In a landmark study that redefines the boundaries of neuroanatomy, researchers at the Medical University of South Carolina (MUSC) have identified a previously unrecognized control point in the brain’s waste clearance system. Published in the journal iScience, the research provides the first direct evidence in humans that the middle meningeal artery (MMA), a structure long considered solely a component of the circulatory system, serves as a primary conduit for the drainage of cerebrospinal and interstitial fluids. This discovery, facilitated by advanced imaging technology originally developed for NASA, marks a significant shift in the scientific understanding of how the human brain maintains its internal environment and removes metabolic waste.
For decades, the prevailing medical consensus was that the brain existed in a state of "immune privilege," separated from the body’s lymphatic system by the robust barriers of the meninges. However, over the last ten years, this paradigm has been steadily dismantled. The MUSC team, led by Onder Albayram, Ph.D., an associate professor in the Department of Pathology and Laboratory Medicine, has now pinpointed a specific anatomical "hub" where the brain’s waste-removal processes intersect with the body’s broader lymphatic network. By focusing on the MMA, the study highlights a critical mechanism that could be central to the development of neurodegenerative diseases, traumatic brain injuries, and age-related cognitive decline.
The Evolution of Neuroanatomical Understanding
The human brain is an incredibly metabolically active organ, consuming roughly 20% of the body’s energy despite accounting for only 2% of its weight. This high level of activity produces a significant volume of metabolic byproducts, including proteins like amyloid-beta and tau. In a healthy brain, these waste products are efficiently cleared out. When this system fails, the accumulation of these proteins is a hallmark of diseases such as Alzheimer’s and Parkinson’s.
Historically, the lymphatic system—a network of vessels that drains fluid from tissues and returns it to the bloodstream—was thought to be absent from the central nervous system. It was believed that the brain relied entirely on the slow diffusion of waste into the cerebrospinal fluid (CSF). This view began to change in 2015 when researchers identified lymphatic vessels in the meninges of mice. Dr. Albayram’s latest work builds upon this foundation, transitioning from animal models to provide high-resolution, real-time evidence of these processes occurring within the living human brain.
The focus on the middle meningeal artery is particularly revolutionary. The MMA is the largest of the three arteries that supply the dura mater, the outermost layer of the meninges. By identifying it as a lymphatic pathway, the MUSC study suggests that the brain’s drainage system is far more integrated with major cranial structures than previously realized.
NASA Technology Meets Neuroscience
The breakthrough was made possible through a unique technological collaboration involving NASA. The imaging tools employed by the MUSC team were originally engineered to investigate Spaceflight-Associated Neuro-ocular Syndrome (SANS), a condition where astronauts experience vision changes and brain swelling due to fluid shifts in microgravity. These advanced real-time MRI (Magnetic Resonance Imaging) sequences allow for the visualization of fluid movement at a much finer temporal and spatial resolution than standard clinical scans.
Using this technology, the research team monitored five healthy volunteers over a continuous six-hour period. This extended observation window was crucial for distinguishing between different types of fluid movement. While blood moves through the brain with high velocity and pulsatility driven by the cardiac cycle, the lymphatic drainage of the brain operates on a much slower, more rhythmic cadence.
The MRI data revealed that fluid was moving along the outer walls of the MMA at a slow, steady pace. "We saw a flow pattern that didn’t behave like blood moving through an artery; it was slower, more like drainage, showing that this vessel is part of the brain’s cleanup system," Dr. Albayram explained. This distinction is vital, as it confirms that the fluid is not part of the high-pressure circulatory system but is instead traveling through the "perivascular spaces" that surround the blood vessels—a key characteristic of the glymphatic and lymphatic systems.
Decoding the Mechanics of Fluid Transit
The study’s findings suggest a sophisticated "dual-purpose" role for the middle meningeal artery. While the interior of the artery carries oxygenated blood to the meninges, the exterior surface of the vessel appears to act as a scaffolding for lymphatic channels. This arrangement allows the brain to utilize the existing anatomical architecture of the circulatory system to facilitate waste removal.
