The landscape of type 2 diabetes management and cardiovascular risk reduction in the United States has been significantly reshaped with the official launch of Ozempic as an oral pill, a groundbreaking development by pharmaceutical giant Novo Nordisk. This pivotal introduction marks a new era in patient care, offering a convenient and effective alternative to injectable therapies. The oral Ozempic will be manufactured entirely within the U.S. and will be accessible through an extensive network of over 70,000 pharmacies nationwide, ensuring broad availability for patients across the country. This strategic domestic production and distribution plan underscores a commitment to supply chain security and patient access.
The U.S. Food and Drug Administration (FDA) has granted approval for this oral formulation of Ozempic, a glucagon-like peptide-1 (GLP-1) receptor agonist. The medication is designed to work in conjunction with diet and exercise to help adults with type 2 diabetes manage their blood sugar levels and, critically, to reduce their risk of major adverse cardiovascular events (MACE). This dual-action capability positions Ozempic as a vital tool in a comprehensive approach to managing a chronic condition that carries significant cardiovascular comorbidities. The approval signifies a major step forward, providing a non-injectable option for a class of drugs that has demonstrated substantial clinical benefits.
A New Era for GLP-1 Therapy: Oral Availability and Expanded Dosing
The Ozempic oral pill is slated to become available in three distinct dosage strengths: 1.5mg, 4mg, and 9mg, commencing on May 4th. This range of options allows for tailored treatment regimens, catering to the individual needs and responses of patients. What sets this launch apart is its status as the first oral peptide GLP-1 medication approved in the U.S. for both glycemic control and the reduction of cardiovascular risk. This distinction is significant, as it addresses a long-standing preference among many patients for oral administration over injections, potentially improving adherence and overall treatment satisfaction.
Historically, oral semaglutide, the active ingredient in Ozempic, was available under the brand name Rybelsus. However, Rybelsus utilized a different formulation and operated within a distinct dosage range. The rebranding and reformulation under the established Ozempic umbrella are intended to streamline treatment choices for both healthcare providers and patients, fostering greater recognition and simplifying the decision-making process within the therapeutic landscape of type 2 diabetes. This consolidation aims to leverage the strong brand equity of Ozempic and present a unified offering of semaglutide-based therapies.
Domestic Production and Widespread Accessibility
A key element of Novo Nordisk’s strategy for the oral Ozempic launch is its commitment to U.S.-based manufacturing. The entire production process for the oral pill will take place within the United States. This decision not only bolsters domestic pharmaceutical manufacturing capabilities but also offers potential advantages in terms of supply chain resilience and responsiveness to market demand. The drug will be distributed through a vast network encompassing over 70,000 pharmacies across the nation, ensuring that the medication can reach a broad spectrum of the patient population efficiently.
For individuals seeking self-pay options, Novo Nordisk is facilitating access through its NovoCare Pharmacy and select telehealth providers. This marks the first instance of NovoCare offering an oral semaglutide pill for type 2 diabetes management through its platform, further enhancing patient convenience and choice. The availability of these channels is crucial for individuals who may face challenges with insurance coverage or prefer direct access to their medications.
Expanding the Therapeutic Horizon: From Injection to Oral Tablet
Ozempic has been a recognized and trusted injectable medication for type 2 diabetes for the past eight years. Its efficacy in improving glycemic control and its established role in reducing cardiovascular and kidney-related complications, particularly in adults with chronic kidney disease, have solidified its position in the therapeutic arsenal. While the injectable form has a proven track record, the advent of an oral pill significantly broadens its appeal and accessibility. It is important to note that neither the injectable nor the oral formulation of Ozempic is currently approved for use in pediatric populations.
