xiao Lu and senior author Professor Xiaoguang Duan from the School of Chemical Engineering, highlights a process known as solar-driven photoreforming. Unlike traditional recycling, which often results in "downcycling" materials into lower-quality products, this method functions as a form of high-tech upcycling. It leverages the inherent chemical energy stored within the carbon-hydrogen bonds of plastic, using nothing more than sunlight and specialized catalysts to trigger the transformation.
The Global Plastic Crisis as a Resource Reservoir
To understand the significance of the Adelaide research, one must consider the staggering scale of the problem it seeks to solve. According to the Organisation for Economic Co-operation and Development (OECD), global plastic production has doubled over the last two decades, reaching approximately 460 million tonnes annually. Of this, less than 10% is successfully recycled. The remainder is either incinerated—releasing significant greenhouse gas emissions—or ends up in landfills and the natural environment, where it can persist for centuries.
However, from a chemical perspective, this waste is a massive, untapped reservoir of energy. Plastics are primarily composed of long chains of hydrocarbons. "Plastic is often seen as a major environmental problem, but it also represents a significant opportunity," Ms. Lu noted during the presentation of the findings. "If we can efficiently convert waste plastics into clean fuels using sunlight, we can address pollution and energy challenges at the same time."
The research identifies that the carbon and hydrogen density in common plastics like polyethylene (PE), polypropylene (PP), and polyvinyl chloride (PVC) makes them ideal candidates for fuel conversion. By viewing these materials as a "secondary raw material" rather than trash, the team aims to facilitate a transition toward a truly circular economy, where the end-of-life for one product serves as the feedstock for the next generation of energy.
The Science of Solar Photoreforming
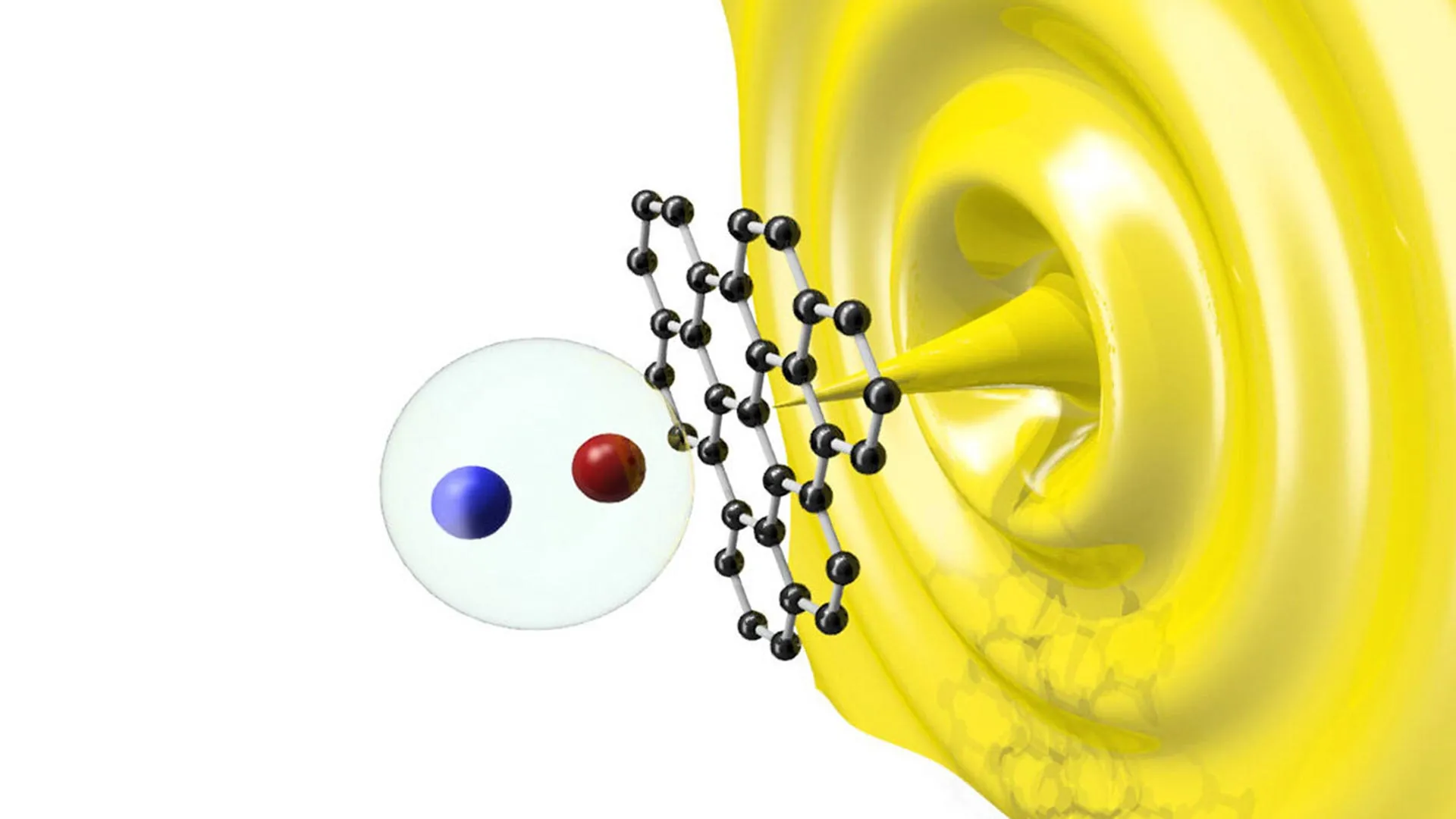
The core of the Adelaide team’s innovation lies in the use of photocatalysts—materials that can absorb solar energy and use it to drive chemical reactions. In the photoreforming process, these catalysts are placed in contact with plastic waste in an aqueous solution. When exposed to sunlight, the catalysts generate electron-hole pairs. these highly reactive species break the sturdy chemical bonds of the plastic polymers.
