In a landmark study published in the journal Nature on April 15, 2026, a collaborative team of scientists from the Dalian Institute of Chemical Physics (DICP) of the Chinese Academy of Sciences (CAS) and the Southern University of Science and Technology (SUSTech) announced a discovery that challenges five decades of established catalytic theory. Led by Professor Tao Zhang and Professor Yanqiang Huang of DICP, alongside Professor Wei Liu of DICP and Professor Yanggang Wang of SUSTech, the research team has for the first time directly observed and tracked the movement of oxygen atoms within the interior, or "bulk," of a catalyst support material. This phenomenon, known as bulk oxygen spillover, represents a paradigm shift in how scientists understand the interaction between metal nanoparticles and their oxide supports, potentially unlocking the 90% of catalyst volume that was previously considered "dead space" in chemical reactions.
The Science of Spillover: Moving Beyond the Surface
Catalysis is the backbone of the modern industrial world, essential for everything from refining petroleum and manufacturing plastics to converting automotive exhaust into less harmful gases. At the heart of heterogeneous catalysis is the concept of "spillover"—the migration of adsorbed species, such as hydrogen or oxygen atoms, from a metal catalyst particle onto the surface of the supporting material, or vice versa. Traditionally, this process was thought to occur exclusively along the two-dimensional surface of the catalyst.
For decades, the scientific community operated under the assumption that the interior lattice of the support material remained largely inert, serving merely as a structural scaffold for the active surface sites. While surface spillover has been well-documented and utilized to improve reaction rates, the possibility of a "non-surface pathway" involving the bulk of the material remained a subject of intense theoretical debate but lacked empirical proof. The recent findings by the DICP and SUSTech team provide the first visual and measurable evidence that oxygen atoms do not just crawl across the surface; they dive into and emerge from the depths of the crystal lattice, driven by differences in chemical potential.
Experimental Design and the Role of Rutile Titanium Dioxide
The researchers focused their study on ruthenium supported by rutile titanium dioxide (Ru/r-TiO2). Titanium dioxide (TiO2) was selected as the model support due to its exceptional "oxygen reservoir" capabilities. TiO2 is a reducible oxide, meaning it can lose and gain oxygen atoms relatively easily, changing its oxidation state in the process. This flexibility makes it an ideal candidate for studying oxygen transport.
Using ruthenium (Ru), a highly active noble metal, the team created a system where the metal-support interaction could be scrutinized at the atomic level. The choice of the rutile phase of TiO2 was strategic; its specific crystal structure provides well-defined channels that the researchers suspected could facilitate the movement of ions. To observe this at a scale invisible to traditional instruments, the team employed Environmental Transmission Electron Microscopy (ETEM). Unlike standard electron microscopy, which requires a vacuum, ETEM allows scientists to observe catalysts in situ—meaning they can watch the atoms move in real-time while the catalyst is exposed to reactive gases and varying temperatures.
