Photosynthetic bacteria, particularly cyanobacteria, are undeniably the architects of our oxygen-rich planet, having fundamentally reshaped Earth’s atmosphere and paved the way for the evolution of complex life. Their role in the Great Oxygenation Event, approximately 2.5 billion years ago, remains one of the most significant transformations in our planet’s history. Now, groundbreaking research from the Institute of Science and Technology Austria (ISTA) has unveiled a remarkable evolutionary twist concerning these vital organisms. A biological system long understood to be involved in DNA segregation has, in an unexpected evolutionary turn, been repurposed to control the very shape of cyanobacterial cells. This discovery, published in the prestigious journal Science, offers profound insights into the dynamic nature of protein systems over evolutionary timescales and sheds new light on the developmental pathways that led to multicellularity in these ecologically crucial bacteria.
Cyanobacteria: Earth’s Ancient Oxygen Factories and Modern Ecosystem Pillars
Cyanobacteria, often referred to as blue-green algae, are a phylum of bacteria that exhibit oxygenic photosynthesis. Their photosynthetic capabilities are not merely a biological curiosity; they represent a pivotal moment in Earth’s biological and geological history. Before the advent of oxygenic photosynthesis, Earth’s atmosphere contained negligible amounts of free oxygen. The proliferation of cyanobacteria approximately 2.5 to 2.3 billion years ago triggered the Great Oxygenation Event (GOE). This period saw a dramatic increase in atmospheric oxygen levels, a phenomenon that was initially toxic to most existing life forms, leading to a mass extinction. However, it also created the environmental conditions necessary for the evolution of aerobic respiration, a far more efficient form of energy production, which in turn enabled the emergence of complex, multicellular organisms, including animals.
Even today, the ecological significance of cyanobacteria cannot be overstated. They are primary producers, contributing substantially to global biomass and playing critical roles in the planet’s carbon and nitrogen cycles. Their adaptability is extraordinary, allowing them to thrive in diverse and often extreme environments, from the frigid expanses of the Arctic to the scorching heat of hot springs, and even colonizing urban surfaces like rooftops and walls.
Among the vast diversity of cyanobacteria, the species Anabaena sp. PCC 7120, commonly known as Anabaena, has served as a model organism for over three decades, providing a rich platform for studying the intricacies of multicellular cyanobacterial biology and evolution. It is within this well-studied organism that scientists have uncovered the surprising evolutionary adaptation of a DNA segregation system.
A Cellular Cinderella Story: From DNA Custodian to Structural Engineer
The research, spearheaded by Benjamin Springstein, a postdoctoral researcher in the group of Professor Martin Loose at ISTA, in collaboration with international partners from the Institut Pasteur de Montevideo (Uruguay), Kiel University (Germany), and the University of Zurich (Switzerland), has revealed a significant evolutionary repurposing. They found that Anabaena, and likely other multicellular cyanobacteria, have undergone a profound evolutionary shift. An ancient protein system, historically understood to be responsible for the accurate segregation of genetic material during cell division, has been re-engineered into a cytoskeleton-like structure. This newly identified function is crucial for determining and maintaining the characteristic shape of cyanobacterial cells.
Understanding Bacterial DNA: The Foundation of Reproduction
At the heart of bacterial life lies the process of cell division, or binary fission. Like all bacteria, Anabaena reproduces by dividing into two daughter cells. This process is critically dependent on the precise replication and distribution of the bacterial genome, ensuring that each new cell receives a complete and functional set of genetic instructions. Bacterial DNA is organized into chromosomes, which are compact structures containing the essential genes required for survival. In many bacteria, including Anabaena, these chromosomes are often present in multiple copies.
Beyond the main chromosomes, bacteria also possess plasmids. These are smaller, circular DNA molecules that carry auxiliary genes, often conferring advantageous traits such as antibiotic resistance or metabolic capabilities. Plasmids are particularly mobile genetic elements; they can be transferred between bacteria, facilitating rapid adaptation and the spread of beneficial genes throughout a population. The efficient segregation of both chromosomes and plasmids is paramount for bacterial viability and propagation.
The ParMR System: An Ancient Legacy Reimagined
Benjamin Springstein’s long-standing fascination with Anabaena led him to delve into its biological and evolutionary intricacies. During the unexpected pause in laboratory work imposed by the COVID-19 pandemic, Springstein dedicated time to an extensive review of scientific literature. It was during this period of deep contemplation that he stumbled upon a "serendipitous observation."
He noted the presence of a system known as ParMR within the chromosomal DNA of Anabaena and some of its close relatives. Conventionally, the ParMR system is associated with plasmid segregation, and it had primarily been identified on plasmids themselves, not integrated into the bacterial chromosome. This atypical localization sparked a hypothesis in Springstein: could this system have evolved to play a role in chromosome segregation instead of, or in addition to, plasmid segregation?
