For over a century, scientists have marveled at the orderly arrangement of mitochondrial DNA (mtDNA) within the cell’s powerhouses, a phenomenon that has long defied a clear explanation. Now, groundbreaking research by a team at the EPFL’s Laboratory of Experimental Biophysics (LEB) has illuminated a previously overlooked biophysical process, "mitochondrial pearling," as the key mechanism responsible for maintaining this crucial cellular organization. This discovery, published in a recent scientific journal, not only deepens our understanding of fundamental cell biology but also holds significant promise for deciphering the origins of a host of debilitating diseases.
The Crucial Role of Mitochondrial DNA and Its Mysterious Organization
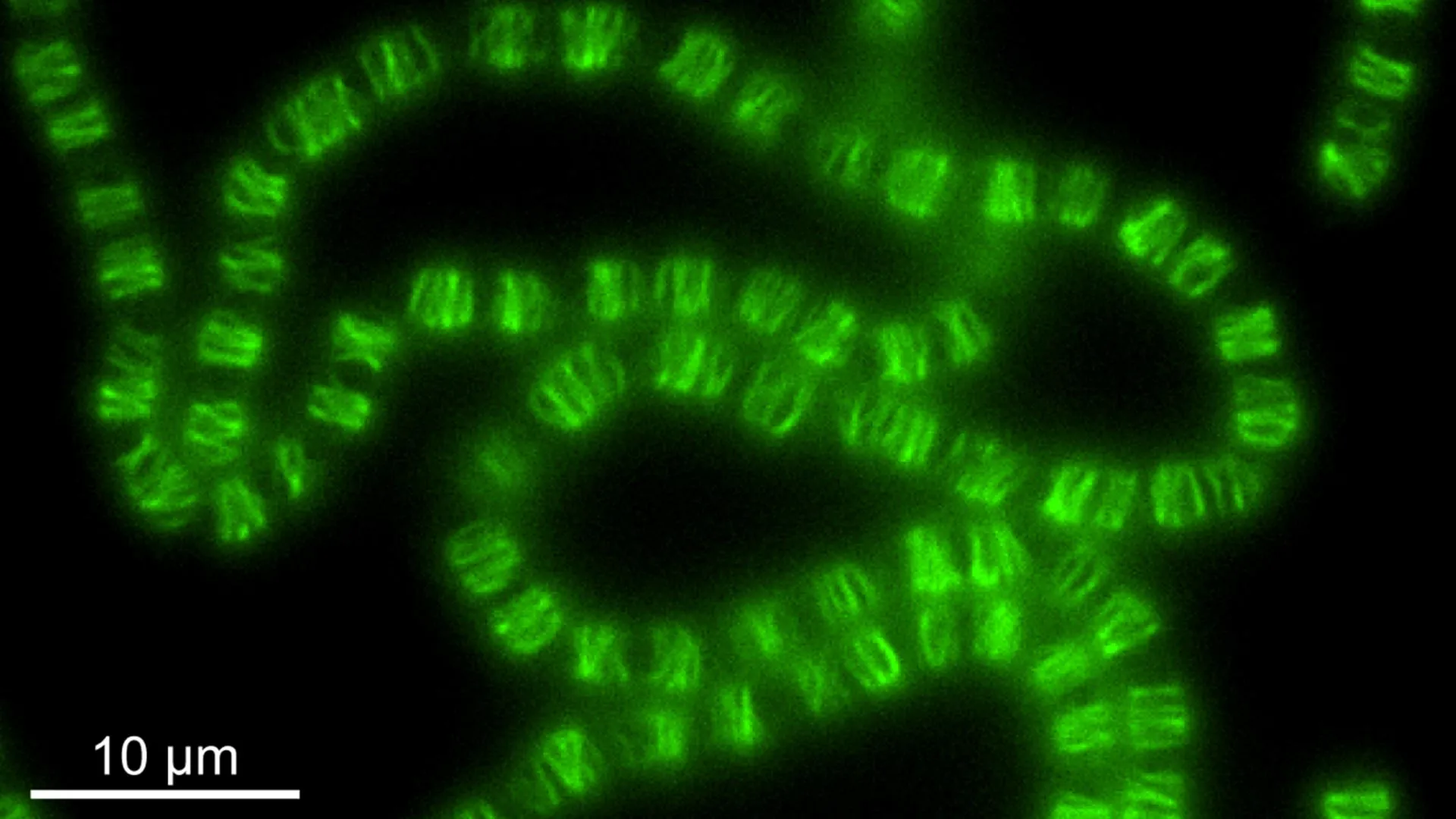
Mitochondria, colloquially known as the "power plants" of the cell, are indispensable organelles responsible for generating the vast majority of the adenosine triphosphate (ATP) that fuels cellular functions. Their ability to perform this vital task is intrinsically linked to their own unique genetic material, mitochondrial DNA (mtDNA). Unlike the nuclear DNA found in the cell’s nucleus, mtDNA exists in multiple copies, typically ranging from hundreds to thousands per cell, and is organized into compact structures called nucleoids.