The fluid observed moving along the MMA consists of a mixture of cerebrospinal fluid (CSF) and interstitial fluid (ISF). ISF is the fluid that bathes the brain’s cells, collecting metabolic waste as it circulates. The movement of this fluid into the lymphatic channels around the MMA represents a critical "exit ramp" for toxins. Once the waste enters these channels, it is transported toward the cervical lymph nodes in the neck, where the body’s immune system can process and eliminate it.
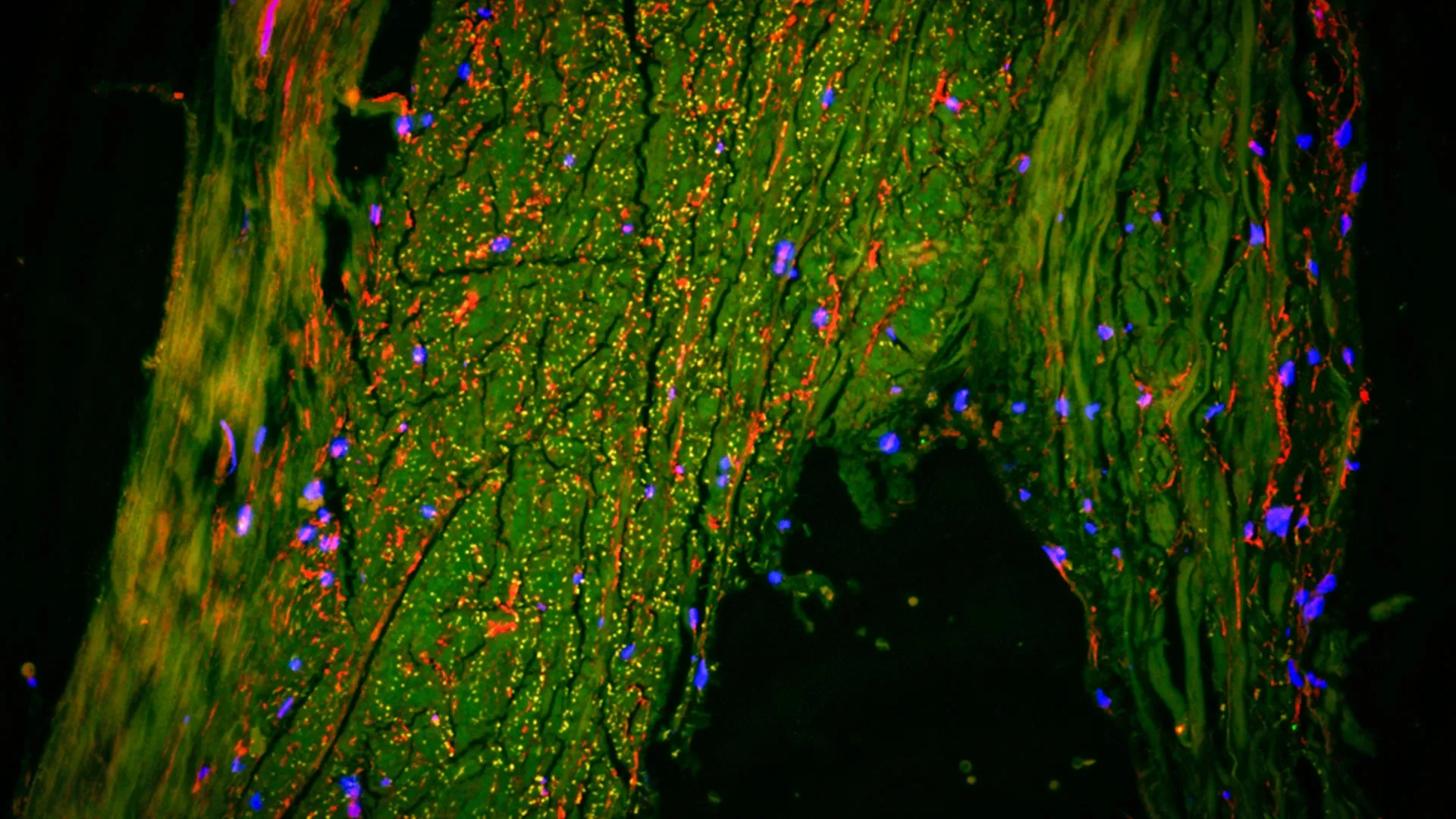
To validate the MRI findings, the MUSC team collaborated with researchers at Cornell University to perform ultra-high-resolution imaging on human brain tissue samples. Using multiplexed immunofluorescence—a technique that allows for the simultaneous visualization of multiple cell types—the researchers confirmed the presence of lymphatic endothelial cells (LECs) in the region surrounding the MMA. These cells are the building blocks of lymphatic vessels and are characterized by specific protein markers, such as LYVE-1 and PROX1. The presence of these cells provides biological confirmation that the slow-moving fluid detected by the NASA-derived MRI was indeed traveling through a dedicated lymphatic pathway.