Direct Observation: Tracking Atoms Beneath the Surface
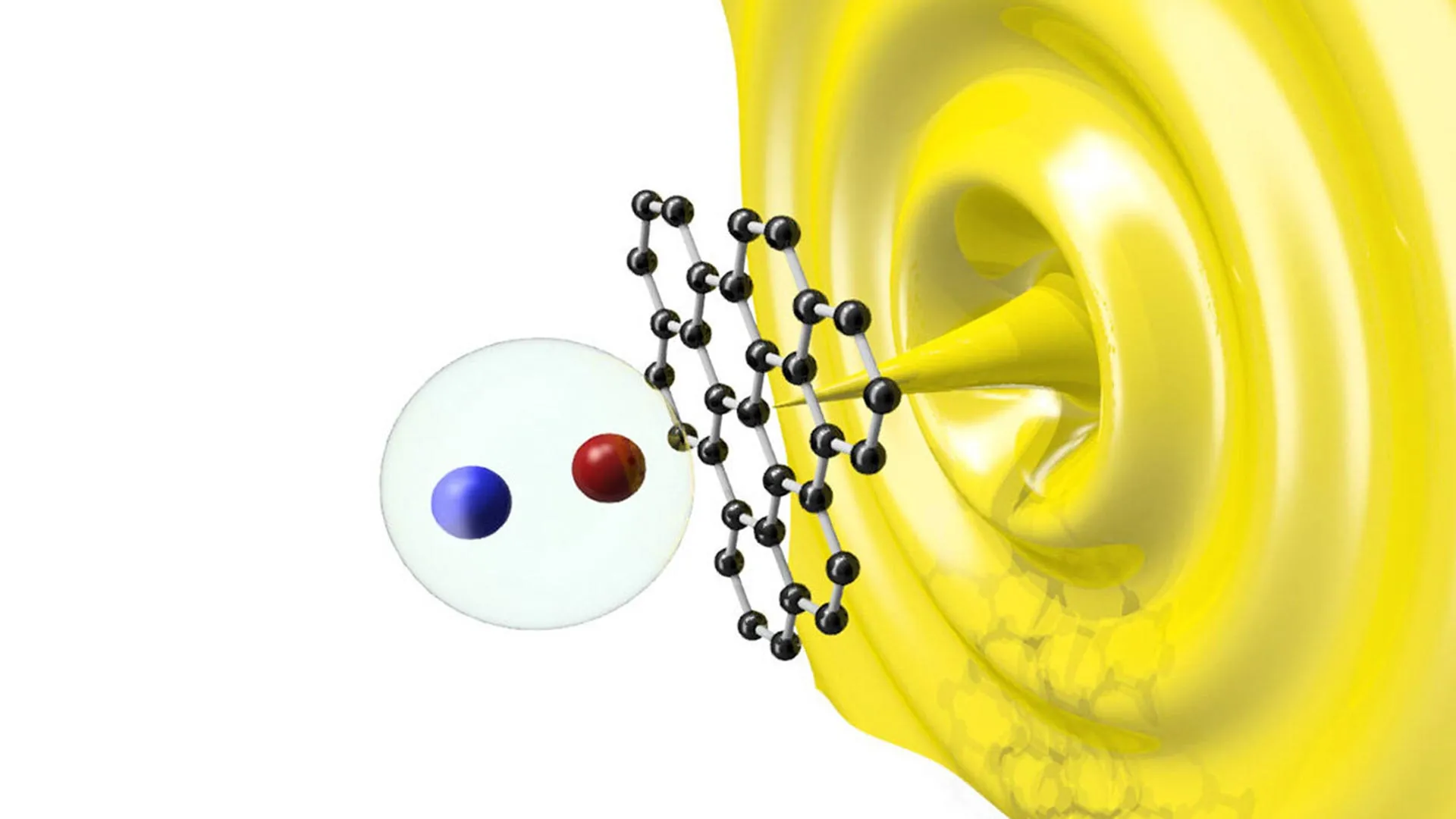
The breakthrough occurred when the team tracked oxygen movement from layers located three to five atoms below the surface of the rutile TiO2 directly to the ruthenium metal interface. This observation proved that the "bulk" of the support was actively participating in mass transfer.
According to the data presented in the study, the metal-support interface acts as an "atomic-scale guard." This interface determines the threshold at which oxygen atoms are permitted to transition from the internal lattice to the active metal site. The researchers found that this movement is not random but is precisely dictated by the oxygen chemical potential gradient. When the metal particle becomes deficient in oxygen during a reaction, it exerts a "pull" that draws oxygen from the interior of the support.
"A channel has been disclosed in the TiO2 support to facilitate oxygen spillover, meanwhile the metal-support interface acts like an atomic-scale guard, controlling whether oxygen spillover can pass through," explained Professor Wei Liu. This discovery effectively redefines the role of the support material from a passive platform to an active participant that can replenish the reaction site from within its own structure.