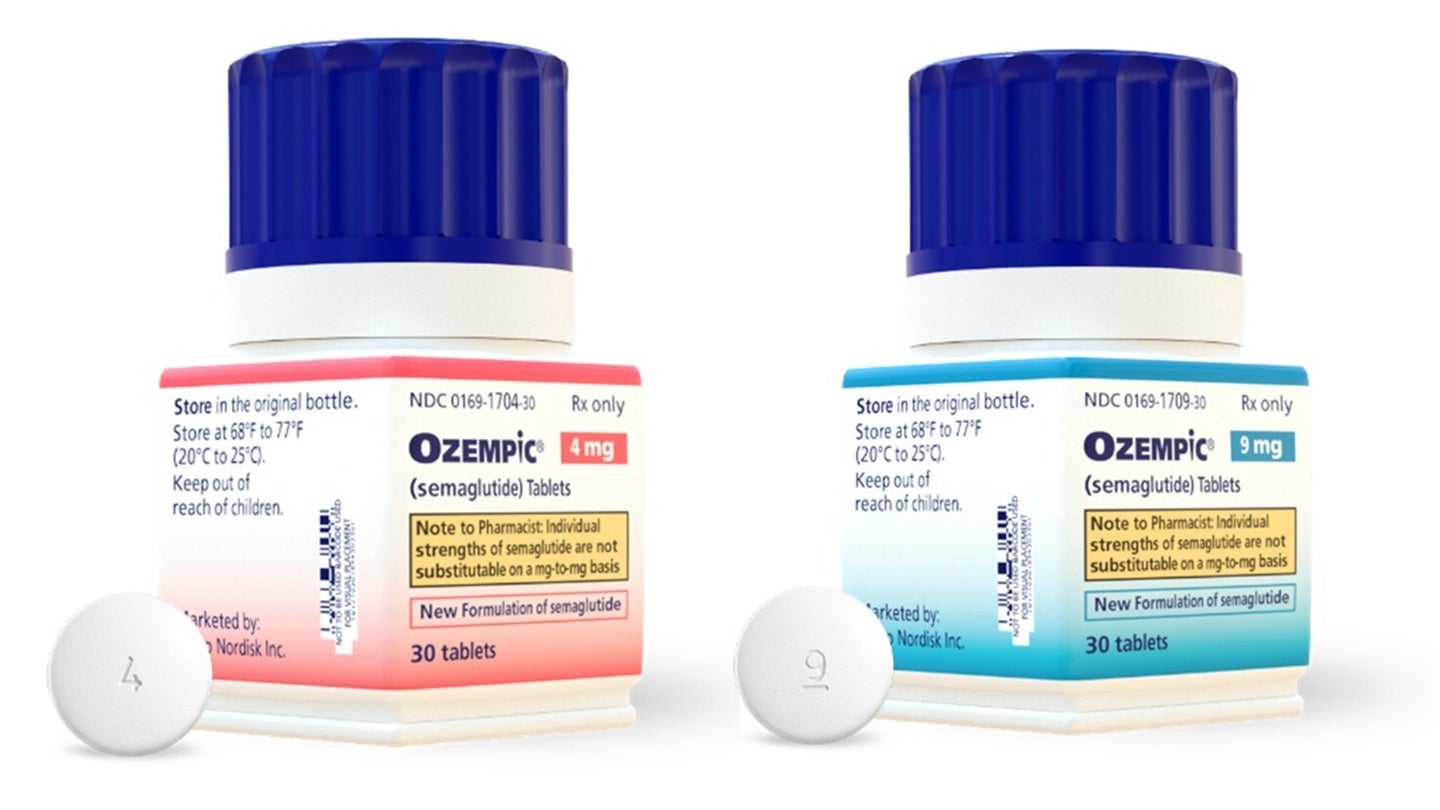
The oral Ozempic tablet contains reformulated strengths of semaglutide, building upon the existing knowledge and clinical experience with the drug. The previous Rybelsus tablets were offered in 3mg, 7mg, and 14mg doses. The strategic rebranding aims to create a more cohesive and recognizable product line for semaglutide, simplifying the selection process for healthcare professionals when choosing the most appropriate treatment for their patients. This move is expected to reduce confusion and facilitate more consistent prescribing patterns.