A persistent observation in cell biology has been the remarkably regular spacing of these mtDNA-containing nucleoids within the mitochondrial network. This precise arrangement is not merely an aesthetic curiosity; it is fundamental to cellular health. It ensures the reliable segregation of mtDNA during cell division, preventing genetic imbalances, and promotes the uniform expression of mtDNA-encoded genes throughout the mitochondrion. When this intricate system falters, the consequences can be severe, leading to a spectrum of metabolic and neurological disorders. Conditions such as liver failure, encephalopathy, and age-related neurodegenerative diseases like Alzheimer’s and Parkinson’s have all been linked to disruptions in mitochondrial function and mtDNA integrity.
Decades of Inquiry and the Elusive Answer
The challenge of understanding how cells achieve and maintain this consistent nucleoid spacing has been a long-standing puzzle. Previous hypotheses centered on dynamic processes like mitochondrial fusion and fission, or on molecular tethering mechanisms. However, these explanations proved inadequate, as nucleoid spacing was observed to persist even when these dynamic processes were experimentally disrupted.
"Proposed mechanisms related to mitochondrial fusion, fission, or molecular tethering cannot explain it, since nucleoid spacing is maintained even when they are disrupted," stated Suliana Manley, a professor at the LEB and a lead author on the study. This persistent enigma prompted the research team to explore alternative explanations, leading them to an unexpected but elegant solution.
Mitochondrial Pearling: A Rediscovered Mechanism Takes Center Stage
The EPFL researchers, led by Professor Manley and postdoctoral fellow Juan Landoni, have now identified mitochondrial pearling as the pivotal mechanism responsible for nucleoid spacing. This process, a transient reshaping of the mitochondrion into a "beads on a string" configuration, had been largely relegated to the footnotes of cell biology literature, often dismissed as a cellular artifact or a response to stress.
During pearling, mitochondria undergo a temporary but dramatic transformation. They form a series of evenly spaced constrictions along their length, creating distinct, bead-like compartments. Crucially, it is within these transiently formed "pearls" that clusters of mtDNA are effectively separated and redistributed. This dynamic redistribution allows the nucleoids to spread out more uniformly, establishing and preserving their characteristic regular spacing.
Illuminating the Process: Advanced Imaging Techniques in Action
To unravel the intricacies of mitochondrial pearling, the research team employed a sophisticated suite of advanced imaging technologies. Their arsenal included super-resolution microscopy, which allows for visualization of cellular structures at unprecedented detail; correlated light and electron microscopy (CLEM), which combines the dynamic insights of light microscopy with the ultrastructural precision of electron microscopy; and phase contrast microscopy, which provides high-contrast images of living cells without staining.
These cutting-edge tools were instrumental in enabling the researchers to track individual nucleoids in real-time, capture the rapid and dynamic changes in mitochondrial shape during pearling events, and gain a deeper understanding of the internal organizational principles at play within these vital organelles. The ability to observe these processes within living cells provided crucial physiological context that had been missing from earlier, static analyses.
The Mechanics of Pearling: From Constriction to Redistribution
The live-cell imaging experiments revealed that mitochondrial pearling is a remarkably frequent occurrence, with events happening several times per minute in healthy cells. During these brief episodes, the mitochondria visibly constrict, forming a series of evenly distributed bulges. The distance between these newly formed "pearls" was found to closely mirror the typical spacing observed between mtDNA nucleoids.