Historical Context: Expanding the Strong Metal-Support Interaction (SMSI)
To understand the weight of this discovery, one must look back nearly 50 years to the late 1970s. In 1978, S. J. Tauster and his colleagues at Exxon described a phenomenon they called Strong Metal-Support Interaction (SMSI). They observed that when metal catalysts supported on reducible oxides like TiO2 were treated at high temperatures in hydrogen, the support would actually migrate and "creep" over the metal particles, partially burying them. This was long thought to be a surface-dominated interaction that often reduced the metal’s ability to adsorb molecules like carbon monoxide or hydrogen.
For half a century, SMSI was the primary framework for understanding how supports influence metals. However, the DICP study expands this concept into the third dimension. While Tauster’s SMSI focused on the physical encapsulation of the metal by the support surface, the new research shows that there is a deep-seated chemical exchange occurring simultaneously. This "surface-interface-bulk" synergy suggests that the internal regions of the catalyst, which were previously considered inaccessible to reactants, can contribute to the overall catalytic cycle.
Implications for Catalyst Engineering and Industrial Application
The ability to harness the bulk of a catalyst support has profound implications for the design of more efficient and durable catalysts. Most industrial catalysts rely on expensive precious metals like platinum, palladium, and ruthenium. If the support material can be engineered to actively supply oxygen or hydrogen to the metal from its interior, it could allow for the use of smaller amounts of these precious metals while maintaining or even increasing reaction efficiency.
- Green Energy and Hydrogen Production: In the quest for sustainable energy, catalysts are used to split water into hydrogen and oxygen. Understanding bulk spillover could lead to the development of catalysts that are more resistant to "poisoning" or deactivation, as the support can act as a buffer, regulating the supply of reactive species.
- Carbon Capture and Utilization: Converting CO2 into useful fuels requires highly controlled oxygen management. The "atomic guard" mechanism identified by the DICP team offers a new lever for scientists to control the selectivity of these reactions, ensuring that CO2 is converted into the desired hydrocarbons rather than unwanted byproducts.
- Environmental Protection: Automotive catalytic converters and industrial scrubbers rely on oxygen storage capacity. By utilizing the bulk of the support, manufacturers could create smaller, more potent converters that operate effectively even under fluctuating oxygen levels in exhaust streams.
Institutional Collaboration and Future Research Directions
The success of this research is attributed to the multi-disciplinary collaboration between DICP and SUSTech. The Dalian Institute of Chemical Physics has long been a global leader in catalysis, particularly under the guidance of Professor Tao Zhang, whose work in single-atom catalysis has already earned international acclaim. The integration of SUSTech’s advanced theoretical modeling, led by Professor Yanggang Wang, allowed the team to verify their microscopic observations with computational simulations, providing a complete picture of the energetic landscape of bulk spillover.
Professor Yanqiang Huang emphasized that this is only the beginning. "This unique oxygen spillover in our work enables the bulk of a catalyst, which is otherwise inaccessible to reactants, to contribute to mass transfer during catalytic reactions, underscoring the critical importance of interface engineering in controlling spillover behavior."
The next phase of the research will involve testing other metal-oxide combinations to see if bulk spillover is a universal phenomenon. The team aims to develop "practical catalysts" that are specifically designed to maximize this 3D synergy. By engineering the crystal lattice of the support to have more efficient internal "channels," they hope to create a new generation of high-performance materials for the global chemical industry.
Conclusion: A New Dimension in Chemistry
The publication of this study in Nature marks a definitive moment in the history of heterogeneous catalysis. By proving that oxygen spillover is not merely a surface-level event but a deep-reaching process involving the bulk of the material, the team led by Prof. Tao Zhang and Prof. Yanqiang Huang has opened a new frontier in material science. The transition from two-dimensional surface chemistry to a three-dimensional "surface-interface-bulk" model provides a fresh roadmap for interfacial atomic engineering. As the world seeks more efficient ways to manage chemical energy and reduce industrial footprints, the ability to utilize the "useless" interior of a catalyst may prove to be one of the most significant technological leaps of the decade.













Leave a Reply