Chronology of the Discovery
The identification of the MMA’s role is the culmination of years of targeted research. In 2021 and 2022, Dr. Albayram and his colleagues published work in Nature Communications that first visualized meningeal lymphatic vessels in humans using post-mortem tissue and specialized MRI. That earlier work established the existence of the vessels; however, it did not demonstrate their function in real-time.
The current study, published in 2024, represents a significant leap forward by capturing the actual flow of fluid. By moving from static anatomical observation to functional physiological monitoring, the researchers have bridged a major gap in the field. The timeline of this research suggests a rapidly accelerating understanding of the brain’s "plumbing," moving from theoretical models in the early 2010s to direct human observation in the 2020s.
Broader Implications for Clinical Medicine
The discovery of a major control point for brain waste clearance has profound implications for a wide range of medical conditions. The most immediate impact is expected in the study of Alzheimer’s disease. Current theories suggest that Alzheimer’s may be caused, at least in part, by a "clearance deficit"—a state where the brain produces waste faster than it can be removed. If the MMA pathway becomes restricted or damaged due to age or cardiovascular disease, it could lead to a backup of toxic proteins, accelerating the onset of dementia.
Beyond neurodegeneration, the findings offer new insights into:
- Traumatic Brain Injury (TBI): Following a head injury, the brain often experiences significant swelling and an influx of inflammatory debris. Understanding how the MMA facilitates drainage could lead to treatments that "prime" the lymphatic system to reduce intracranial pressure and clear inflammatory markers more quickly.
- Psychiatric Disorders: Emerging research has linked neuroinflammation to conditions such as depression and schizophrenia. If the brain’s "cleanup" system is inefficient, chronic low-grade inflammation may persist, affecting neurotransmitter function and mood.
- Aging and Lifestyle: As the body ages, blood vessels naturally become stiffer (arteriosclerosis). Because the lymphatic channels are physically attached to the MMA, the health of the circulatory system likely directly impacts the efficiency of the lymphatic system. This provides a clear biological link between heart health and brain health.
Future Directions and Research Objectives
The MUSC team is already moving into the next phase of their research. While this study established a baseline using healthy individuals, Dr. Albayram is now applying these imaging techniques to patients with diagnosed neurodegenerative disorders. The goal is to determine if the fluid flow along the MMA is visibly altered in patients with Alzheimer’s or mild cognitive impairment.
"A major challenge in brain research is that we still don’t fully understand how a healthy brain functions and ages," Dr. Albayram noted. "Once we understand what ‘normal’ looks like, we can recognize early signs of disease and design better treatments."
The potential for early diagnosis is particularly promising. If MRI can detect a "slowing" of the drainage along the MMA before clinical symptoms of memory loss appear, it could provide a window for preventative interventions. Such interventions might include lifestyle changes, medications to improve vascular elasticity, or even new therapies designed to physically stimulate lymphatic flow.
As the scientific community digests these findings, the MUSC study stands as a testament to the power of interdisciplinary collaboration. By combining NASA’s engineering prowess with Cornell’s cellular imaging expertise and MUSC’s clinical insights, researchers have uncovered a fundamental aspect of human biology that remained hidden in plain sight for centuries. The middle meningeal artery, once viewed as a simple delivery pipe for blood, is now recognized as a vital guardian of the brain’s internal environment.














Leave a Reply