This process offers several distinct advantages over existing waste-to-energy technologies:
- Temperature Efficiency: Traditional gasification or pyrolysis requires temperatures exceeding 500 to 800 degrees Celsius, requiring massive energy inputs. Photoreforming occurs at near-ambient temperatures, significantly reducing the carbon footprint of the conversion process itself.
- Lower Energy Barriers: Producing hydrogen through traditional water splitting is energy-intensive because the oxygen evolution reaction is difficult to trigger. In photoreforming, the plastic acts as a "sacrificial agent" that is easier to oxidize than water, thereby lowering the overall energy required to produce hydrogen gas.
- Product Diversity: The process does not just yield hydrogen. Depending on the catalyst and the conditions, it can produce syngas (a mixture of hydrogen and carbon monoxide), acetic acid, and even diesel-range hydrocarbons, all of which have established markets in the chemical and transport industries.
Chronology of Development and Experimental Milestones
The journey toward solar-powered plastic conversion has evolved rapidly over the last decade. Early experiments in the mid-2010s focused on pure, laboratory-grade plastics and often yielded only trace amounts of fuel. However, the University of Adelaide’s recent study represents a significant leap forward in terms of both stability and output.
During the experimental phase, the researchers tested various photocatalyst compositions to identify which could best withstand the harsh chemical environments created by breaking down complex polymers. The team reported that some of their optimized systems were able to run continuously for over 100 hours without significant degradation. This level of stability is a crucial benchmark for moving technology from a laboratory setting to a pilot plant.
Furthermore, the yields recorded were higher than many previous benchmarks. The researchers successfully demonstrated the production of high-purity hydrogen, which is increasingly viewed as the "fuel of the future" for heavy industry and long-haul shipping. The simultaneous production of acetic acid—a key component in the manufacturing of textiles, films, and solvents—adds a secondary revenue stream that could make the technology more economically attractive to private investors.