While most of these beaded segments contained a nucleoid situated near their center, the study also noted that pearling structures could form even in the absence of mtDNA. This observation suggests that the physical machinery for pearling is independent of the genetic material itself, but that the process is exquisitely tuned to interact with and organize the nucleoids.
As the pearling process progresses, larger agglomerations of nucleoids were observed to fragment. These smaller nucleoid clusters then migrate and settle into adjacent, newly formed pearls. Upon the mitochondrion’s return to its characteristic tubular shape, the nucleoids remain in their newly segregated positions, thus maintaining the crucial even distribution that had been established during the pearling phase. This dynamic redistribution ensures that even if a mitochondrion undergoes multiple pearling cycles, the overall evenness of nucleoid spacing is preserved.
Unveiling the Regulators: Calcium and Internal Membranes
Beyond describing the morphological changes, the researchers delved into the regulatory mechanisms that drive and control mitochondrial pearling. Through a series of genetic and pharmacological experiments, they identified two key factors that play a significant role.
Firstly, the influx of calcium ions into the mitochondria was found to act as a potent trigger for the pearling process. Calcium signaling is a fundamental cellular communication pathway, and its role here suggests a direct link between cellular metabolic state and the organization of its genetic material.
Secondly, the internal membrane structures within mitochondria were implicated in maintaining the separation of nucleoids. These intricate folds and compartments within the mitochondrion likely provide a physical scaffold or guide for the nucleoids during the pearling process, preventing them from re-aggregating.
When these regulatory factors were experimentally disrupted, the study observed a clear tendency for nucleoids to clump together, failing to achieve the characteristic even spacing. This finding underscores the delicate balance of molecular and physical forces that govern mitochondrial organization.
A Historical Perspective: From Anomaly to Essential Mechanism
The significance of mitochondrial pearling has been dramatically re-evaluated in light of this new research. As Juan Landoni noted, "Since Margaret Reed Lewis first sketched mitochondrial pearling in 1915, it has largely been dismissed as an anomaly linked to cellular stress." For over a century, this striking visual phenomenon was largely ignored or misinterpreted.
The current study elevates pearling from a curiosity to an elegantly conserved and fundamental mechanism at the core of mitochondrial biology. The researchers propose that this biophysical process offers a remarkably simple and energetically efficient means for cells to manage and distribute their mitochondrial genome, ensuring its proper function and inheritance.
Implications for Health and Disease: A New Avenue for Therapeutic Intervention
The implications of this discovery extend far beyond academic curiosity. Understanding the precise mechanisms by which cells organize their mtDNA holds profound potential for addressing a wide range of human diseases. As the research highlights, dysfunction in mitochondria and mtDNA is implicated in numerous serious conditions.
The findings suggest that the cellular reliance on physical processes, alongside complex molecular machinery, for maintaining internal organization is a critical aspect of cell biology. By elucidating the workings of mitochondrial pearling and its regulatory pathways, scientists may gain invaluable insights into the pathogenesis of diseases linked to mitochondrial dysfunction.
This fundamental knowledge could pave the way for novel therapeutic strategies. If conditions like Alzheimer’s, Parkinson’s, and various metabolic disorders are indeed exacerbated or initiated by faulty mtDNA organization, then targeting the pearling mechanism or its regulators could offer entirely new avenues for treatment. Developing interventions that promote or restore healthy pearling could potentially mitigate the accumulation of mtDNA damage, improve mitochondrial function, and ultimately slow or reverse disease progression.
Future Directions and Collaborative Efforts
The research team at EPFL is continuing to explore the nuances of mitochondrial pearling. Future investigations are expected to focus on the specific molecular components that mediate the constrictions and guide nucleoid movement during the process. Furthermore, researchers aim to explore how this mechanism interacts with other cellular processes and how its efficiency might vary across different cell types and physiological conditions.
The collaborative nature of modern scientific discovery is evident in this work, which brings together expertise in biophysics, advanced imaging, and cell biology. The successful resolution of this century-old mystery serves as a testament to persistent scientific inquiry and the power of innovative technological approaches. The journey from a historical sketch to a fundamental biological mechanism underscores the importance of re-examining even long-dismissed phenomena, as they may hold the keys to unlocking some of biology’s most enduring secrets and addressing some of humanity’s most pressing health challenges.















Leave a Reply