Addressing Cardiovascular Risk: A Primary and Secondary Prevention Tool
The clinical significance of oral Ozempic extends beyond blood sugar management. It is specifically approved to reduce the risk of major adverse cardiovascular events (MACE) such as heart attack, stroke, or death. This benefit is particularly crucial for adults with type 2 diabetes who also have a high cardiovascular risk profile. The approval encompasses both primary prevention, for individuals who have not yet experienced a cardiovascular event but are at high risk, and secondary prevention, for those who have a history of such events. This broad application underscores the multifaceted benefits of the medication in addressing the complex health needs of individuals with type 2 diabetes.
Affordability and Access: Navigating Prescription Costs
Novo Nordisk is also addressing the financial aspect of medication access. For patients with insurance coverage, the Ozempic pill may be available for as little as $25 for a three-month prescription, making it a more affordable option for many. For those paying out-of-pocket, monthly costs are estimated to range from $149 to $299, depending on the prescribed dosage. These out-of-pocket costs are subject to certain restrictions, highlighting the importance of verifying coverage and understanding program details. The tiered pricing structure aims to balance affordability with the value proposition of the medication.
Future Developments and Strategic Outlook
Looking ahead, Novo Nordisk has submitted an application to the FDA for approval of a 25mg Ozempic tablet. A decision on this higher dosage strength is anticipated by the end of 2026, which could further expand treatment options and potentially cater to patients requiring more intensive glycemic control.
Ed Cinca, Senior Vice President of Marketing and Patient Solutions at Novo Nordisk, expressed enthusiasm for the oral launch, stating, "Ozempic has become a leading, trusted brand for people with type 2 diabetes, and the science is deep. Ozempic pill delivers strong blood sugar control and proven cardiovascular risk reduction, and people may also lose weight." He further elaborated on the patient-centric approach: "With Ozempic now available as both an injection and oral option under one brand, treatment can be individualized so patients are getting the proven semaglutide medicine that best fits their needs." This sentiment emphasizes Novo Nordisk’s commitment to personalized medicine and patient empowerment in managing chronic conditions.
This development follows another significant milestone for Novo Nordisk in March 2026, when the company received FDA approval for a high-dose version of Wegovy (semaglutide), its weight-loss therapy. This parallel advancement in semaglutide-based treatments occurs as patent protections supporting the drug’s market exclusivity are approaching expiry in certain global markets. This strategic timing suggests a concerted effort by Novo Nordisk to solidify its market position and maximize the utility of its semaglutide portfolio across various therapeutic areas.
Broader Implications for Diabetes Care and Public Health
The introduction of an oral Ozempic pill has far-reaching implications for the management of type 2 diabetes and the reduction of cardiovascular disease in the United States. The increased convenience of an oral formulation is expected to improve patient adherence to treatment regimens, a critical factor in achieving long-term health outcomes. Improved adherence can lead to better glycemic control, a reduced incidence of diabetes-related complications, and ultimately, a lower burden of disease on individuals and the healthcare system.
Furthermore, the drug’s proven efficacy in reducing cardiovascular risk addresses a major cause of morbidity and mortality among individuals with type 2 diabetes. By offering a readily accessible option for both blood sugar management and cardiovascular protection, oral Ozempic has the potential to significantly impact public health outcomes. The domestic production strategy also contributes to the economic landscape by supporting U.S. manufacturing jobs and bolstering the domestic pharmaceutical supply chain. As the U.S. population continues to grapple with the growing prevalence of type 2 diabetes and associated cardiovascular diseases, innovations like the oral Ozempic pill represent vital advancements in the ongoing fight against these chronic health challenges. The comprehensive network of pharmacies, coupled with accessible self-pay options, aims to democratize access to this important therapeutic innovation.















Leave a Reply