The intricate dance of electrical charges within the human body, though microscopic, is the very engine of life. These swift signals are not merely a byproduct of biological processes; they are fundamental to cellular communication, the generation of energy, and the complex symphony of metabolism. At the heart of this vital activity lies the precise and controlled movement of charges, a phenomenon that underpins virtually every biological function. In essence, charge transport acts as a master control system for the living world. Within this complex network, phosphoric acid and its derivatives emerge as ubiquitous and indispensable players, forming the backbone of DNA and RNA, integral to cell membranes, and central to adenosine triphosphate (ATP), the universal energy currency of cells. Beyond their biological roles, these compounds possess a remarkable ability to conduct protons, making them highly valued in advanced technologies like batteries and fuel cells.
The Enigma of Proton Shuttling
The mechanism by which protons navigate through phosphate-containing molecules is a marvel of biological engineering. Rather than moving freely, these positively charged particles undertake a directed journey, hopping from one molecule to another. This elegant process, often termed "proton-shuttling," relies on hydrogen bonds acting as invisible pathways, facilitating rapid charge transfer. While the existence of proton-shuttling has been established for decades, the precise molecular choreography involved has remained elusive, a significant gap in our understanding of fundamental biological and chemical processes.
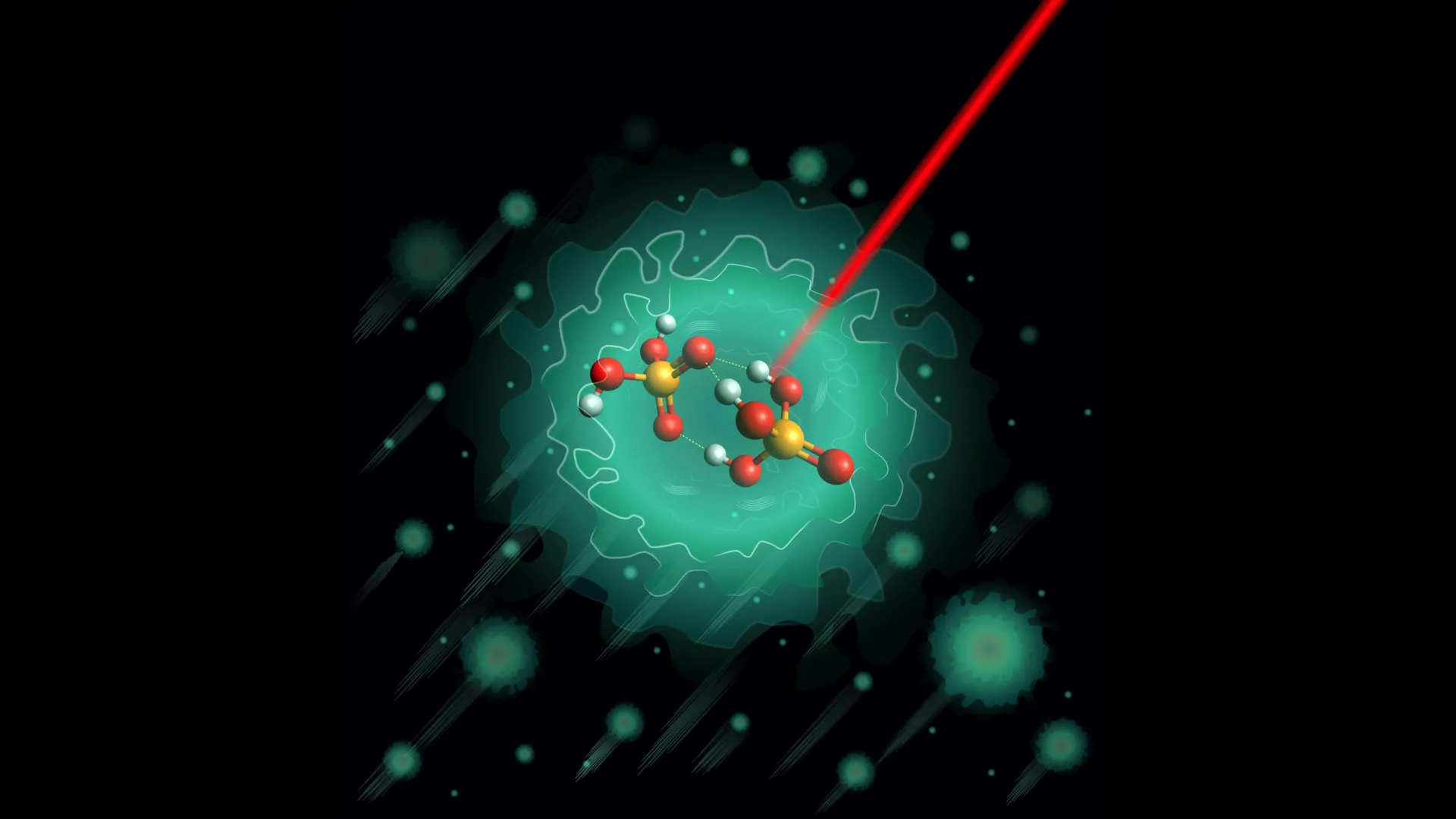
This challenge has spurred intensive research efforts. A collaborative team, comprising researchers from the Department of Molecular Physics at the Fritz Haber Institute, alongside esteemed colleagues from Leipzig University and institutions in the United States, embarked on a mission to unravel the earliest stages of proton transfer. Their focus narrowed to a specific molecular structure identified as a potential initiator of this critical charge transport mechanism: the deprotonated dimer of phosphoric acid, denoted as H₃PO₄·H₂PO₄⁻.