Upon joining ISTA as an IST-Bridge Fellow, Springstein embarked on a series of experiments to rigorously test his hypothesis. The results, however, diverged dramatically from his initial expectations, leading to a discovery that would fundamentally alter the understanding of this ancient bacterial machinery.
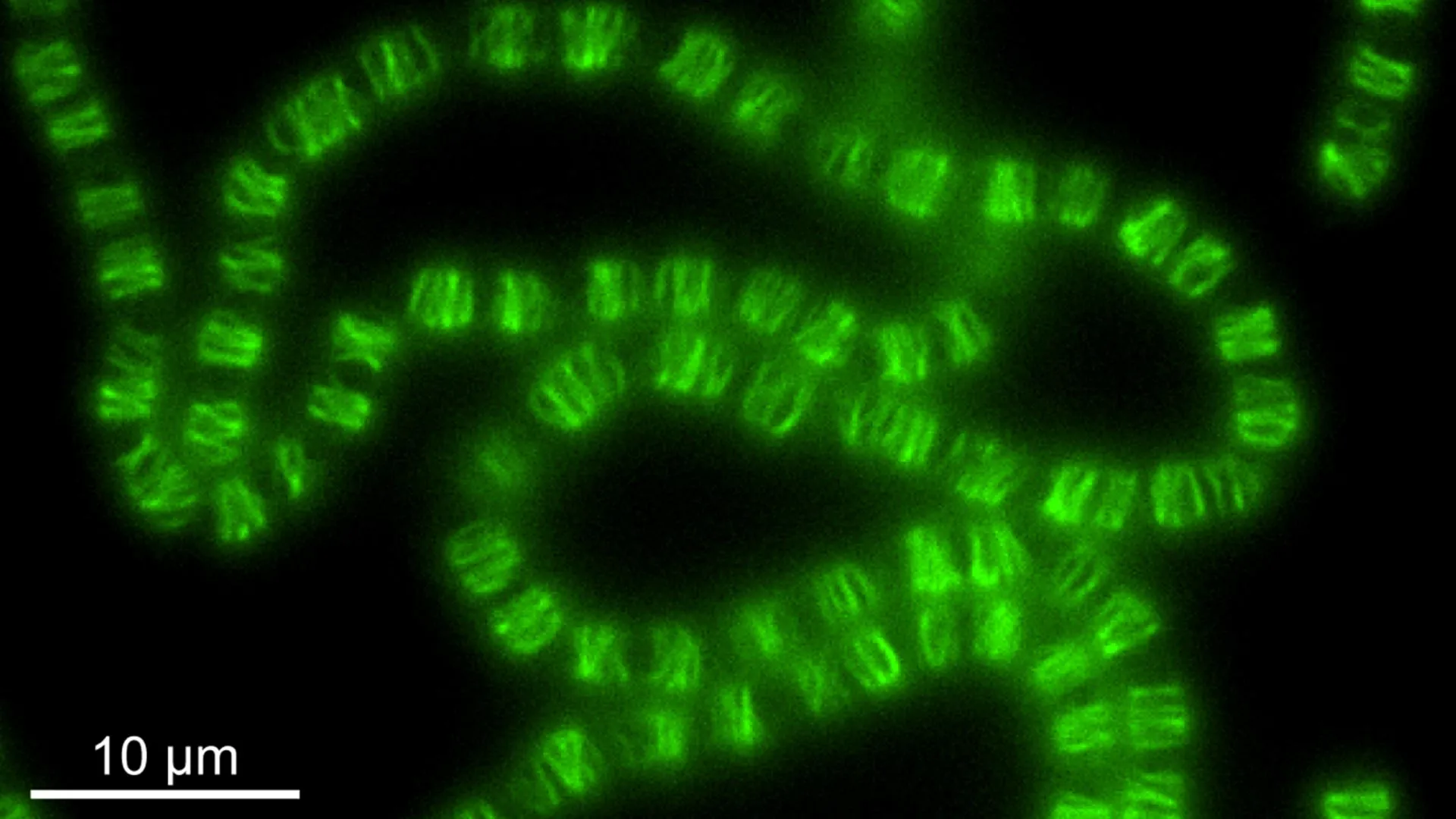
Instead of binding to DNA, one key component of the system, ParR, was found to interact with lipid membranes, particularly the inner membrane of the cell. Concurrently, the other component, ParM, did not assemble into the filament structures typically observed in the cytoplasm for DNA transport. Instead, ParM formed intricate filament networks situated just beneath the inner membrane. These networks created a layer of protein polymers, strikingly resembling a cellular cortex, a structure well-known in more complex eukaryotic cells for providing mechanical support and maintaining cell shape.
The implication was profound: rather than functioning as a typical DNA segregation apparatus that assembles spindle-like structures within the cell’s interior to partition genetic material, this system appeared to be operating at the membrane interface, actively organizing and structuring the cell itself.