Addressing the Complexity of Real-World Waste
Despite the promising laboratory results, the transition to real-world application is fraught with engineering hurdles. Professor Xiaoguang Duan emphasized that "laboratory plastic" is very different from the waste found in a typical recycling bin.
"One major hurdle is the complexity of plastic waste itself," Prof. Duan explained. "Different types of plastics behave differently during conversion, and additives such as dyes, flame retardants, and stabilizers can interfere with the chemical process. Efficient sorting and pre-treatment are therefore essential to maximize performance and product quality."
In a real-world scenario, a solar-powered facility would need to handle a "cocktail" of different polymers. Polyethylene (found in plastic bags) reacts differently than Polyethylene Terephthalate (PET, found in soda bottles). Moreover, the presence of food waste or dirt can "poison" the catalysts, rendering them ineffective. This necessitates a robust front-end sorting system, potentially utilizing AI-driven optical sorting technology, to ensure the feedstock is compatible with the photocatalytic reactor.
Engineering Challenges and the Path to Scalability
Beyond the feedstock, the design of the reactors themselves remains a point of intense focus. For the technology to be viable, it must be able to process tonnes of plastic rather than grams. This requires large-scale solar collectors and reactors that allow for maximum light penetration.
Another significant challenge is product separation. Because the photoreforming process often produces a mixture of liquid and gaseous products, energy-intensive separation techniques are currently required to isolate pure hydrogen or specific chemicals. If the energy used to separate these products exceeds the energy gained from the fuel, the environmental benefit is negated.
To mitigate this, the Adelaide team is exploring "continuous-flow" reactors. Unlike batch reactors, where materials are processed in discrete groups, continuous-flow systems allow for a steady stream of plastic to be fed in and products to be extracted simultaneously. This approach is more compatible with industrial standards and tends to be more energy-efficient. Additionally, researchers are looking into "hybrid" systems that combine solar energy with small amounts of thermal or electrical energy to boost reaction rates during cloudy periods or at night.
Broader Implications for the Global Energy Landscape
The potential impact of this technology extends far beyond waste management. As nations strive to meet the goals of the Paris Agreement, the demand for "green hydrogen" is expected to skyrocket. Currently, most hydrogen is produced from natural gas through a process called steam methane reforming, which releases large amounts of CO2.
Solar-driven plastic-to-hydrogen technology offers a "double-green" alternative: it removes plastic from the environment while producing a carbon-neutral fuel. This could be particularly transformative for developing nations, which often bear the brunt of the global plastic waste trade but lack the expensive infrastructure required for traditional electrolysis or high-heat incineration.
Industrial analysts suggest that if the technology can be scaled, it could disrupt both the waste management and energy sectors. Waste management companies could transition into energy producers, and chemical manufacturers could source their raw materials from recycled carbon rather than virgin petroleum.
Future Outlook: A Decadal Roadmap
The Adelaide research team has outlined a clear roadmap for the next decade. The immediate focus will be on the development of "robust catalysts"—materials that are not only efficient but also cheap to manufacture and resistant to the chemical "poisoning" caused by plastic additives.
"There is still a gap between laboratory success and real-world application," Prof. Duan admitted. "We need more robust catalysts and better system designs to ensure the technology is both efficient and economically viable at scale."
The team expects that pilot-scale facilities could begin appearing within the next five to ten years, likely integrated into existing recycling centers or industrial parks. These pilots will serve as a proof-of-concept for larger, utility-scale installations.
As the world seeks to decouple economic growth from environmental degradation, the work being done at Adelaide University provides a glimpse into a future where "waste" is a word of the past. By harnessing the power of the sun to unlock the energy trapped in our discarded plastics, scientists are not just cleaning up the planet—they are fueling the next industrial revolution.
"This is an exciting and rapidly evolving field," Ms. Lu concluded. "With continued innovation, we believe solar-powered plastic-to-fuel technologies could play a key role in building a sustainable, low-carbon future."













Leave a Reply