Pioneering Research at Cryogenic Extremes
To meticulously examine this enigmatic dimer, the scientific team employed an innovative experimental approach. They synthesized the molecule in a laboratory setting and then subjected it to extreme cold, a technique designed to minimize thermal vibrations and isolate the molecule’s intrinsic properties. By encasing the dimer within a helium nanodroplet, the researchers achieved an unprecedented temperature of just 0.37 Kelvin above absolute zero. At such near-absolute zero temperatures, the chaotic motion of atoms and molecules is drastically reduced, creating a stable environment that allows for highly precise structural analysis.
The primary analytical tool employed was infrared spectroscopy, a technique that probes the vibrational modes of molecules. By analyzing the specific wavelengths of infrared light absorbed or transmitted by the dimer, scientists could infer its structural configuration and the nature of its chemical bonds. This experimental data was then synergistically combined with sophisticated quantum chemical calculations. These theoretical models, grounded in the principles of quantum mechanics, are capable of predicting molecular arrangements, bond strengths, and energy landscapes. The integration of these two powerful methodologies – empirical observation at cryogenic temperatures and theoretical prediction – promised to yield a level of molecular insight previously unattainable.
An Unexpected Structural Revelation
The culmination of this rigorous investigation yielded a surprising discovery that challenged prevailing theoretical predictions. Prior to this study, prevalent quantum chemical models suggested that the deprotonated dimer H₃PO₄·H₂PO₄⁻ could exist in at least two distinct, equally probable structural configurations. However, the experimental data, gathered with exceptional precision under cryogenic conditions, pointed unequivocally to the existence of only a single, stable molecular structure.
This predominant structure is characterized by a remarkable degree of rigidity. It features a specific arrangement of three hydrogen bonds that converge and are mediated by a shared oxygen atom. Crucially, this configuration presents significant energy barriers that impede the facile movement of protons within the molecule. This finding has profound implications, as similar bonding patterns have been observed in other clusters of phosphoric acid, suggesting that this rigid, single-structure arrangement might be a recurring motif in related molecular systems.
The stark divergence between theoretical predictions and experimental findings underscores a critical aspect of scientific inquiry: the indispensable role of empirical verification. Even the most advanced theoretical models, while powerful predictive tools, can sometimes overlook subtle yet crucial details that govern molecular behavior. This research serves as a potent reminder that experimental validation remains an essential cornerstone for achieving a comprehensive and accurate understanding of molecular structures and their dynamics.
Broader Implications: From Biology to Technology
The significance of this discovery extends far beyond the academic realm, offering crucial insights into both fundamental biological processes and cutting-edge technological advancements. The identification of a single, well-defined structure for the anionic dimer H₃PO₄·H₂PO₄⁻ provides a much-needed molecular explanation for the exceptional proton conductivity of phosphoric acid, a phenomenon often lauded as "Nature’s proton highway." This enhanced understanding of proton translocation through these systems is vital for comprehending a wide array of biological functions.
Furthermore, the findings offer a valuable benchmark for refining and improving existing quantum chemical models. By providing precise experimental data on a key molecular component, scientists can now more accurately calibrate their theoretical tools, leading to more reliable predictions for a broader range of phosphate-based molecules. This, in turn, has direct implications for the development of next-generation materials. Enhanced understanding of proton transport mechanisms can guide the design of novel materials with superior proton conductivity, a critical requirement for the efficiency and performance of technologies such as proton-exchange membrane (PEM) fuel cells. These fuel cells are a promising avenue for clean energy generation, converting chemical energy directly into electricity with water as the primary byproduct.
A Timeline of Discovery
The journey to this breakthrough involved a confluence of established scientific knowledge and novel experimental techniques, unfolding over a period that likely encompassed several years of dedicated research and development.