Filaments with Cytoskeletal Tendencies: In Vitro Evidence
To elucidate the precise mechanics of this newly identified system, the research team undertook in vitro reconstitution experiments. By purifying the key protein components and assembling them outside of living cells, they were able to observe their behavior in isolation. These experiments revealed that the ParM filaments exhibit a property known as "dynamic instability." This means they undergo cycles of growth and rapid collapse, a characteristic behavior also observed in microtubules, a critical component of the cytoskeleton in eukaryotic cells.
Further detailed structural analysis was conducted in collaboration with ISTA Professor Florian Schur and his PhD student Manjunath Javoor. Employing cryo-electron microscopy (cryo-EM), a technique that allows for atomic-level visualization of biological molecules, the team meticulously examined the architecture of these protein filaments. Their cryo-EM studies uncovered a critical distinction: unlike the polar filaments formed by similar systems in other bacterial contexts, the filaments in Anabaena are bipolar. This bipolarity means they can elongate and retract from both ends simultaneously, a feature that likely contributes to their ability to exert forces and shape the cell membrane.
Loss of Function Reveals a Structural Role: The Cytoskeletal Imperative
The ultimate confirmation of the system’s true function came when it was experimentally inactivated or removed from living Anabaena cells. The phenotypic consequences were stark and unambiguous. "Cells lacking the system lost their normal rectangular-like cell shape and instead became round and swollen," explained Springstein.
These morphological aberrations are highly indicative of compromised cellular structural integrity. In other bacterial species, disruptions to genes responsible for maintaining cell shape typically manifest in such distortions. The observed outcome strongly suggested that the primary role of this system was not the meticulous distribution of DNA, but rather the active control and maintenance of cellular architecture.
Recognizing its novel function and its distinct localization at the cell membrane, the researchers proposed a new designation for this repurposed system: CorMR, a name reflecting its "cortex-like" and membrane-associated role.
Tracing the Evolutionary Trajectory: From Gene Duplication to Structural Innovation
The evolutionary journey of multicellularity in cyanobacteria is a gradual process, marked by the emergence of specialized cell types and coordinated cellular behaviors from single-celled ancestors. To understand how the CorMR system arrived at its current state, bioinformatic analyses were conducted by collaborator Daniela Megrian from the Institut Pasteur in Montevideo, Uruguay.
The transformation of the ParMR system into CorMR was not a single, sudden event. Instead, it appears to have unfolded through a series of incremental evolutionary steps:
- Gene Relocation: The ParMR system, initially residing on plasmids, migrated to the bacterial chromosome. This chromosomal integration likely provided greater stability and a more consistent presence within the cell.
- Component Modification: The protein components, ParR and ParM, underwent structural and functional changes. These alterations likely refined their interaction with membranes and their filament-forming properties.
- Membrane Affinity Development: The system acquired the ability to bind to the cell’s inner membrane, a crucial step in its transition to a structural role.
- Regulatory Integration: The CorMR system became integrated into the cell’s broader regulatory network, likely coming under the control of additional protein systems that fine-tuned its activity.
These sequential evolutionary modifications effectively transformed an ancient mechanism for managing genetic material into a sophisticated system for shaping the cell itself. This remarkable example underscores the profound adaptability of biological systems and demonstrates how evolution can repurpose existing molecular tools for entirely new and critical functions.
Broader Implications: Understanding Multicellularity and Protein System Evolution
The discovery of the CorMR system in Anabaena has far-reaching implications for our understanding of bacterial evolution and the development of multicellularity. It provides a tangible example of how complex cellular structures and functions can emerge from simpler ancestral components. This finding challenges previous assumptions about the conserved nature of DNA segregation systems and highlights the evolutionary plasticity of bacterial protein machinery.
Furthermore, the study offers valuable insights into the origins of cytoskeletal elements in more complex organisms. While the CorMR system is prokaryotic, its functional parallels with eukaryotic cytoskeletal components, such as dynamic filament formation and cellular shape control, suggest potential deep evolutionary connections or convergent evolutionary pathways.
The research also has implications for the study of microbial ecology and biotechnology. A deeper understanding of how cyanobacteria maintain their structural integrity could lead to new strategies for optimizing their growth and productivity in various applications, such as biofuel production or carbon capture.
The collaborative nature of this research, involving multiple international institutions, underscores the power of global scientific cooperation in tackling complex biological questions. The meticulous experimental work, coupled with sophisticated bioinformatic analysis, has not only solved a specific puzzle within Anabaena but has also opened new avenues for investigating evolutionary innovation across the microbial world. The repurposing of the ParMR system into CorMR serves as a potent reminder that the molecular toolkit of life is not static but is constantly being refined, adapted, and reinvented through the relentless force of evolution.















Leave a Reply