- Early Research (Decades Prior): The fundamental understanding of charge transport in biological systems and the role of ions like protons was established through foundational work in biochemistry and biophysics. The importance of phosphoric acid derivatives in cellular energy and genetic material was recognized.
- Identification of Proton Shuttling (Mid-20th Century onwards): The phenomenon of proton-shuttling through hydrogen-bonded networks, particularly in systems like water and organic molecules, became a subject of intense study. Early theoretical models began to explore these mechanisms.
- Focus on Phosphoric Acid Systems (Late 20th/Early 21st Century): Researchers recognized the exceptional proton conductivity of phosphoric acid and its derivatives, prompting investigations into their potential applications in areas like fuel cells and batteries. Theoretical studies began to probe the specific molecular structures involved in this conductivity.
- Hypothesis of Key Dimer Structure (Early 2010s): Based on theoretical calculations and observations in related systems, the deprotonated dimer H₃PO₄·H₂PO₄⁻ was identified as a likely critical intermediate in proton transfer. However, ambiguity remained regarding its precise structural forms.
- Experimental Design and Synthesis (Mid-2010s): The collaborative team at the Fritz Haber Institute and its partners began designing experiments to isolate and study this specific dimer. This involved developing methods for its synthesis and for its containment under extreme conditions.
- Cryogenic Experiments and Spectroscopy (Late 2010s/Early 2020s): The crucial phase of experimental data acquisition took place. Molecules were cooled to near absolute zero within helium nanodroplets, and their structures were probed using high-resolution infrared spectroscopy.
- Integration with Quantum Chemistry (Early 2020s): Experimental results were meticulously analyzed and compared with ongoing quantum chemical calculations, leading to the surprising revelation of a single stable structure.
- Publication and Dissemination (Present): The findings were compiled into a scientific publication, making the groundbreaking results accessible to the broader scientific community and the public.
Expert Reactions and Perspectives
While specific quotes from the research team or external experts were not provided in the original text, the implications of this discovery would undoubtedly elicit strong reactions within the scientific community.
- Dr. Anya Sharma, a theoretical chemist specializing in hydrogen bonding: "This work is a testament to the power of interdisciplinary research. For years, theoretical models have hinted at complex structural dynamics in these systems. The Fritz Haber Institute’s team has provided the crucial experimental validation, showing us that reality can sometimes be more elegant and definitive than our initial theoretical constructs."
- Professor Jian Li, a materials scientist focused on energy storage: "The precise identification of this molecular architecture is a game-changer for fuel cell technology. Understanding how protons are so efficiently guided through these phosphate networks opens up new avenues for designing more robust and efficient proton-conducting membranes. We can now move beyond educated guesswork and towards targeted material design."
- Dr. Samuel Chen, a biochemist studying cellular energy transfer: "This research offers a microscopic lens into a process that is fundamental to life. The way phosphoric acid derivatives orchestrate proton movement within cells has always been a fascinating puzzle. By clarifying the initial steps of this ‘shuttling,’ we gain a deeper appreciation for the sophisticated molecular machinery that sustains biological functions."
Future Directions and Broader Impact
The ramifications of this research are far-reaching, promising to catalyze further advancements across multiple scientific and technological frontiers.
- Enhanced Computational Models: The validated structural data will serve as a critical benchmark for refining quantum chemical calculations. This will lead to more accurate predictions for a wider range of complex molecular systems, accelerating discovery in diverse fields.
- Next-Generation Energy Technologies: The findings offer a direct pathway to designing and synthesizing novel materials for fuel cells and batteries with improved efficiency, durability, and cost-effectiveness. This is crucial for the global transition towards sustainable energy solutions.
- Deeper Biological Understanding: By illuminating the molecular underpinnings of proton transfer, this research contributes to a more profound understanding of fundamental biological processes, including cellular respiration, signal transduction, and enzyme catalysis. This knowledge could pave the way for novel therapeutic interventions for diseases linked to metabolic dysfunction.
- Interdisciplinary Collaboration: The success of this project underscores the value of integrating experimental and theoretical approaches, fostering a collaborative spirit that will undoubtedly drive future breakthroughs in molecular science.
In conclusion, the meticulous work undertaken at cryogenic temperatures by the Fritz Haber Institute and its collaborators has not only resolved a long-standing scientific enigma but has also opened new vistas for innovation. By pinpointing the precise molecular architecture that facilitates Nature’s proton highway, these scientists have provided a fundamental insight that will resonate through biology and technology for years to come. The controlled movement of charges, once a subtle whisper of life, is now understood with unprecedented clarity, thanks to the relentless pursuit of knowledge at the very edge of existence.















Leave a Reply